СИНТЕЗ НАНОПОРОШКОВ ИЗ FE:MGAL2O4 В ЛАЗЕРНОМ ФАКЕЛЕ
DOI: https://doi.org/10.23670/IRJ.2018.74.8.005
СИНТЕЗ НАНОПОРОШКОВ ИЗ FE:MGAL2O4 В ЛАЗЕРНОМ ФАКЕЛЕ
Научная статья
Осипов В.В.1, Соломонов В.И.2, Платонов В.В.3, Тихонов Е.В.4, Медведев А.И.5, Подкин А.В.6, *
1 ORCID: 0000-0002-6637-6241;
2 ORCID: 0000-0003-1868-2647;
3 ORCID: 0000-0001-9730-0402;
4 ORCID: 0000-0001-7766-6343;
5 ORCID: 0000-0002-5375-2324;
6 ORCID: 0000-0001-8809-6155
1, 2, 3, 4, 5, 6 Институт электрофизики УрО РАН, Екатеринбург, Россия
*Корреспондирующий автор (podkin[at]iep.uran.ru)
Аннотация Исследовались особенности получения нанопорошков Fe:MgAl2O4 путём испарения мишени из смеси простых оксидов (Fe2O3, MgO, Al2O3) импульсно-периодическим излучением CO2 лазера с пиковой интенсивностью излучения I=1,6 МВт/см2 и средней мощностью излучения Paver=600 Вт, также волоконным иттербиевым лазером (I=0,4 МВт/см2 и Paver=300 Вт). Показано, что при использовании CO2 лазера полученный нанопорошок имеет удельную поверхность 56 м2/г и содержит в себе две кристаллических фазы - MgAl2O4 (98,2 масс.%) и MgO (1,8 масс.%) с растворёнными в них ионами Fe. При средней мощности излучения 600 Вт производительность получения нанопорошка составила 16 г/час. В случае применения волоконного иттербиевого лазера полученный нанопорошок имеет в 2 раза большую удельную поверхность (105 м2/г) и содержит в себе 4 фазы: MgAl2O4 (67,5 масс.%), γ-Al2O3 (24,8 масс.%), Fe3O4 (3,2 масс.%) и MgO (4,5 вес.%). При этом производительность получения нанопорошка из-за образования на поверхности мишени «леса» из выступов высотой 4÷5мм, покрытых слоем полупрозрачного оплавленного слоя, составила только 2,7г/час. Значительные различия в фазовых составах полученных этими лазерами нанопорошков связывается с большей скоростью охлаждения лазерного факела в случае волоконного иттербиевого лазера. Ключевые слова: шпинель, нанопорошок, газофазный метод, CO2 лазер, волоконный иттербиевый лазер.SYNTHESIS OF FE:MGAL2O4 NANOPOWDERS INTO LASER PLUM
Research article
Osipov V.V.1, Solomonov V.I.2, Platonov V.V.3, Tikhonov E.V.4, Medvedev A.I.5, Podkin A.V.6, *
1 ORCID: 0000-0002-6637-6241;
2 ORCID: 0000-0003-1868-2647;
3 ORCID: 0000-0001-9730-0402;
4 ORCID: 0000-0001-7766-6343;
5 ORCID: 0000-0002-5375-2324;
6 ORCID: 0000-0001-8809-6155
1, 2, 3, 4, 5, 6 Institute of Electrophysics Ural Division of RAS, Yekaterinburg, Russia
* Corresponding author (podkin[at]iep.uran.ru)
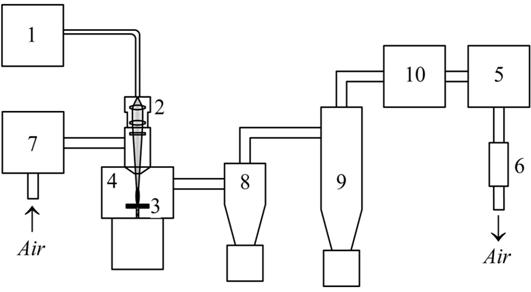
Abstract The features of production of Fe:MgAl2O4 nanopowders by evaporation of targets made from a simple oxide mixture (Fe2O3, MgO, Al2O3) by repetitively pulsed CO2 laser radiation with I=1.6 MW/cm2 peak power density and Paver=600 W average radiation power as well as by ytterbium fiber laser radiation (I=0.4 MW/cm2 and Paver=300 W) were studied. It was demonstrated that the nanopowder produced with the use of the CO2 laser has the specific surface of 56 m2/g and contains two crystalline phases, i.e. MgAl2O4 (98.2 wt%) and MgO (1.8 wt%) with Fe ions dissolved in them. At the average radiation power of 600 W the output of the nanopowder was 16 g/h. For the nanopowder produced using the ytterbium fiber laser twofold increase of the specific surface (105 m2/g) was observed. This nanopowder contains four phases, i.e. MgAl2O4 (67.5 wt%), γ-Al2O3 (24.8 wt%), Fe3O4 (3.2 wt%) and MgO (4.5 wt%). In this case the output of the nanopowder was 2.7 g/h due to formation of a “forest-like” array of 4–5 mm high spikes covered with a semitransparent melt layer. Significant differences in the phase compositions of the nanopowders obtained using these lasers are associated with a higher rate of the laser plume cooling for the ytterbium fiber laser. Keywords: magnesium aluminate spinel, nanopowder, vapor phase method, CO2 laser, ytterbium fiber laser. Introduction Mid-infrared 2–5 μm wavelength range is of considerable interest as it corresponds to the atmospheric window as well as to the absorption maximum of biological tissues (∼2.9 μm). To achieve generation of radiation in this region zinc chalcogenide crystals ZnSe and ZnS doped with transition metal ions (Fe2+, Co2+, Cr2+) are used. Lasing with Cr2+:ZnSe and Cr2+:ZnS crystals was first demonstrated in 1996 [1, P. 885], and in 1999 pulsed generation in 3.98–4.54 μm region was obtained for a Fe2+:ZnSe laser [2, P. 1720]. A promising material for active elements of lasers emitting at 2–5 μm is magnesium aluminate spinel (MgAl2O4) which has the transparency maximum within 2.5–4.5 μm range [3, P. 3341] corresponding to the emission band of Fe2+ doping ion. However, due to high melting temperature of spinel (Tm=2,135 °C) and the difference between the ionic radii of magnesium and iron it is difficult to grow Fe2+:MgAl2O4 monocrystals of high optical quality. To obtain such monocrystals a method of diffusion of iron ions into MgAl2O4 host crystal was employed in [4]. For this purpose an iron film was