THE EFFECT OF SELENIUM, GOLD, AND SILVER NANOPARTICLES, AND MONOCLONAL ANTIBODIES ON RECOMBINANT THYROGLOBULIN Tg-cDNA
THE EFFECT OF SELENIUM, GOLD, AND SILVER NANOPARTICLES, AND MONOCLONAL ANTIBODIES ON RECOMBINANT THYROGLOBULIN Tg-cDNA
Abstract
Thyroglobulin, a thyroid-specific protein, and precursor in the synthesis of thyroid hormones has been proposed as a marker of iodine status not only in the population, reflecting thyroid pathologies, but also iodine deficiency in the general population. In most people, it is measured in serum, and serum concentrations increase with an increase in thyroid mass, with thyroid inflammation, and with stimulation of thyroid-stimulating hormone receptors. Due to the large size of the molecule (thyroglobulin), the possibility of obtaining recombinant thyroglobulin is limited. This work aims to use selenium, gold, silver nanoparticles, and monoclonal antibodies to thyroglobulin to create a new generation of test systems. Experimental series of selenium nanoparticles were carried out, and their main characteristics were determined. In addition, an analysis of the antimicrobial activity of selenium nanoparticles was carried out around the disk with selenium nano-particles. The results indicated the size of nanoparticles as 87 and 98 nm, and the peak values of the spectrophotometric analysis of the selenium suspension NPs at 229 and 290 nm respectively. Furthermore, an analysis of the antimicrobial activity of selenium nanoparticles with selenium NPs identified L. monocytogenes inhibition growth at (10.5 mm) and E. coli (10.8 mm) respectively. Interactions of conjugates of mAb to TG with selenium NPs using the ELISA method determined that in a high concentration of selenium NPs in the synthesized conjugate, the optical density of the signal in ELISA increases.
1. Introduction
Thyroid hormones are synthesized by thyrocytes of vertebrates from a large dimeric protein called thyroglobulin, with a monomer molecular weight of 330 kDa and containing about 2750 residues [1]. The source of thyroid hormones in humans is the iodoglycoprotein thyroglobulin, which is the most expressed protein in the thyroid gland. Interestingly, thyroid hormone receptor-mediated bioactivity has been traced back to aquatic lifeforms that predate the first appearance of the thyroglobulin gene [2], [3]. In particular, like a sea urchin [4], [5], and in Amphioxus, many orthologues of the genes involved in the synthesis of thyroid hormones are already present, although these species lack the gene encoding thyroglobulin, similar to vertebrates. Amphioxus, which does not have a follicular structure of the thyroid gland, accumulates iodide and synthesizes thyroid hormone in the pharyngeal endostyle. This organ has functional equivalence to the vertebrate thyroid gland, including the expression of transcription termination factor 1, homeobox NKX2.1, and paired box protein PAX8, which are required for the specification of the thyroid gland [6], [7].
The complete thyroglobulin gene probably first appeared as a result of intragenic duplication and gene fusion [7]. The first incontrovertible evidence of a complete thyroglobulin gene appears with the development of vertebrates [8], and once having appeared, the entire structure encoded by thyroglobulin, was then preserved in all vertebrate. The earliest thyroglobulin-bearing vertebrates that have been studied to date are lamprey larvae, which exhibit exocrine secretion of iodoprotein-containing thyroid hormones from the pharyngeal endostyle before metamorphic transition into the adult lamprey, which has a true endocrine thyroid gland [8]. When comparing the thyroglobulin protein of lamprey and zebrafish, Xenopus and humans, although there are differences, the general modular structures, including the most important cysteine residues forming disulfide bonds, as well as the regional structure of thyroglobulin, are all preserved [9].
Thyroglobulin, a thyroid-specific protein, and precursor in the synthesis of thyroid hormones has been proposed as a marker of iodine status not only in the population, reflecting thyroid pathologies, but also iodine deficiency in the general population [3], [9]. In most people, it is measured in serum, and serum concentrations increase with an increase in thyroid mass, with thyroid inflammation, and with stimulation of thyroid-stimulating hormone receptors. However, the use of thyroglobulin is hindered by differences between assays [10]. While attempts have been made to minimize this through international standardization, significant differences remain. In addition, it is not clear whether a simultaneous assessment of antibodies to thyroglobulin is necessary, given the possible impact of the test on thyroglobulin, in the context of the population [11].
