MORPHOMETRY OF THE RESPONSE OF MICE EPIDIDYMAL ADIPOSE TISSUE TO NANOSECOND REPETITIVE PULSED MICROWAVES
Керея А.В.1, Большаков М.А.2, Жаркова Л.П.3, Иванов В.В.4, Князева И.Р.5, Кутенков О.П.6, Ростов В.В.7
1Аспирант, 2Доктор биологических наук, профессор, 3Кандидат биологических наук, Национальный исследовательский Томский государственный университет; 4Кандидат биологических наук, доцент, 5Кандидат биологических наук, доцент, Сибирский государственный медицинский университет; 6Ведущий инженер, 7Доктор физико-математических наук, профессор, Институт сильноточной электроники Сибирского отделения Российской академии наук
Работа выполнена при финансовой поддержке проекта АВЦП № 2.1.1/2777–2.1.1/13778
МОРФОМЕТРИЧЕСКАЯ ОЦЕНКА РЕАКЦИИ ЭПИДИДИМАЛЬНОЙ ЖИРОВОЙ ТКАНИ МЫШЕЙ ПОСЛЕ ВОЗДЕЙСТВИЯ НАНОСЕКУНДНЫМ ИМПУЛЬСНО-ПЕРИОДИЧЕСКИМ МИКРОВОЛНОВЫМ ИЗЛУЧЕНИЕМ
Аннотация
Исследовано влияние ежедневного в течение 10 дней воздействия 4000 импульсов наносекундного импульсно-периодического микроволнового излучения (ИПМИ) c частотами повторения 8–25 имп./с, пиковой плотностью потока мощности (пППМ) 70, 700 и 1500 Вт/см2 на состояние эпидидимальной жировой ткани лабораторных мышей, оцененное по размерам адипоцитов.
Ключевые слова: импульсное микроволновое излучение, размеры адипоцитов.
Kereya A.V.1, Bolshakov M.A.2, Zharkova L.P.3, Ivanov V.V.4, Knyazeva I.R.5, Kutenkov O.P.6, Rostov V.V.7
1 Postgraduate student, 2Doctor of Biological Sciences, Professor, 3PhD in Biological Sciences, National Research Tomsk State University; 4 PhD in Biological Sciences, associate professor, 5 PhD in Biological Sciences, associate professor, Siberian State Medical University, 6 Lead Engineer, 7Doctor of Physical and Mathematical Sciences, Professor, Institute of High Current Electronics Siberian Branch of the Russian Academy of Sciences
MORPHOMETRY OF THE RESPONSE OF MICE EPIDIDYMAL ADIPOSE TISSUE TO NANOSECOND REPETITIVE PULSED MICROWAVES
Abstract
The purpose was to study the effect of 10-day daily exposure of epididymal adipose tissue of mice to repetitive pulsed microwaves (RPM) with a pulse repetition frequency of 8–25 Hz and peak power density of 70, 700, and 1500 W/cm2. The RPM effect was judged from changes in adipocyte sizes.
Keywords: repetitive pulsed microwaves, adipocyte sizes.
The effect of electromagnetic radiation (EMR) on biological objects is defined in many respects by the degree of radiation penetration into an object. The estimates of EMR energy absorption by Schwan [1] demonstrated that the absorbed radiation passes successively through skin, hypodermic fat layer with a low water content, and deeper tissues with a high water content. Because the electrical properties of skin, subcutaneous adipose tissue, and muscles differ greatly, the pattern of EMR penetration into a body is rather complex. The adipose tissue is thus considered as a passive element which allows penetration of more or less energy into a body depending on the EMR frequency and adipose tissue thickness. In particular, the EMR penetration depth for adipose tissue at a frequency of 10 GHz varies between 2 and 4 cm. The so large, though relatively, radiation penetration depth ensures that the radiation can reach the tissues and organs beneath the adipose tissue and affect their state. In the adipose tissue as such, up to 35% of the electromagnetic energy is thus absorbed and converted to heat [2]. Hence, it is not improbable that this tissue can also experience the action of EMR the result of which can be functionally important for the whole organism.