deposited onto the host crystal with subsequent annealing at high temperature for several days. It was shown that under these conditions the obtained Fe:MgAl2O4 contains iron ions in divalent state and features a broad emission band within 3–6 μm region. A similar approach was used for doping ZnSe polycrystals with iron. In this case the maximum doping depth in ZnSe was achieved with deposition of an iron film with the thickness of ~1 μm on both sides of the sample with subsequent isostatic pressing at 1,270 °C and 100 MPa for 54 hours. The resulting concentration of iron ions on the surface of the sample was 8.6×1018 cm-3 and at the depth of 0.5 mm it was decreased to 4.0×1018 cm-3 [5, P. 211]. To achieve lasing in 3–5 μm region it seems to be reasonable to employ Fe:MgAl2O4 ceramics as an active element. In this case MgAl2O4 can be doped with iron at the stage of the nanopowder production. This fact can significantly improve the situation as it actually provides the solution for the issue of doping uniformity and depth. Production of nanopowders by laser ablation of a target appears to be promising for these purposes. Within this approach the synthesis of nanopowders occurs in the laser plume, i.e. at high temperature with rapid cooling. This provides a decent uniformity of the nanoparticle composition [6, P. 565]. Such nanopowders have the average nanoparticle size of 10–15 nm and narrow size distribution of the nanoparticles (45 nm) [7, P. 116]. The powders produced by the laser synthesis method were vastly employed for fabrication of highly transparent ceramics which provided highly efficient laser performance [8, P. 52], [9, P. 47]. Continuous CO2 laser radiation [10, P. 2561] and repetitively pulsed ytterbium fiber laser radiation [11, P. 472] have already been used to obtain Eu:MgAl2O4 nanoparticles. However, the effect of high transparency of magnesium aluminate spinel at 1.07 μm on the efficiency and productivity of the nanopowder synthesis using ytterbium fiber laser was not estimated in [11, P. 472]. Previously it was shown in [12, P. 716], [13, P. 724] that this effect results in the decrease of the output of the Nd:Y2O3 nanopowder obtained using the ytterbium fiber laser. It should also be noted that, to the best of our knowledge, the synthesis of Fe:MgAl2O4 by the laser method has not been reported. The goal of the present work was to study the features of production of magnesium aluminate spinel Fe:MgAl2O4 nanopowder from a physical mixture of MgO, Al2O3, Fe2O3 powders using a repetitively pulsed CO2 laser (λ=10.6 μm) and an ytterbium fiber (λ=1.07 μm) laser, and also to characterize the obtained nanoparticles. Experimental setup and methods of nanopowder characterization We synthesized the nanopowders of magnesium aluminate spinel doped with iron using a “LAERT” repetitively pulsed СО2 laser and an “LS-07N” continuous-wave ytterbium fiber laser operating in a repetitively pulsed mode. The designs of the experimental setups for the nanopowder synthesis are similar in general. The block diagram of the experimental setup for production of the nanoparticles using the fiber laser is shown as an example in Fig. 1.Fig. 1 – Block diagram of experimental setup for nanopowder synthesis using laser evaporation method
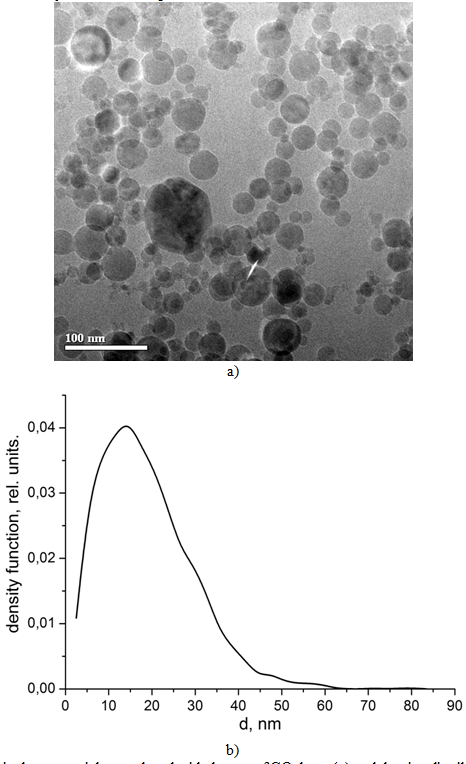
Laser radiation (1) was focused at a target (3) inside an evaporation chamber (4) by an optical head (2). A membrane-based pump was used to pump the air through the system. Volume flow rate of air was measured by a rotameter (6). The input air was purified by a dust filter system (7). The products of target evaporation were first driven by the airflow into a cyclone (8) where large drops and target fragments were separated, and, then, onto a bag filter used for collecting the nanoparticles. Prior to release into the atmosphere the air was finally purified in a filter (10). The conditions for the nanopowder production were close to the optimal ones determined previously for our synthesis of Nd:Y2O3 nanopowder. The targets were evaporated by rectangular radiation pulses from the LS-07N ytterbium fiber laser (l=1.07 μm) with the pulse duration of 120 μs and relative pulse duration of 2. Such pulse duration is insufficient to stimulate spattering of numerous large melt drops from the crater which results in the decrease in the efficiency of Nd:Y2O3 nanopowder production with employment of continuous radiation of the fiber laser [13, P. 724], [14, P. 821]. In this case the average radiation power was 300 W with the peak pulse power of 600 W. The targets were also evaporated by the СО2 laser (l=10.6 μm), which provided laser pulses of a more complex shape [7, P. 116] with the duration of 370 μs (at a level of 0.1), peak power of 8 kW and energy of 1.2 J. At 500 Hz pulse repetition rate the average radiation power was 600 W. The surface of the target was placed into the centre of the waist of the laser