Due to the large size of the molecule, the possibility of obtaining recombinant thyroglobulin is limited. As a result, the only available source of purified human thyroglobulin is thyroid tissue. Unfortunately, the population of human thyroglobulin from tissue donors is heterogeneous in terms of the level of glycosylation and iodination, which creates problems with its use in diagnostics. In addition, different variants of thyroglobulin affect the results of ELISA, which are used to screen for diseases of the cytoid gland [12], [13].
The goal of this research is to study the effects of nanoparticles and monoclonal antibodies on the recombinant thyroglobulin, by creating recombinant thyroglobulin (Tg-cDNA) and inserting it in the microorganisms such as; L. monocytogenes and E. coli. The analysis of the antimicrobial activity of nanoparticles shows the zones of inhibition of the growth and records the results. The tasks of this article are analyzing recombinant thyroglobulin proteins, studying the experimental series of selenium nanoparticles and determination of their main characteristics, an analysis of the antimicrobial activity of selenium nanoparticles to find out their function and sensitivity to nanoparticles, and determining the conjugation of mAb to TG with selenium NPs.
2. Materials and methods
For this research, four pairs of thyroglobulin primers (Е1, Е2, М1 и М2) and the cDNA were used.
Е1 (Forward):5′-GACACAAGGAAGCTAGAGGA-3′, и Е1 (Reverse): 5′-ACAGGCGGGCTGGCTCCT GGTCA-3′. Е2 (Forward): 5′-AGCTGACACAAGGAAGGAAGCTAGAGGA-3′, и Е2 (Reverse): 5′-GA TCACAGGCGGGCTGGCTCCTG GTC-3′. M1 (Forward): 5′-GGGGACCTAGGGCAAGCAG-3′, и M1 (Reverse): 5′-GCCTTTCGGAGGAGGCACAAGAT-3′. M2 (Forward): 5′-AGCTGGGGACCTAGGGC AAGCAG-3′, и M2 (Reverse): 5′- GATCGCTTTCGAGGCACAAGAT-3′.
Moreover, the total volume RNA was isolated from patients venous blood, which was 1ml and obtained from standard venipuncture and transferred immediately into sterile tubes containing TRIzol LS reagent. Therefore, RNA isolated around 100 mg thyroid tissue obtained from patients.
For reverse transcription polymerase chain reaction (RT-PCR), RNA was reverse transcribed to complementary DNA (cDNA), which was made according to the manufacturer's information and instructions. It included 40 μL using 250 ng random hexamer primers (Life Technologies, Inc.), 10 U ribonuclease inhibitor (Life Technologies, Inc.), 200 U Superscript II (Life Technologies, Inc.), 50 mmol/L Tris-HCl (pH 8.3), 75 mmol/L KCl, 3 mmol/L MgCl2, 500 μmol/L of each deoxy-NTP, and 10 mmol/L dithiothreitol.
PCR test: PCR was performed in a total volume of 55 μl. (18.5 µl NFW (Nuclease Free Water), 5 µl Buffer 5x, 0.5 µl dNTP, 0.5 µl Taq polymerase, 1 µl Primers, forward (Forward F.), 1 µl Reverse primers (Revers R.), 1 µl cDNA, 30µl Oil). Then the Samples were subjected to 35 cycles of denaturation (94°C, 1 min), annealing (55°C, 1 min), and elongation (72°C, 1 min). Then 6 µl of the PCR product was mixed with 2 µl of 4* (gel loading dye) and run for 70 minutes on an agarose gel in TAE buffer (24.23 g Tris, 8.96 µl CH3COOH, 1.86 g EDTA, and 73.3 µl H 2 O). The gel was stained with ethidium bromide and bands, amplified fragments, were seen on the UV table. Samples without cDNA were used as negative controls.
Analysis of the obtained sequences of PCR products by sequencing; BioEdit 7.2 (BioEdit - Biological Sequence Alignment Editor - Computer software) is used for editing and analyzing the main sequence. Other analyzes and information are made on the following websites:
genome.eerie.fr, for nucleotide sequence alignment;
- expasy.hcuge.ch, for protein analysis and alignment;
- embl-heidelberg.de, for recognizing repeats in a protein sequence;
- ncbi.nlm.nih.gov, for searching the sequence database.
Nanoparticle (NP) Research
Selenium NP size determination: Using the NANOPHOX PCCS instrument, the size of the NP was determined.