Adipose tissue is a type of connective tissue which has a complex structure and vital functions [3]. Previously, it was thought that adipose tissue cells (adipocytes) form merely passive tissue for energy storage as fat. However, recent studies show strong evidence that adipocytes can act as endocrine and paracrine secretory cells. According to [3, 4], adipose tissue lies in the center of a network of autocrine, paracrine, and endocrine signal systems which cooperate with neuroendocrine regulators. The paths of signal transduction in adipose tissue are formed on a hierarchical principle and represent an important mechanism allowing an organism to adapt to various adverse conditions: starvation, stress, infections, and large excess of energy delivered with food substrates [4, 5, 6]. Epididymal adipose tissue is one of the much used model objects in studies of various properties of adipose tissue, because it makes up the largest portion (45 ± 3%) of the total adipose tissue of an abdominal cavity [7] and allows more accurate weighing due to easy extraction. The morphology of this fat depot is more homogeneous than, e.g., inguinal fat which contains much brown adipose tissue. The superficial localization of epididymal fat, due to its direct accessibility for irradiation and microwave energy absorption, provides a clear advantage in studying the response of adipose tissue to RPM irradiation.
By now, there have been developed new repetitive pulsed microwave sources based on electron accelerators with a short (several to tens of nanoseconds) pulse duration and high peak pulse power. This type of radiation is capable of producing a variety of biological effects in different cells and tissues. In particular, repetitive pulsed microwaves can change the electrical conductivity of hepatocytes and hepatocyte mitochondria [8], affect the level of reactive oxygen species (ROS) with subsequent oxidative modification of lipids and proteins [9], and change the activity of mitochondrial antioxidant enzymes [10]. The irradiation influences the functional state of mitochondria; in particular, some irradiation modes stimulate respiration against the background of reduced coupling of oxidation and phosphorylation and increased expenditure of oxidation substrates [11]. If these processes are possible in adipocytes, they can cause enhanced utilization of fats with respective volume changes in adipose tissue [12].
In the context of the foregoing, it was worthwhile to investigate the effect of nanosecond RPM on epididymal adipose tissue of mice. The main indicator from which to judge the state of fat cells was chosen to be their linear dimensions. This choice was dictated by the possibility of clear and easy observation of the changes occurring in the cells under the action of external factors [13, 14, 15] and by the functional significance of this indicator. As shown earlier [15], the size of adipocytes is an important factor in the production and secretion of adipokines. Moreover, the fat cell size redistribution is closely associated with a change in adipose tissue mass [25]. Reasoning from the above, the objective of the present study was to examine the morphometric characteristics of adipose tissue irradiated by nanosecond RPM.
Materials and methods
Laboratory animals. In the experiments, 108 inbred white male mice of line S57B1/6 weighing 25–30 g were used. The mice were purchased from a nursery of the Institute of Pharmacology (Tomsk Scientific Center, SB RAMS). The animals were kept under standard conditions at constant temperature and humidity with food and drinks available at any time; the light conditions were 12:12. The study was conducted according to the ethical standards for laboratory animals and sanitary rules for facilities, equipment, and maintenance of experimental biological clinics [16]. The animals were divided into two groups: a sham-irradiated group and an irradiated group, with 6 animals for each used irradiation mode.
