ТЕХНОЛОГИЯ ЩЕЛОЧНОГО ФОСФОРНОГО УДОБРЕНИЯ НА ОСНОВЕ РАЗЛИЧНЫХ ПРОМЫШЛЕННЫХ ОТХОДОВ
Хузиахметов Р.Х.1, Хузиахметова А.Р.2
1Кандидат химических наук, доцент, 2Ассистент, Казанский национальный исследовательский технологический университет
ТЕХНОЛОГИЯ ЩЕЛОЧНОГО ФОСФОРНОГО УДОБРЕНИЯ НА ОСНОВЕ РАЗЛИЧНЫХ ПРОМЫШЛЕННЫХ ОТХОДОВ
Аннотация
Представлены результаты опытов по переработке низкокачественного Вятско-Камского фосфорита (ФВК) на щелочное фосфорное удобрение с использованием различных промышленных отходов. Установлено, что на основе сульфатно-содовой смеси – ССС (отход глиноземного производства) и органического шлама (отход некоторых производств нефтехимического синтеза) возможно получение термофосфата (ТФ) с содержанием 15-18 % усвояемого Р2О5 [Р2О5(УСВ)].
Ключевые слова: фосфорит, сульфатно-содовая смесь, шлам, продукт переработки шлама (ППШ), термофосфат.Khuziakhmetov R.Кh.1, Khuziakhmetova A.R.2
1PhD in Chemistry, Associate Professor, 2Assistant, Кazan National Research Technological University
THE TECHNOLOGY OF ALKALINE FHOSPHORUS FERTILIZER ON THE BASIS OF VARIOUS INDUSTRIAL WASTE
Abstract
The results of experiments on the processing of low-grade Vyatka-Kama phosphate (VKP) for alkaline phosphoric fertilizer using different industrial wastes are presented in this paper. It was found that it was available to get the thermal phosphate (TP) with the content of 15-18 % of the digestible P2O5 [P2О5(dig)] on the basis of sulfate-soda mixture – SSM (waste alumina production) and organic sludge (waste of some of the industries of petrochemical synthesis).
Keywords: phosphate, sulfate-soda mixture, sludge, sludge recycling product (SRP), thermal phosphate.Introduction
Despite the fact that Russia is one of the world leaders in the production of fertilizers (19,6 million tone NPK– the 4th place after China, USA, India) and cereals (116 million tons of grain), the country's agricultural sector is relatively poorly developed (low yield, high cost production). One of the reasons of low yields (in 2016 – 26,3 centner/ha) is a small quantity of fertilizers applied to the soil – 40 kg/ha (1,9 million tons NPK), because approx.80% oft hem are exported.
The least produced fertilizers are phosphate fertilizers – 3 million tons of P2О5 (N – 8,2 million tons, K2О – 8,4 million tons). The raw material for their production are apatites (the largest deposits of apatite are located in the United States, Brazil, Russia) and sedimentary phosphates (mainly in Morocco – about 64 billion tons). The main problems of production and application of phosphoric fertilizers are the following: monopolization of apatite deposits; continuous price increase for domestic consumers; high transport costs for the transport of concentrate to processing enterprises.
As per above, low cost low-quality phosphates of fields, which are located in agricultural regions, must be involved in the processing. Primarily it is VKP (approx. 50% of the reserves of phosphate, ~ 325 million tons P2О5) [1, P.169].
The most perspective possible way of processing of low-grade phosphate materials is the thermal alkaline processing method, which is suitable for processing of low quality phosphate raw material with high content of Fe2О3 and Al2О3. Special attention is paid for the option of combined thermal alkaline processing with simultaneous utilization of alkaline industrial wastes (e.g., soda-sulfate mixture of alumina production, alkaline liquid combustible organic sludge from some production of petrochemical synthesis, as well as SRP [2].
The aim of this research is to develop the methods for producing alkaline phosphorous fertilizers based on the Vyatka-Kama’s phosphate by using some industrial waste-additives mentioned above (WA).
Material and methods
Vyatka-Kama’s phosphate has been chosen as a phosphorus raw material due to the following main factors: low cost, ease of concentrating the ore by washing (in Р2О5 ore~10-18%, in the concentrate – 20-24%); the impossibility of it’s processing by classic acid method (the value of МFe-Р = Fe2О3/Р2О5 = 0,2 at the maximum allowable value МFe-Р = 0,14).