In this study, we used bovine serum albumin (bovine serum) as sodium selenite (Na2SeO3) reducing agent and surface modifier to control Nano-Se formation. The stock solution of 0.1 M Na2SeO3 was prepared by dissolving 0.3459 g of Na2SeO3 powder in 20 µl of H2O. Na2SeO3 was mixed with 20 µl of bovine serum albumin (BSA) and DPBS buffer as stabilizers or closing agents. The mixture was then centrifuged at 3000 rpm for 15 minutes, the supernatant was incubated at 37°C for 5 days, and then the average particle size in the prepared sample was measured using a NANOPHOX instrument.
Study of the antibacterial activity of NPs
Antimicrobial activity was recorded by the presence of transparent zones of no growth around the droplets with the test sample. This experiment was carried out to verify the effect of nanoparticles on the proliferation of bacterial cells, which was carried out on two types of cells (E.coli, & L.Monocytogenes). Which may be used in the treatment of thyroglobulin and thyroid diseases in the future.
Creation of a conjugate of selenium NPs with monoclonal antibody (mAb)
Immobilization of monoclonal antibody (mAb) with nanocarriers was carried out through the method of physical adsorption by mixing mAb directed to human TH and MAb 1 (concentration 2.0 μg/ml in a volume of 1.0 ml) with 1.0 ml of nanoparticles solution at a concentration of 1, 2, 4, 8, 16, 32, 64 μg/ml. Then, it was incubated at +37°C for 16 hours. In this experiment, selenium nanoparticles with average diameters of 87 nm and 98 nm were used as carrier nanoparticles.
3. Results and discussion
The PCR was performed in a total volume of 55 μl. (18.5 µl NFW (Nuclease Free Water), 5 µl Buffer 5x, 0.5 µl dNTP, 0.5 µl Taq polymerase, 1 µl Primers, forward (Forward F.), 1 µl Reverse primers (Revers R.), 1 µl cDNA, 30µl Oil). Then the Samples were subjected to 35 cycles of denaturation (94°C, 1 min), annealing (55°C, 1 min), and elongation (72°C, 1 min). Then 6 µl of the PCR product was mixed with 2 µl of 4* (gel loading dye) and run for 70 minutes on an agarose gel in TAE buffer (24.23 g Tris, 8.96 µl CH3COOH, 1.86 g EDTA, and 73.3 µl H 2 O). The gel was stained with ethidium bromide and bands, amplified fragments, were seen on the UV table. Samples without cDNA were used as negative controls.
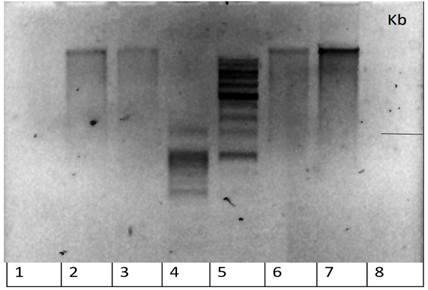
Figure 1 - The results of electrophoresis of RNA samples (primers) from thyroid tissue from 3 patients and 1 donor Lanes:
1, 8 – empty; 2 – sample 1 (patient with DTG) (Dmitrieva); 3 – sample 2 (patient with DTG) (Voronina); 4 – marker 100 b.p.; 5 - marker 1 Kb; 6 - sample 3 (patient with DTG) (Molostova); 7 - sample 4 (healthy donor) (Kuranov)

Figure 2 - Electrophoresis result of PCR - thyroglobulin cDNA with primer M1
Note: samples without cDNA were used as negative controls. In this experiment we applied a lot of protocols from different sources to get PCR results for thyroglobulin; but despite everything, we only got results for one primer of thyroglobulin, which is M1, which was on 940 bp; this result was analyzed and used in the rest of this research work with nanoparticles and their effect on bacteria cells
4. Nanoparticle (NP) Research
Selenium NP size determination: Through NANOPHOX PCCS instrument, the size of the NP was determined.