RPM irradiation. In the study, the sham-irradiated group was subjected to the same manipulations as the irradiated group, except that the microwave generator was off. The experimental animals were daily exposed to 4000 RPM pulses of duration 100 ns with a repetition frequency of 8, 13, 16, 22, and 25 Hz for 10 days. The irradiation was realized at the same time (from 9 to 11 a.m). During the irradiation, the mice were in plastic containers placed at an open waveguide end (of cross-section 10 × 33 mm) to ensure irradiation of their groin where the bulk of epididymal adipose tissue was concentrated. The rest part of their body was covered with radio-absorbing materials to eliminate possible irradiation of the whole body. The exposure time was varied from 3 to 9 min depending on the pulse repetition frequency. The RPM source was a laboratory generator based on a MI-505 magnetron (Russia) with a carrier frequency of 10 GHz, pulse duration of 100 ns at half power level, pulse repetition frequency of 8–25 Hz, and peak power density of 70, 700, and 1500 W/cm2. The peak power density was estimated by the procedure described in detail elsewhere [17]. The intensity of exposure with 70, 700, and 1500 W/cm2 at 8–25 Hz was chosen due to high biological effectiveness of radiation at these parameters, as evidenced earlier by the studies on wound healing [18], mitochondrial respiration [11], and mitochondrial antioxidant enzyme activity [10].
Adipose tissue preparations. Euthanasia of the animals on completion of the full irradiation cycle was by cervical dislocation. For morphometric studies, adipose tissue slices were fixed in 10% neutral buffered formalin (Biovitrum, Russia), dehydrated in IsoPrep isopropyl alcohol solution (Biovitrum, Russia), and immersed in paraffin (Histomix, Russia) according to the procedure [14]. Sections of thickness 5–7 mm were prepared on a MZP-01 microtome (Technom, Russia), mounted on glass slides, and stained with hematoxylin and eosin. The obtained slides were examined on a computer-compatible Biomed-3M transmitted light microscope (Russia). Images of the slices were digitized for subsequent evaluation of adipocyte sizes. The digitized images with clearly defined cell boundaries were subjected to morphometric processing with the ImageJ 1.46 computer program [19]. The sizes of adipocytes were estimated using the point counting method [14, 20] which consisted in measuring the minimum and maximum diameters of cells and finding their half-sum. In total, 324 images were analyzed (18 images per group of 6 mice). For each irradiation mode, the average diameter of 400 fat cells was determined.
Statistical analysis. The RPM effect on adipose tissue was evaluated from variations in the average diameter of adipocytes and their size distribution. The obtained results were subjected to statistical processing in which the arithmetic average of an indicator and its standard error were calculated. The statistical significance of the difference between the indicators of the irradiated and sham-irradiated animals was determined by the Mann-Whitney U-test [21]. In all cases, the P values less than or equal to 0.05 were considered to be statistically significant.
Experimental results
The experiments show that the daily exposure of adipose tissue to 4000 RPM pulses within 10 days affects both the average size of adipocytes and their size distribution. The effect thus depends on the pulse repetition frequency and intensity of exposure.
Average adipocyte size after RPM irradiation. The irradiation causes the average size of adipocytes to change significantly compared to the average size of sham-irradiated cells. The RPM exposure with a peak power density of 70 and 700 W/cm2 at pulse repetition frequencies of 13, 16, 22, and 25 Hz significantly decreased the average adipocyte size (Table 1). The irradiation of adipose tissue with a peak power density of 1500 W/cm2 at a pulse repetition frequency of 22 Hz also decreased the average adipocyte size, whereas that at 13 and 16 Hz increased this parameter (Table 1).
Thus, the change in the average cell diameter was due to the cell size redistribution with attendant increase or decrease in the percentage of both larger and smaller cells (Fig. 1).