beam focused by a KCl or quartz lens. In the case of ytterbium fiber laser the diameter of the focal spot was 430 μm and the radiation power density I in the spot was 0.4 MW/cm2. In the case of the СО2 laser the spot had an elliptic shape with the size of 0.7 × 0.9 mm, and the peak radiation intensity was 1.6 MW/cm2. The mechanism for moving the target provided its motion in such a way that during the evaporation process on average its surface was kept in the plane of the laser beam waist with the laser beam rotating along the spiral at the linear speed of 67 cm/s in the case of the fiber laser and at 35 cm/s in the case of the СО2 laser. The air consumption during the evaporation of the target by each of the two lasers was 6.3–6.6 m3/h; the evaporation chamber was kept at atmospheric pressure (p=0.1 MPa). The targets were fabricated from commercial Al2O3 (purity 99,99%), MgO (purity 99,5%) and Fe2O3 (purity 99,99%) micropowders mixed in the proportion corresponding to the chemical composition of Fe0.034MgAl2.644O5.016 batch mixture. The oxides were mixed for 24 hours and then sintered at T=1,300 °C to accomplish the solid-phase synthesis of spinel. After that the powder was grinded by an LDI-65 disk grinder and pressed in a uniaxial hydrostatic press to obtain cylindrical pellets with the diameter of 65 mm, which were sintered in air at 1,400 °C for 5 hours. Phase compositions of the nanopowders were determined by means of the X-ray phase analysis using a D8 DISCOVER diffractometer (Cu, Kα, λ=0.1542 nm). The processing of the obtained X-ray diffraction patterns was carried out using the program «TOPAS-3» with the Rietveld algorithm for the refinement of the structural parameters. The size of the nanoparticles was obtained by the BET method with employment of a TriStar3000 analyzer. To distinguish the luminogen ions pulsed cathodoluminescence was measured by a CLAVI setup. Results and discussion The particles of the nanopowders obtained by evaporation using the ytterbium and СО2 lasers exhibit almost no agglomeration and their shape is close to spherical. Photographs of the nanoparticles produced by evaporation with the use of the CO2 laser and the size distribution of the nanoparticles are presented in Fig. 2. It can be seen that the majority of the nanoparticles have the diameters ranging from 3 nm to 60 nm. Their average size is 18.7 nm. The specific surface of the nanopowder obtained using the ytterbium laser appears to be twice (105 m2/g) as large as that of the nanopowder produced using the СО2 laser (56 m2/g). This fact indicates that the nanopowder synthesized with employment of the ytterbium laser contains smaller nanoparticles. The average size of these nanoparticles is 10–12 nm. Given that in our experiments the air pressure inside the evaporation chamber was constant the mentioned difference between the average sizes of the nanoparticles is due to the difference in the dynamics of the target evaporation by the lasers and vapor cooling in air. Probably, in the case of the target evaporation by the СО2 laser, the produced nanoparticles remain at high temperature for a longer period.Fig. 2 – TEM images of spinel nanoparticles produced with the use of CO2 laser (a) and the size distribution of nanoparticles (b)
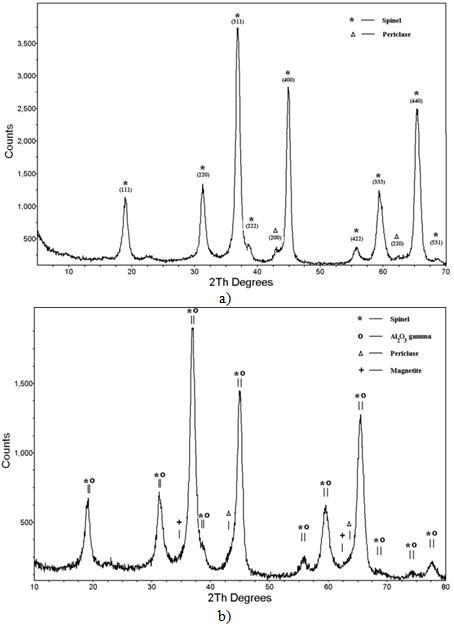
Fig. 3 – X-ray patterns of Fe:MgAl2O4 nanopowders obtained with the use of repetitively pulsed ytterbium fiber laser radiation (a) and pulsed CO2 laser radiation (b)
There are significant differences in other characteristics of the nanopowders as well. The X-ray patterns of the obtained nanopowders are shown in Fig. 3. In both cases the nanopowders include several crystalline phases (see Table 1). There are four phases in the nanopowder produced with the use of the ytterbium laser, i.e. spinel (MgAl2O4), aluminum oxide (γ-Al2O3), periclase (MgO) and magnetite (Fe3O4). The γ-Al2O3 phase contains numerous defects and its structure is similar to the spinel phase. The lattice constant measured for γ-Al2O3 is significantly higher than the value reported in the literature and is close to the lattice constant of MgAl2O4 spinel. It is likely that there are magnesium atoms dissolved in the crystalline lattice of γ-Al2O3, that lead to its distortion. A fraction of magnesium oxide formed MgO in cubic phase and its weight content is 4.5 wt%. Surprisingly iron oxide was not dissolved in the phases considered above and formed a separate magnetite (Fe3O4) crystalline phase with its weight content being higher than the detection threshold (≈1 wt%) and equal to 3.2 wt%. There are only two crystalline phases present in the nanopowder obtained using the СО2 laser. It contains spinel (MgAl2O4) with its structure being distorted and imperfectly ordered, and periclase (MgO). In this case iron oxide was dissolved in these two phases. At the same time the measurements of the pulsed cathodoluminescence spectra in 450–850 nm range revealed that both nanopowders contain Fe3+ included into the spinel structure. These ions provide characteristic wide emission band at l»730 nm. Unfortunately, the emission range of Fe2+ ions (3–6 μm) is beyond the functionality of our detecting instruments and, therefore, it is inconclusive that these ions are present in the obtained spinel powders.Table 1 – Phase composition of spinel nanopowders obtained with the use of ytterbium fiber laser and CO2 laser