Used additives-wastes predominantly contain Na2СО3 and Na2SО4 in their composition. Average composition of sulfate-soda mixture (SSM) – the waste of Bogoslavskih aluminum factory (Sverdlovsk region) – is the following: Na2СО3- 20 %, Na2SО4 - 70 %, Al2O3 - 3 %; the sludge of «Nizhnekamskneftehim»: NaOH -10 %, organic combustibles ~ 10 %, SRP: Na2СО3 - 90 %, others are impurities.
Experiments of the TP receiving using the different waste were conducted as per scheme shown in fig. 1, the method of thermal phosphate producing was identical to method of thermal phosphate producing using Na2CO3 [3].
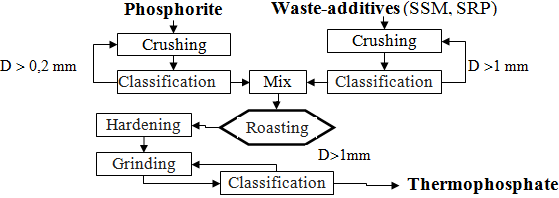
Input parameters were varied in the following range: temperature – 900-1100 degrees; τ =15-60 minutes; the ratio of WA:VKP = 0,2÷1,5 weight; Na2СО3 concentration in the SSM and SRP (and also conditional Na2СО3 in the sludge) – 10-98 %. Output parameters in the experiments were the following: content of total phosphorus (P2О5) and digestible phosphorus [ P2О5(dig)].
The optimization criteria is: К(dig) = (P2О5(dig) / P2О5)×100, %. ResultsCalculation of the mixture of raw materials composition in the studied systems («VKP–sludge», «VKP–Na2CO3», «VKP–SRP», «VKP–SSM») were conducted depending on the state of aggregation of additives (suspension or solid), the content of the reactants (Na2CO3, Na2SO4) in them, the excessive amounts of coal. It was found that at the usage of an initial sludge the ratio of components is:
VKP : sludge = 41 % : 59 % = 1 : 1,45 wt. (NaOH sludge ~11 %).
Thermal phosphate obtaining in the «VKP-Na2CO3» («VKP-SRP») system is based on the assimilable СаNaPO4 formation by the reaction:
Са5F(PO4)3 + 2Na2CO3 + SiO2 = [3СаNaPO4 + Ca2SiO4 + NaF] +2СО2.
Thermal phosphate obtaining in the «VKP-sludge» system during it’s disposal process (by burning «phosphate + sludge» suspension) allows to obtain a finished fertilizer in one step. The burning process of an organic part of the sludge with the formation of Na2CO3 and its interaction with the phosphate occurs simultaneously (SiO2 required as part of VKP):
2NaOH + CO2 = Na2CO3 + Н2O, ΔНР= – 126 кJ;
2NaOH + CO + 0,5О2 = Na2CO3 + Н2O, ΔНР= – 410кJ;
Са5F(PO4)3 + 2Na2CO3 + SiO2 = [3СаNaPO4+Ca2SiO4+NaF] + 2CO2.
Thermal phosphate obtaining in the «VKP-SSM» system is distinguished by the usage of additional coal (for Na2SO4 rehabilitation to Na2O):
Na2SO4 + 2C + 1,5O2 = Na2O + 2CO2 + SO2;
Са5F(PO4)3 + 2Na2O + SiO2 = [3СаNaPO4 + Ca2SiO4 + NaF]
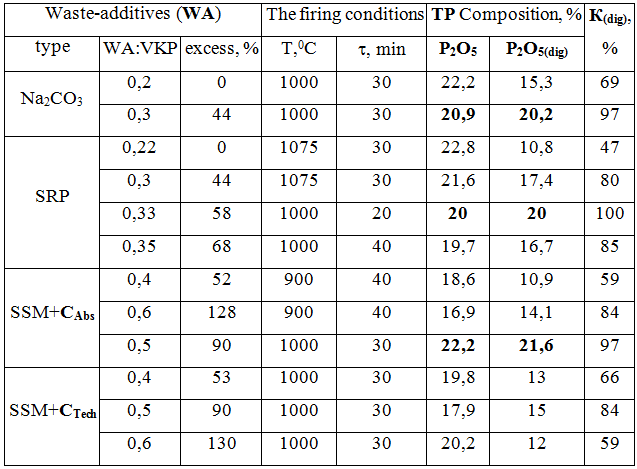
The experiments’ results for obtaining alkaline phosphorous fertilizers based on VPK using the above industrial wastes are presented in table. 1.
Table 1 – Obtaining conditions and the composition of thermal phosphates, obtained using various waste-additives (WA)
Discussion
A method of producing thermal phosphate in the «VKP–sludge» system, despite the reduction of energy consumption is quite difficult because of the low concentration of NaOH and the necessity of evaporating large volume of water (the amount of К(dig) does not exceed 40-50 %).