Table 1 - Average adipocyte diameter after RPM irradiation of adipose tissue
| Pulse repetition frequency, Hz | Average adipocyte diameter, mm | ||
| RPM, W/cm2 | |||
| 70 | 700 | 1500 | |
| Sham | 47.6±1.58 | 47.6±1.58 | 47.6±1.58 |
| 8 | 45.2±2.05 | 45.1±2.05 | 49±1.59 |
| 13 | 42.6±1.05* | 45.4±1.27 | 54±3.26* |
| 16 | 42.±1.21* | 42.±1.20* | 52±1.61* |
| 22 | 42.9±1.46* | 38.9±0.32* | 41.2±1.89* |
| 25 | 44.3±1.37* | 44.3±1.36* | 45.1±1.57 |
| Note: the presented values are arithmetic mean values ± error of mean; an asterisk stands for a statistically significant difference, р ≤ 0.05, compared to the data for the reference (sham) group. | |||

Fig. 1 - Histological characterization of epididymal adipose tissue after irradiation. Three populations have been distinguished according to their diameter: А – sham-irradiated group – from 41 to 50 μm; B – increase in the number of small cells – less than 41 μm (after exposure with a peak power density of 70 and 700 W/cm2); C – increase in the number of big cells – greater than 50 mm (after exposure with a peak power density of 1500 W/cm2).
Adipocyte size distribution after RPM irradiation. The adipocytes of sham- irradiated animals featured the size ranging from 11 to 100 mm with a clearly defined average of 41–50 μm, which agrees with the available data [22].
The adipocyte size distribution was found to be a more sensitive indicator of the RPM effect than the average cell size. In particular, after irradiation at a frequency of 8 Hz with any peak power density used, a statistically significant difference was observed in the amount of adipocytes of one or another size compared to that in the sham-irradiated group, which escaped detection in estimating the average cell size. The irradiation with a peak power density of 70 W/cm2 at all pulse repetition frequencies caused an increase in the number of small cells and a decrease in that of large cells (Fig. 2).
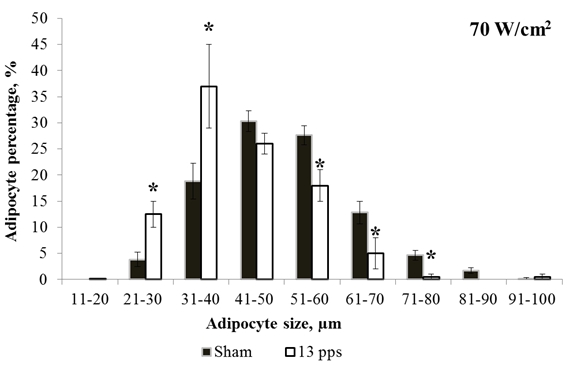
Fig. 2 - Adipocyte size distributions for sham irradiation and irradiation with 70 W/cm2 at 13 Hz. * – statistically significant difference, p ≤ 0.05
A similar change in the cell size distribution was observed on RPM exposure at a peak power density of 700 W/cm2 and pulse repetition frequencies of 16 and 22 Hz. However, the irradiation at 8 Hz decreased the cell percentage with a size of 51–60 mm, whereas that at 13 Hz increased the percentage of cells of average size compared to their percentage in the sham-irradiated group (Fig. 3).
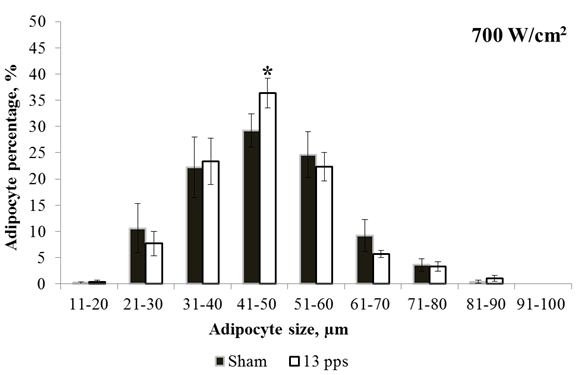
Fig. 3 - Adipocyte size distributions for sham irradiation and irradiation with 700 W/cm2 at 13 Hz. * – statistically significant difference, p ≤ 0.05
The irradiation at 1500 W/cm2 caused the adipocyte size both to increase and to decrease compared to the cell size in the sham-irradiated group. The irradiation of the mice at 8 Hz decreased the cell percentage with a size of 31–40 μm and increased that with a size of 71–80 μm. On irradiation at 13 Hz, the cell percentage with average size decreased and that with a size of 71–80 μm increased (Fig. 4).