| Phase | Weight content, wt% | Size of coherent scattering region, nm | Lattice constant a, Å | Lattice constant taken from JCPDS database, Å |
| Ytterbium fiber laser | ||||
| MgAl2O4 | 67.5 | 9 | 8.08(2) | 8.0831 (PDF # 00-021-1152) |
| γ-Al2O3 | 24.8 | 13 | 8.03(2) | 7.939 (PDF # 00-050-0741) |
| MgO | 4.5 | 5 | 4.15(2) | 4.2112 (PDF # 00-045-0946) |
| Fe3O4 | 3.2 | 2.3 | 8.41(5) | 8.396 (PDF # 00-019-0629) |
| CO2 laser | ||||
| MgAl2O4 | 98.2 | 12 | 8.06(2) | 8.0831 (PDF # 00-021-1152) |
| MgO | 1.8 | 14 | 4.201(7) | 4.2112 (PDF # 00-045-0946) |
Fig. 4 – Target after evaporation by СО2 laser radiation (a) and ytterbium fiber laser radiation (b)
Note: Inset shows magnified image of irregularities formed on the surface of the target during its evaporation by fiber laser
A completely different picture is observed when the target is irradiated by the ytterbium fiber laser. It can be seen from Fig. 4b that unlike the case of evaporation by the СО2 laser the surface of the target after evaporation becomes very irregular and appears as a “forest-like” array of closely spaced sharp spikes with the height up to 4–5 mm. In this case the output of the nanopowder was only 2.7 g/h. This value is approximately 6 times lower than that obtained with the CO2 laser. This difference cannot be exclusively attributed to the difference in the average radiation power of the lasers as in our experiments the average power of ytterbium fiber laser radiation was just two times lower than that of the CO2 laser. Analogously, due to nonuniform evaporation of the target the output of the nanopowder was decreased to 19 wt%. The reduction of the nanopowder output caused by formation of such needle-shaped spikes on the target surface has been observed previously in our experiments on evaporation of Nd:Y2O3 target by continuous radiation from an ytterbium fiber laser [13, P. 724] with the intensity of 0.4–1.2 MW/cm2 in the beam waist. Notably, decreasing the radiation intensity to the level of 0.4 MW/cm2 by expanding the laser spot to the diameter of 430 μm provided a means to reduce the height of the irregularities and increase the average output of the nanopowder from 16 g/h to 23 g/h. Two possible reasons for formation of such structure were proposed, i.e. spattering of numerous liquid melt drops from the crater with the size of these drops being on the order of tens of micrometers and formation of a semitransparent melt layer on the target surface. We consider that the formation of the needle-like spikes is most likely due to high opacity of spinel in the visible and near-IR spectral ranges. In particular, at the wavelength of 1.0 μm the transmittance of a 4-mm-thick ceramic plate of MgO:1.3Al2O3 composition was found to be »83% [15, P. 2887]. Spinel doped with Fe ions is also sufficiently transparent and its refractive index at 1.25 μm is varied within 1–40 cm-1 [16, P. 68] range depending on the iron content. In this case laser radiation is primarily absorbed by different defects in the crystalline structure including color centers or doping ions, with iron ions used for doping in our case. Subsequently, this leads to local heating of the material and reduction of its transparency due to formation of new defects, and, finally, to laser ablation of the material [12, P. 716]. The original target composed of sintered microparticles with the target porosity of »50% reveals a rather high concentration of the defects. Therefore, the penetration depth for the radiation is only several micrometers as similar to the case involving CO2 laser radiation. However, after multiple evaporation, a melt layer which is semitransparent to ytterbium fiber laser radiation is formed on the surface of the target with the layer thickness varying within 25–550 μm range from place to place. Laser radiation is absorbed in either widely-spaced defects in this layer, or in the nonablated layer of the target. Therefore, volume absorption is observed actually. Due to varying thickness of the melted layer, random arrangement of the defects in this layer or different content of Fe ions in different regions, the targets are evaporated at different rates. This results in formation of the irregular surface. Moreover, the growth of the irregularities can be accelerated due to strong reflection of the incident radiation from the inclined sides of the spikes to the valleys between them. As a result, for these valleys the evaporation rate is higher