In the «VKP-SRP» system with a stoichiometric ratio SRP:VKP = 0,22, the experimental degree of К(dig) is very small (due to the occurrence of different reactions with impurities). Digestible form of P2О5(dig) becomes optimally acceptable (К(dig) > 90 %) only with a considerable excess of additives up to 60 %.
Usage of SSM system in the experiments with the absorbent carbon (САbs) under substantially identical optimal firing conditions (1100 degrees, 30 min) the fertilizer with the content of P2О5(dig)~ 20 % has been received (as in the case of Na2СО3). However, when the absorbent carbon is replaced by the technical coal (СТеch) the decrease in the content of P2О5(dig) ~ 16 % can be observed (due to the large amount of ash in the coal ~ 10 %).
Data taken by the X-ray diffraction analyses shows (fig. 2) that thermal phosphates compositions derived on basis of Na2СO3 and Na2SO4 are identical.
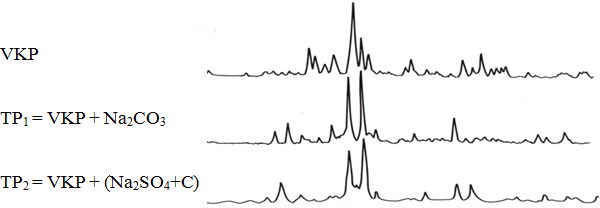
Fig. 2 – Diffraction pattern of initial phosphate (VKP) and thermal phosphate
Fertilizers derived by thermal method, which is based on VKP and mentioned waste, meet the requirements for simple superphosphate, in terms of P2О5(dig) content. [4].
Conclusion
Thus, based on the provided research the following conclusion can be made: usage of sulfate-soda mixture and product disposal of organic sludge (waste of some of the industries of petrochemical synthesis) at the optimum firing conditions provides a phosphorus fertilizer with 20-21 % content of P2О5(dig) .
Список литературы / References
- Андреев М. В. Технология фосфорных и комплексных удобрений/ М. В. Андреев, А А.Бродский, Ю.А. Забелешинский и др.; под ред.С.Д. Эвенчика и А.А.Бродского, M.:,Химия, 1987. 464 с.
- Способ получения термофосфата / Р.Х. Хузиахметов, Н.П. Мирошкин. Заявка №2015126585 от 02.07.2015 г. Решение о выдачи патента РФ от 07.09.2016 г.
- Хузиахметов Р.Х. Технология фосфорных и комплексных удобрений из низкокачественных фосфоритов различных месторождений / Р.Х. Хузиахметов, A.М. Губайдуллина, И.П. Бреус // Вестник Казанского технологического университета. – 2009. – Т.12, № 6. – С. 52-56.
- ГОСТ Р 5956-78. Суперфосфат простой гранулированный. Технические условия. – Введ. 1979-0101. – М.: Изд-во стандартов, 1978. – 31 с.
Список литературы на английском языке / References in English
- Andreev M. V. Technologija fosfornyh i kompleksnyh udobrenij [Technology of phosphoric and complex fertilizers] / М. V. Andreev, А.А.Brodskiy, U.А.Zabeleshenskiy and others; / edited by D. Evenchik and A. A. Brodskiy, –M.; Khimiya, 1987. – 464 р. [in Russian]
- Sposob polucheniya termofosfata [A method of thermophosphate producing] / Kh. Khuziakhmetov, N. P. Miroshkin // Zajavka №2015126585 ot 02.07.2015. Reshenie o vydachi patenta RF ot 07.09.2016 [Request №2015126585 from 02.07.2015. The decision on the patent RF issuance from 07.09.2016] [in Russian]
- Khuziakhmetov R. Kh. Technologija fosfornyh i kompleksnyh udobreniy iz nizkokachestvennyh fosforitov razlichnyh mestorozhdenij [Technology of phosphoric and complex fertilizers from low quality phosphate rock of different fields] / R. Kh. Khuziakhmetov, A.М. Gubaydullina, I.P. Breus // Vestnik Кazanskogo tehnologicheskogo universiteta [Bulletin of Kazan Technological University]. – – Vol. 12, № 6. – Р. 52-56 [in Russian]
- GOST R 5956-78. Superfosfat prostoy granulirovanniy. Tehnicheskie uslovija [The simple superphosphate granular. Technical conditions]. – 1979–01–01. – M. : Izd-vo standartov, 1978. – 31 p. [in Russian]