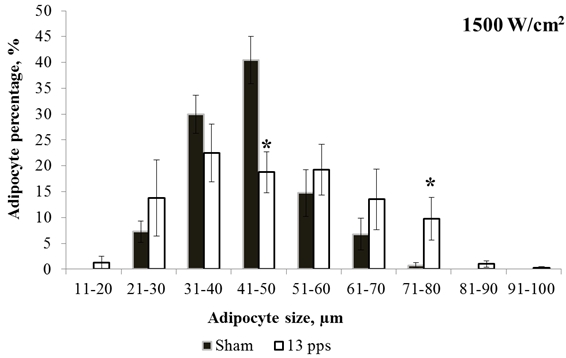
Fig. 4 - Adipocyte size distributions for sham irradiation and irradiation with 1500 W/cm2 at 13 Hz. * – statistically significant difference, p ≤ 0.05
The irradiation at 16 Hz also caused a decrease in cell percentage with a size of 31–40 and 41–50 μm and an increase in that with a size of 61–70 and 71–80 μm. In contrast, the irradiation at 22 Hz increased the cell percentage with smaller and average sizes. The irradiation at 25 Hz decreased the cell percentage with a size of 31–40 μm and increased that with a size of 51–60 μm.
Discussion
The experimental results suggest that nanosecond RPM irradiation affects adipose tissue. The response of the tissue shows up as variation in the average size of adipocytes and their size redistribution. On exposure to nanosecond RPM, the adipose tissue reveals five types of reactions: a shift of the adipocyte size distribution curve toward higher values (70%) and lower values (10%); an increase in cell percentage with a size (average size) characteristic of sham cells (8%); a decrease in cell percentage with an average size (6%); and a decrease in cell percentage with a size smaller than 40 μm (6%). The character of the response depended on the peak power density. At a peak power density of 70 and 700 W/cm2, the response was dominated by a shift of the cell distribution toward smaller sizes (70%). At 1500 W/cm2, the cell size distribution shifted toward both increasing and decreasing sizes. By and large, the variation of the cell size in all exposure modes fit in the variation of the average adipocyte size (Table 1).
The effects of microwave radiation can be both thermal and non-thermal [23]. For this reason, the obtained results were compared with the research data on temperature effects in human fat cells [24]. On heating, adipose tissue cells decrease greatly in size and then die [25]. In view that the response to RPM was dominated by the decrease in cell size, one could admit microwave-induced overheating of fat cells. Therefore, the increase in fat tissue temperature on nanosecond repetitive pulsed irradiation was estimated. Let us consider the electric intensity in a fat layer in air at a power density S = 1500 W/cm2 = 1.5 ∙ 107 W/m2 (in SI units) for complex relative permittivity at f = 1010 Hz [1]. In this case, we have ε1 ≈ 4 and ε2 ≈ 1. Compared to muscular tissue, the reflection coefficient for a normal plane incident wave decreases from ≈ 50% to ≈ 10%. Because of the continuous tangential E field component, the electric intensity E approximates the intensity in air: E ≈ 27 ∙ S1/2 ≈ 105 V/m. The incident microwaves decrease to 1/e =0.368 (i.e., 63% absorbed) with penetration depth Dp which is roughly estimated as:
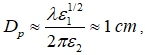
where λ = c/f = 3 cm, c is the velocity of light in vacuum. It is significant that Dp is much (about 8 times) larger than the penetration depth for muscular tissue. In fact, a few millimeters of the entire fat layer may be considered as an optimum thickness to avoid inhomogeneous field distribution. At the same time, deeper penetration means lower values of the specific absorbed rate (SAR) and specific absorbed energy (SAE) per microwave pulse duration t:
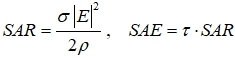