than that for the inclined sides. To understand the reasons responsible for the differences in the phase compositions of the nanopowders obtained with the use of the CO2 and ytterbium fiber lasers let us briefly consider the kinetics of the processes that take place at the stage of the laser plume cooling using the published data [14, P .821], [17, P. 541], [18, P. 134]. The nanoparticles are formed during vapor condensation in the laser plume, which is propagating perpendicularly to the target surface and is gradually cooling [14, P .821], [17, P. 541]. Spectroscopic measurements carried out for the glow of the laser plume formed during evaporation of Nd:YAG and ZrO2:Y2O3 targets by a CO2 laser pulse demonstrate that the vapor temperature in the crater mouth reaches the boiling point of the target material. The temperature decreases unevenly with distance from the target. Vapor condensation starts at 4–5 mm distance from the target, where local increase in temperature due to simultaneous release of the latent heat of evaporation was observed. In these conditions vortical mixing of air and condensing material of the laser plume was observed at the edge of the laser plume and on the top of it facilitating rapid cooling of the plume and, consequently, formation of the nanoparticles. It is known [19] that condensation of a multicomponent vapor starts with condensation of the component having the highest boiling temperature with enrichment of the vapor by high-boiling components. The latter are condensed when the vapor mixture is cooled to the boiling temperature of the corresponding component. This process is slowed down due to vapor heating and formation of the nanoparticles with condensation of the first component. From this point of view, the differences between the thermophysical characteristics of magnesium, aluminum and iron oxides can be one of the key factors affecting the phase composition of the nanoparticles produced in the plume. Magnesium oxide MgO (Tb=3,600 °C) has the highest boiling temperature among the oxides listed above [20]. Therefore, when the laser plume cools, it is MgO vapor that becomes supersaturated first, and condensation centers (i.e. nucleation centers of the nanoparticles) appear. When the temperature of the material in the laser plume is decreased below the boiling temperature of Al2O3 (Tb=2,980 °C), condensation of the vapors of this oxide on the surface of the formed liquid nanoparticles is started. It should be noted that the boiling temperature of Al2O3 is only slightly greater than the melting point of MgO (Tm=2,800 °C). The vapors of iron oxide FeO are the last to condense as it has the minimal boiling temperature (Tb=2,512 °C) among the three oxides. Thus, each subsequent vapor component is condensed on the surface of the nanoparticles at the temperature which is only slightly greater than their melting temperature. In this case interdiffusion of the atoms of the three oxides is required for formation of spinel crystalline structure in the nanoparticles; this process takes some time. The process occurs most efficiently at temperatures close to the melting temperature of the nanoparticles and, as the particles are cooled, the process is significantly slowed down. There is no accurate information on the cooling dynamics for the material that appears in the laser plume during evaporation of the spinel target with the use of the СО2 or fiber laser radiation. However, it is known that the process is very fast and its characteristic time is shorter than 1 ms [17, P. 541]. The presence of multiple phases in the magnesium aluminate spinel nanopowders can be attributed to their “freezing” with rapid cooling of the vapor and nanoparticles in the laser plume. This conclusion is in agreement with the results of paper [11, P. 472] where the presence of MgO crystalline phase in Eu:MgAl2O4 nanopowder obtained with the use of repetitively pulsed radiation of ytterbium fiber laser was ascribed to this effect. The reasoning given above provides a means to qualitatively explain the differences between the phase compositions of the nanopowders obtained using the CO2 and ytterbium lasers by different cooling rates of the plumes formed during evaporation of Fe0.034MgAl2.644O5.016 targets by radiation of each laser. For this purpose it is necessary for the cooling of the material in the laser plume created by the CO2 laser radiation to occur for a longer period than that corresponding to the plume formed during evaporation of the target by the ytterbium laser. It is possible to satisfy this condition as the diameter of the beam waist of the СО2 laser, and, consequently, the diameter of the vapor column of the plume are almost two times as large as the parameters corresponding to the case of the ytterbium laser. In turn, different time of high-temperature exposure of the materials will affect the diffusion rates and diffusion lengths