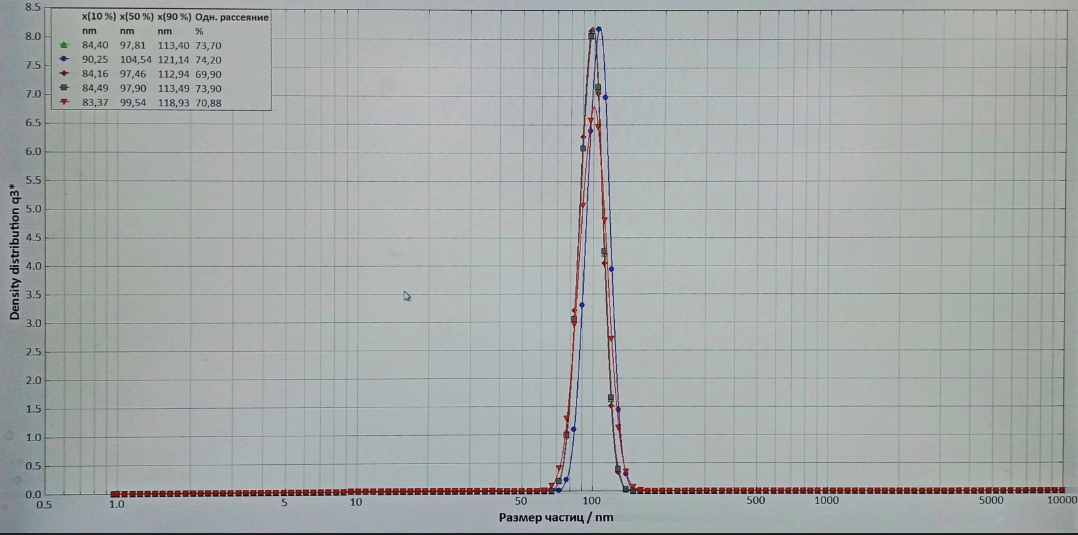
Figure 3 - Absorption spectra of Na2SeO3 nanoparticles of various concentrations
Absorption spectra were measured at room temperature for the visible wavelength in the range of 83.37–121.14 nm for five wavelengths. As it can be seen, the optical density values differ and usually increases with increasing concentration of Na2SeO3 nanoparticles, which indicates the total number of particles in the solution. In addition, the absorption peak shifts towards lower energy or higher wavelengths with increasing nanoparticle size.
Figure 4 - Typical time evolution of the cross-correlation function with lag time -part 1
Figure 5 - Typical time evolution of the cross-correlation function with lag time -part 2
5. Study of the antibacterial activity of NPs
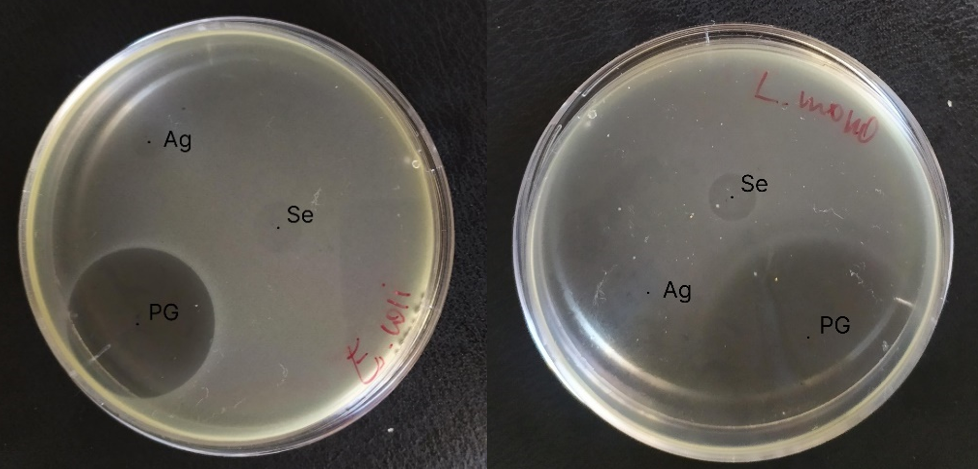
Figure 6 - Result of an E. coli and L. Monocytogenes activity test
Antimicrobial activity was recorded by the presence of transparent zones of no growth around the droplets with the test sample. The larger the diameter of the zone around the disk, the more sensitive the bacterium to test samples.
Table 1 - Diameters of the microbial growth inhibition zones around the discs impregnated with test samples
Test samples/ Microorganism | E. coli (Diameter) | L. monocytogenes (Daiameter) |
Ag nanoparticles | 6,8 | 0 |
Se nanoparticles | 10,8 | 10,5 |
Penicillin G | 33 | 53,8 |
Creation of a conjugate of selenium NPs with monoclonal antibody (mAb):
In this experiment, selenium nanoparticles with average diameters of 87 nm and 98 nm were used as carrier nanoparticles.