Here σ = 2πε0ε2f is electrical conductivity (ε0 = 8,85∙10-12 F/m), ρ is density (≈ 800 kg/m3). The calculation gives the following maximum specific absorbed rate and energy: SAR ≈ 3 ∙ 106 W/kg and SAE ≈ 0.3 J/kg. So, the increase in temperature for the upper layer is no more than 0.0002°. For a typical repetition frequency of 10 Hz, the average SAR (3 W/kg) hardly looks as thermal exposure. We suppose that the intense microwaves produce an excess number of electrons, protons, and ions near the developed surface of mitochondrial membranes, thus impairing some of the key activities. The specific size of mitochondria measures a few microns. The additional potential can reach several hundreds of millivolt, which seems to be a critical level capable of breaking the normal energy status of membranes. It is conceivable that nanosecond RPM initiates similar processes. As shown earlier, the RPM exposure changes the ROS level (superoxide anion and hydrogen peroxide) in hepatocyte mitochondria and the antioxidant enzyme activity. There is evidence that dysfunction of mitochondria affects the size and differentiation of adipocytes by changing the level of ROS which participate in modulation of the amount of white adipose tissue and inhibition of lipogenesis, thus limiting the development of adipose tissue [26].
In addition, it was demonstrated that secretion of adipocytokines depends on adipocyte sizes [27]. Increasing the adipocyte size increases the production and secretion of anti-inflammatory adipokines by adipocytes. This response of adipose tissue to nanosecond RPM can provide an appropriate change in the body state.
ConclusionThus, results of our study suggest that nanosecond RPM irradiation affects adipose tissue. The response of the tissue shows up as variation in the average size of adipocytes and their size redistribution. Undoubtedly, studying the reactions of adipose cells to nanosecond repetitive pulsed microwaves is important for understanding the general principles and mechanisms of the RPM effects. The obtained results can be of practical significance in terms of hygienic standardization of nanosecond repetitive pulsed microwave radiation and its use in correction of redundant adipose tissue.
References
- Schwan H.P., Foster K.R. Biophysics, 1977. Vol. 17, pp. 193–197.
- Kudryashov Yu.B., Perov Yu.F., Rubyn А.B. Radiation biophysics. RF and microwave electromagnetic radiation. Moscow.: Fizmatlit, 2008. 184 p.
- Gimeno R.E., Klaman L.D. Adipose tissue as an active endocrine organ: recent advance // Current Opinion in Pharmacology, 2005. Vol. 5, Issue 2, pp. 122–128.
- Ahima R.S. Adipose Tissue as an Endocrine Organ // Special Issue: The Neurobiology of Obesity, 2006. Vol. 14, Issue S8, pp. 242S–249S.
- Frigolet M. E., Vela V., Torres N., et al. White Adipose Tissue as Endocrine Organ and Its Role in Obesity // Archives of Medical Research, 2008. Vol. 39, Issue 8, pp. 715–728.
- Gale S.M., Gastracane V.D., Mantzoros C.S. Energy homeostasis, obesity and eating disorders: recent advances in endocrinology // J. Nutr., 2004. Vol. 134, N. 2, pp. 295–298.
- Komarek V. Gross Anatomy // In: Hedrich H.J., Bullock G. The laboratory mouse. Amsterdam, Elsevier, 2004.
- Zharkova L.P., Afanas’ev K. V., Bolshakov M.A., et al. Estimation of the effect of repetitive pulsed X-ray and microwave radiation on biological structures by measuring the impedance characteristics // TSU Vestnik Journal, 2008. No. 312, pp. 180–183.
- Bolshakov M.A., Knyazeva I.R., Rostov V.V., et al. Initiation of free-radical oxidation in albino mice by exposure to pulse periodic microwaves and X-rays // Biophysics, 2005. Vol. 50, Suppl. 1, pp. 104–109.