and, consequently, solubility of the particles. In other words, in the case involving the CO2 laser the material in the laser plume is kept at high temperature for a longer time; this fact facilitates the diffusion of Al and Fe atoms in the nanoparticles, i.e. formation of the magnesium aluminate spinel crystalline lattice with Fe ions dissolved in the lattice. Different rates of the laser plume cooling during evaporation of the spinel target by the ytterbium laser and CO2 laser are evidenced by the fact that the size of the nanoparticles obtained in the first case is 1.5 times larger than that obtained in the second one. Indeed, faster rates of the laser plume cooling during evaporation of the target by the fiber laser result in formation of a larger number of nucleating seeds for the nanoparticles. Moreover, the produced nanoparticles are kept in molten state for a shorter time and, consequently, there is less time for their coalescence resulting in appearance of larger particles. This is the reason for formation of the nanoparticles with small average size during the target evaporation by the ytterbium fiber laser. Correlation between the formation of an additional crystalline phase and the average size of the nanoparticles is confirmed by the analysis of the results obtained in the papers [10, P. 2561], [11, P. 472], where Eu:MgAl2O4 were synthesized using continuous radiation of a CO2 laser and repetetively pulsed radiation of an ytterbium fiber laser. Eu:MgAl2O4 nanopowders produced with employment of the fiber laser contained the nanoparticles with the average size of 13 nm [11, P. 472]. In this case, most of the europium oxide formed a separate Eu2O3 crystalline phase in the nanopowder. This phase appears as the ionic radii of Al3+ (51 pm) and Mg2+ (65 pm) are significantly smaller as compared to the value corresponding to Eu3+ ions (95 pm); this fact hinders their diffusion into MgAl2O4 и MgO crystalline lattices. Probably, the time required for the nanoparticle cooling is insufficient for the europium oxide to get dissolved. On the contrary, in the nanopowder synthesized using the CO2 laser [10, P. 2561] the europium oxide was almost completely dissolved in the spinel phase and the average size of the nanoparticles (»30 nm) was greater than that obtained with employment of the fiber laser. This can with great probability be the evidence of slower rate of laser plume cooling in this case as compared to the case involving utilization of the ytterbium fiber laser. To clarify the correctness of the proposed assumptions it is necessary to supplement the mentioned literature data with additional experimental results and the development of a hydrodynamic model describing spattering of the laser plume that takes the appearing vortices into account. Conclusion Thus, the method of laser evaporation of a solid target composed of a mechanical mixture of simple oxides provides a means to obtain the nanopowders of the specified phase composition including those of magnesium aluminate spinel doped with iron ions. As compared to the case of the target evaporation by the ytterbium fiber laser, the process involving the СО2 laser provides achievement of more favorable conditions for production of the nanopowder with the specified phase composition (magnesium aluminate spinel) and higher productivity of the nanopowder synthesis with higher weight output achieved during the evaporation of a single target. The model providing the explanation of the differences in the phase compositions of the nanopowders obtained using these lasers is proposed.| Финансирование Работа выполнена в рамках Государственного целевого проекта № 0389-2014-0027 с частичной финансовой поддержкой Российского фонда фундаментальных исследований по проекту № 17-08-00064 A. | Funding The work was carried out within the framework of the State task project No. 0389-2014-0027 with partial financial support of the Russian Foundation for Basic Research under project No. 17-08-00064 A. |
| Благодарности Авторы благодарны В.В. Лисенкову за полезные обсуждения исследования, Т.М. Деминой за измерения удельной поверхности нанопорошков, О.Р. Тимошенковой и А.М. Мурзакаеву за ТЭМ-изображений наночастиц и В.А. Шитову и К.Е. Лукьяшину за формулировку целей. | Acknowledgement The authors are grateful to V.V. Lisenkov for useful discussions of the research, to T.M. Demina for measuring the specific surface of the nanopowders, to O.R. Timoshenkova and A.M. Murzakaev for TEM imaging of the nanoparticles, and to V.A. Shitov and K.E. Lukyashin for fabrication of the targets. |
| Конфликт интересов Не указан. | Conflict of Interest None declared. |
Список литературы / References
- DeLoach L.D. Transition metal-doped zinc chalcogenides: spectroscopy and laser demonstration of a new class of gain media / L.D. DeLoach, R.H. Page, C.D. Wilke and others // IEEE Journal of Quantum Electronics. – 1996. – № 32. – P. 885-895.