Spectral analysis of conjugates of selenium NPs (64 and 2 µg/mL) with mAb to TG:
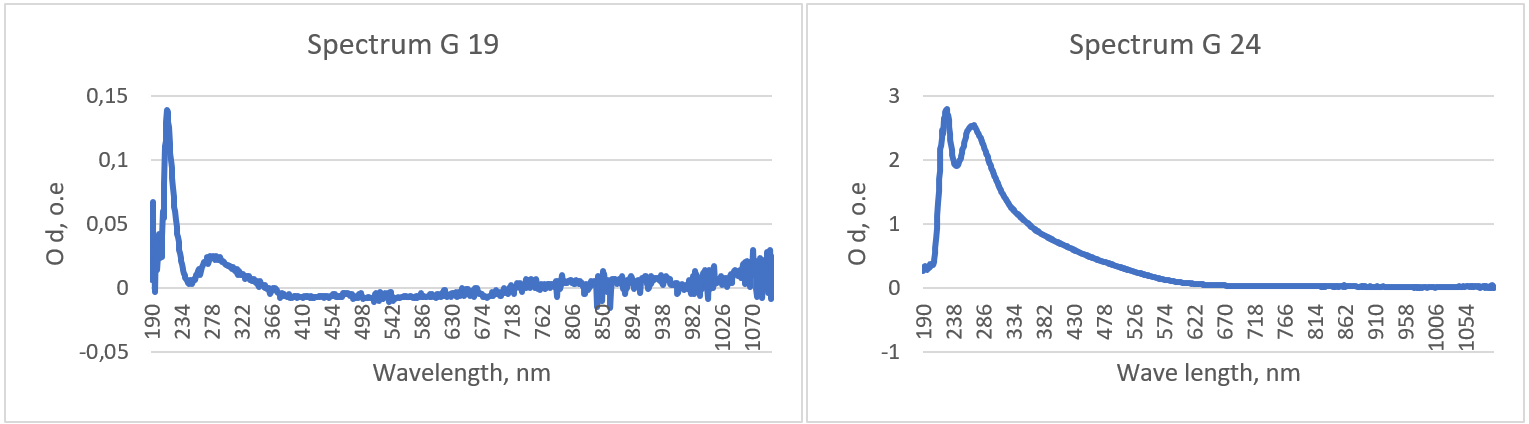
Figure 7 - Characteristic plots of the absorption spectra of conjugates of selenium NPs with mAb to TH
Table 2 - Peak values of the results of spectrophotometric analysis of conjugates of selenium NPs with mAb to TH
NP concentration, µg/ml | Wavelength, nm | Optical density, о. е. |
64 | 228 | 2,799 |
276 | 2,463 | |
2 | 214 | 0,138 |
287 | 0,024 |
6. Conclusion
The expression of recombinant thyroglobulin proteins for the site corresponding to the TG was limited by primers M1 845 bp and studying of experimental series of selenium nanoparticles and their main characteristics indicated the size of nanoparticles as 87 and 98 nm. In addition, the peak values of the spectrophotometric analysis of the suspension selenium NPs were achieved at 229 and 290 nm respectively. Moreover, an analysis of the antimicrobial activity of selenium nanoparticles around the disk with selenium NPs showed that there are zones of inhibition of the growth of L. monocytogenes (10.5 mm) and E. coli (10.8 mm). Microorganisms were also proved to be sensitive to nanoparticles. Conjugating mAbs to TG with selenium NPs determined that the peaks of the absorption spectrum of MAb conjugation to TH with selenium NPs shifted to the right with an increase in the concentration of NPs due to falling at a shorter wavelength. Therefore, with an increase in the concentration of NPs, the value of the OD of the conjugate increases because conjugates with larger NPs have lower OD. Interactions of conjugates of mAb to TG with selenium NPs using the ELISA method determined that at a high concentration of selenium NPs in the synthesized conjugate, the optical density of the signal in ELISA increases. The concentration of NPs in the conjugate 64 μg/ml gives the most intense signal in the ELISA, after which there is a gradual drop when a smaller amount of NPs is added. The conjugates are most stable at NP concentrations of 64 and 32 μg/mL respectively.