- Bolshakov M.A., Zharkova L.P., Ivanov V.V., et al. Activity of antioxidant enzymes of liver mitochondria of mice after exposure to nanosecond repetitive pulsed microwaves // TSU Vestnik Journal, 2012. No. 3(19), pp. 122–136.
- Knyazeva I.R., Ivanov V.V., Zharkova L.P., et al. Effect of repetitive pulsed microwaves on functional activity of isolated mitochondria of mice liver // TSU Vestnik Journal, 2011. No. 4 (16), pp.113–123.
- Knyazeva I.R., Bolshakov M.A., Ivanov V.V., et al. Response of mice liver mitochondria to repetitive pulsed microwaves and X-rays // Proceedings of the Universities. Physics, 2012. No. 10/3, pp. 194–199.
- Poglio S., Galvani S., Bour S., et al. Adipose tissue sensitivity to radiation exposure // American Journal of Pathology, 2009. Vol. 174, No. 1, pp. 44–53.
- Osman O.S., Selway J.L., Kępczyńska M.A., et al. A novel automated image analysis method for accurate adipocyte quantification // Adipocyte, 2013. July 1; 2 (3), pp. 160–164.
- Gavrilova Jo, O., Pack S., Jou W., et al. Hypertrophy and/or Hyperplasia: Dynamics of Adipose Tissue Growth // PLoS Comput Biol., 2009. 5(3): e1000324.
- Euro guide on the accommodation and care of animals used for experimental and other scientific purposes. (Based on the revised Appendix A of the European Convention ETS 123) FELASA: Federation of European Laboratory Animal Science Associations, London, UK. 2007. 17 p. www.felasa.eu.
- Knyazeva I.R., Ivanov V.V, Zharkova L.P., et al. Functional state of mitochondria exposed to nanosecond repetitive pulsed microwave // Proceedings of the 9th International Symposium on Electromagnetic Compatibility and Electromagnetic Ecology. St. Petersburg., 2011. PP. 549–553.
- Zharkova L.P., Buldakov M.A., Knyazeva I.R., et al. Sensitivity of some biological objects to repetitive submicrosecond microwave pulses // Journal of Energy and Power Engineering, 2012. V. 6, No. 6, pp. 925–931.
- Collins T.J. ImageJ for microscopy // Biotechniques, 2007. Jul; 43 (1 Suppl): 25–30.
- Avtandilov G.G. Medical morphometry // M.: Medical, 1990. 384 p.
- Efimov V.M., Kovaleva V.Yu. Multivariate analysis of biological data. St. Petersburg, 2008. 86 p.
- Hara-Chikuma M., Sohara E., Rai T., et al. Progressive Adipocyte Hypertrophy in Aquaporin-7-deficient Mice // Journal of Biological Chemistry, 2005. 280, pp. 15493–15496.
- Markov M. Thermal vs. nonthermal mechanisms of interactions between electromagnetic fields and biological systems // Bioelectromagtetics, 2006. 4-15, pp. 3–13.
- Prins J.B., Walker N.I., Winterford C.M., et al. Apoptosis of human adipocytes in vitro // Biochem. Boiphys Res. Commun., 1994. Vol. 201, No. 2, pp. 500–507.
- Ponomareva E.G., Cherkasova O.A., Simonenko G.V., et al. Effect of bacterial lectin and increased temperature on adipocytes // Proceedings of Samara Scientific Center, Russian Academy of Sciences 2012. Vol. 14, No. 1, pp. 283–287.
- Pauw A.D., Tejerina S., Raes M., et al. Mitochondrial dysfunction in adipocyte dedifferentiation and systemic metabolic alterations // American Journal of Pathology, 2009. Vol. 175, No. 3, September. pp. 927–939.
- Skurk T., Alberti-Hube C., Herder C., et al. Relationship between Adipocyte Size and Adipokine Expression and Secretion // Journal of Clinical Endocrinology & Metabolism, 2007. Vol. 92, No. 3, March 1, pp. 1023–1033.