- Adams J.J. 4.0–4.5-µm lasing of Fe:ZnSe below 180 K, a new mid-infrared laser material / J.J. Adams, C. Bibeau, R.H. Page and others // Optics Letters. – 1999. – № 24. – P. 1720-1722.
- Rubat du Merac M. and ohers / Journal of the American Ceramic Society. – 2013. – № 96. – P. 3341.
- Sackuvich R.K. Spectroscopic characterization of Ti3+:AgGaS2 and Fe2+:MgAl2O4 crystals for mid-IR laser applications // R.K. Sackuvich, J.M. Peppers, N.-S. Myoung and others // Proceedings. – 2012. https://doi.org/10.1117/12.909156
- Dormidontov A.E. High-efficiency room-temperature ZnSe:Fe2+ laser with a high pulsed radiation energy / A. E. DormidonovK. N. Firsov, E. M. Gavrishchuk and others // Applied Physics B. – 2016. – № 122 – P. 211
- Garanin S.G. Laser ceramic. 1. Production methods / S. G. Garanin, N. N. Rukavishnikov, A. V. Dmitryuk and others / Journal of Optical Technology. – 2010. – Vol. 77, Issue 9. – P. 77, 565-576.
- Osipov V.V. Laser synthesis of nanopowders / V.V. Osipov, Yu.A. Kotov, M.G. Ivanov and others // Laser Physics. –2006. – Vol. 16, Issue 1. – P. 116–125.
- Osipov V.V. and others / Photonics. – 2017. – № 67.
- Bagayev N. Ho:YAG transparent ceramics based on nanopowders produced by laser ablation method: Fabrication, optical properties, and laser performance / S.N. Bagayev, V.V. Osipov, S.M. Vatnik and others // Optical Materials. – 2015. – Volume 50, Part A. – P. 47-51
- Wenish C. and others / Journal of the American Ceramic Society. – 2016. – № 99. – P. 2561.
- Beketov I.V. Synthesis and luminescent properties of MgAl2O4:Eu nanopowders / I.V. Beketov, A.I. Madvedev, O.M. Samatov and others // Journal of Alloys and Compounds. – 2014. – Vol. 586. – P. 472-475.
- Osipov V.V. Effect of pulses from a high-power ytterbium fiber laser on a material with a nonuniform refractive index. I. Irradiation of yttrium oxide targets / V.V. Osipov, V.V. Lisenkov, V.V. Platonov nd others // Technical Physics. – 2014. – Vol. 59, Issue 5. – P. 716–723.
- Osipov V.V. Effect of pulses from a high-power ytterbium fiber laser on a material with a nonuniform refractive index. II. Production and parameters of Nd:Y2O3 nanopowders / V.V. Osipov, V.V. Lisenkov, V.V. Platonov and others // Technical Physics. – 2014. – Vol. 59, Issue 5. – P. 724–732
- Osipov V.V. Laser plume evolution in the process of nanopowder preparation using an ytterbium fibre laser / V.V. Osipov, V.V. Lisenkov, V.V. Platonov and others // Quantum Electronics. – 2016. – № 46:9. – P. 821–828.
- Krell A. Influence of the structure of MgO· nAl2O3 spinel lattices on transparent ceramics processing and properties/ A. Krell, K. Waetzig, J. Klimke // Journal of the European Ceramic Society. – 2012. – Vol. 32. – P. 2887-2898.
- Zeidler S. and others / Astronomy and Astrophysics. – 2011. – Vol. 526, A68.
- Osipov V.V. and other / Quantum Electron. – 2009. – 39, 541.
- V. Osipov and others / Laser Phys. – 2006. – 16, 134.
- Химическая термодинамика / Пер. с англ. И. Пригожин, Р. Дефэй ; Под ред. В.А. Михайлова // Новосибирск : Наука. – 1966. – 509 с.
- Самсонов Г.В. Физико-химические свойства окислов / Самсонов Г.В., Борисова AJI., Жидкова ТХ. и др. // М.: Металлургия, 1978.- 472 с.
Список литературы на английском языке / References in English
- DeLoach L.D. Transition metal-doped zinc chalcogenides: spectroscopy and laser demonstration of a new class of gain media / L.D. DeLoach, R.H. Page, C.D. Wilke and others // IEEE Journal of Quantum Electronics. – 1996. – № 32. – P. 885-895.
- Adams J.J. 4.0–4.5-µm lasing of Fe:ZnSe below 180 K, a new mid-infrared laser material / J.J. Adams, C. Bibeau, R.H. Page and others // Optics Letters. – 1999. – № 24. – P. 1720-1722.
- Rubat du Merac M. and ohers / Journal of the American Ceramic Society. – 2013. – № 96. – P. 3341.
- Sackuvich R.K. Spectroscopic characterization of Ti3+:AgGaS2 and Fe2+:MgAl2O4 crystals for mid-IR laser applications // R.K. Sackuvich, J.M. Peppers, N.-S. Myoung and others // Proceedings. – 2012. https://doi.org/10.1117/12.909156
- Dormidontov A.E. High-efficiency room-temperature ZnSe:Fe2+ laser with a high pulsed radiation energy / A. E. DormidonovK. N. Firsov, E. M. Gavrishchuk and others // Applied Physics B. – 2016. – № 122 – P. 211
- Garanin S.G. Laser ceramic. 1. Production methods / S. G. Garanin, N. N. Rukavishnikov, A. V. Dmitryuk and others / Journal of Optical Technology. – 2010. – Vol. 77, Issue 9. – P. 77, 565-576.
- Osipov V.V. Laser synthesis of nanopowders / V.V. Osipov, Yu.A. Kotov, M.G. Ivanov and others // Laser Physics. –2006. – Vol. 16, Issue 1. – P. 116–125.
- Osipov V.V. and others / Photonics. – 2017. – № 67.
- Bagayev N. Ho:YAG transparent ceramics based on nanopowders produced by laser ablation method: Fabrication, optical properties, and laser performance / S.N. Bagayev, V.V. Osipov, S.M. Vatnik and others // Optical Materials. – 2015. – Volume 50, Part A. – P. 47-51
- Wenish C. and others / Journal of the American Ceramic Society. – 2016. – № 99. – P. 2561.
- Beketov I.V. Synthesis and luminescent properties of MgAl2O4:Eu nanopowders / I.V. Beketov, A.I. Madvedev, O.M. Samatov and others // Journal of Alloys and Compounds. – 2014. – Vol. 586. – P. 472-475.
- Osipov V.V. Effect of pulses from a high-power ytterbium fiber laser on a material with a nonuniform refractive index. I. Irradiation of yttrium oxide targets / V.V. Osipov, V.V. Lisenkov, V.V. Platonov nd others // Technical Physics. – 2014. – Vol. 59, Issue 5. – P. 716–723.
- Osipov V.V. Effect of pulses from a high-power ytterbium fiber laser on a material with a nonuniform refractive index. II. Production and parameters of Nd:Y2O3 nanopowders / V.V. Osipov, V.V. Lisenkov, V.V. Platonov and others // Technical Physics. – 2014. – Vol. 59, Issue 5. – P. 724–732
- Osipov V.V. Laser plume evolution in the process of nanopowder preparation using an ytterbium fibre laser / V.V. Osipov, V.V. Lisenkov, V.V. Platonov and others // Quantum Electronics. – 2016. – № 46:9. – P. 821–828.
- Krell A. Influence of the structure of MgO· nAl2O3 spinel lattices on transparent ceramics processing and properties/ A. Krell, K. Waetzig, J. Klimke // Journal of the European Ceramic Society. – 2012. – Vol. 32. – P. 2887-2898.
- Zeidler S. and others / Astronomy and Astrophysics. – 2011. – Vol. 526, A68.
- Osipov V.V. and other / Quantum Electron. – 2009. – 39, 541.
- V. Osipov and others / Laser Phys. – 2006. – 16, 134.
- Khimicheskaya termodinamika [Chemical Thermodynamics]/ trans. By I. Prigozhin, R. Defey, ed. by A. Mikhaylov // Novosibirsk : Nauka. – 1966. – 509 p. [in Russian]
- Samsonov G.V. Fiziko-khimicheskiye svoystva okislov [Physicochemical Properties of Oxides] / G.V. Samsonov, A.L. Borisova, T.G. Zhidkova and others // Moscow : Metallurgiya. – 1978. – 472 p. [in Russian]