МИКРОБИОЛОГИЧЕСКИЕ И МОРФО-ФУНКЦИОНАЛЬНЫЕ ОСОБЕННОСТИ ЭЯКУЛЯТА ИНФЕРТИЛЬНЫХ МУЖЧИН С БЕССИМПТОМНОЙ БАКТЕРИОСПЕРМИЕЙ
Годовалов А. П.1, Карпунина Т. И.2
1ORCID: 0000-0002-5112-2003, кандидат медицинских наук, 2ORCID: 0000-0003-2511-4656, доктор биологических наук, ФГБОУ ВО Пермский государственный медицинский университет имени академика Е.А. Вагнера Минздрава России
Работа выполнена при поддержке гранта РФФИ №16-44-590429 и Администрации Пермского края
МИКРОБИОЛОГИЧЕСКИЕ И МОРФО-ФУНКЦИОНАЛЬНЫЕ ОСОБЕННОСТИ ЭЯКУЛЯТА ИНФЕРТИЛЬНЫХ МУЖЧИН С БЕССИМПТОМНОЙ БАКТЕРИОСПЕРМИЕЙ
Аннотация
В настоящем исследовании показано, что выявление условно патогенных микроорганизмов, адгезированных на сперматозоидах, как правило, сопровождается изменением качественных и количественных характеристик сперматозоидов. Высокую степень обсемененности можно рассматривать как триггер выработки антиспермальных антител, которые изменяют качественные и количественные характеристики сперматозоидов, их двигательную активность. Бессимптомную бактериоспермию целесообразно учитывать при планировании беременности и применении вспомогательных репродуктивных технологий.
Ключевые слова: Бессимптомная бактериоспермия, условно патогенные микроорганизмы, антиспермальные антитела, снижении фертильности.Godovalov A. P.1, Karpunina T. I.2
1ORCID: 0000-0002-5112-2003, MD, 2ORCID: 0000-0003-2511-4656, PhD in Biology, Acad. E.A. Wagner Perm State Medical University, Perm
MICROBIOLOGICAL AND MORPHO-FUNCTIONAL FEATURES OF EJACULATE FROM INFERTILE MEN WITH ASYMPTOMATIC BACTERIOSPERMIA
Abstract
This study shows that for asymptomatic bacteriospermia potentially pathogenic microorganisms which adhere to the sperm cells, can reduce the fertile properties of ejaculate. A high degree of contamination can be seen as a production trigger of sperm antibodies which change the quality and quantity of sperm cells, their motility. It’s advisable to consider asymptomatic bacteriospermia when planning a pregnancy and using the assisted reproductive technologies.
Keywords: Asymptomatic bacteriospermia, potentially pathogenic microorganisms, sperm antibodies, impairment of fertility.Background. According to the published data of long-term studies in Russia, 10-17% of families are infertile (4-4.5 million married couples), with half of cases connected with men's reproductive dysfunction, which is consistent with the results of such studies abroad [1]. It is generally accepted that the infectious and inflammatory diseases of the testicles, appendages and prostate, the incidence of which is increasing every year, negatively affect the spermatogenesis and fertility [3, 13]. There is a data published, explaining the reduction in fertile properties of ejaculate by impaired physical and chemical parameters of ejaculate, damaging effects of reactive oxygen species, direct or indirect influence of pathogens, development of pathological autoimmune reactions. At the same time, there is no convincing information that asymptomatic bacteriospermia can affect the quality of sperm and cause male infertility.
The purpose of study is to examine the parameters of the ejaculate of men who are in infertile couples and have asymptomatic bacteriospermia.
Materials and methods. A laboratory study has been carried out with use of ejaculate samples of 71 men aged 37.7±0.7 years who are in infertile marriage. Surveyed men had no clinical symptoms of infectious and inflammatory diseases. However, the ultrasound investigation detected diffuse changes of the prostate by type of chronic prostatitis in more than half of the cases. Period from the beginning of sexual activity without contraception before treatment ranged from 12 to 60 months. The material was taken and studied according to standardized methods proposed by WHO experts [14].
The immersion microscopy of native material smears calculated the total number of sperm cells, and after eosin staining the viable cells were analyzed. To evaluate the composition and morphological features of cells we used the preparations stained according to Romanovsky-Giems method.
The bacteriological analysis of samples used decimal dilutions of starting material, assuming significant for further analysis the quantity of bacteria in excess of 103 colony-forming unit per 1 ml (CFU/ml). Streptococci were isolated in blood agar, enterococci – in Enterococcus agar, staphylococci were isolated in vitelline-salt agar, Candida fungi - in Sabouraud's medium, enterobacteria were identified by plating on Endo agar, anaerobes - in rich medium for the control of sterility, spilled in "high column" in test tubes. The number of microorganisms isolated was expressed as decimal logarithm of CFU/ml number of the biological material.
Concentration of antispermal antibodies (ASA) was determined with ELISA using Bioserv Diagnostics test-systems (Germany) according to the manufacturer's instructions.
The unpaired Student's t-test was used for statistical analysis of data.
Reults. The average volume of ejaculate was 3.7±0.2 ml, pH 7.8±0.02, the number of sperm cells was 81.9±7.1 mln/ml, including 84.8±1.7% survived. There were immature sperm cells in seminal plasma samples (1.0±0.1%). The number of leucocytes did not exceed 1.4±0.5, and macrophages – 0.6±0.04 per field. Sperm cells motility was characterized by various indicators (Table 1).
Table 1 – Sperm cells motility indicators
| Sperm cells motility | Relative indicators, % | Absolute value, mln/ml |
| fast forward | 34.2±2.3 | 29.9±3.3 |
| slow forward | 18.8±1.5 | 18.1±3.1 |
| nonforward | 10.7±0.7 | 9.1±1.4 |
| none | 36.4±2.9 | 25.0±3.0 |
As it can be seen from Table 1, more than half of sperm cells were progressive movable, and a third part of cells was immovable. The average line speed of sperm cells was 12.6±0.6 μm/s.
In assessing morphological characteristics almost half (46.1±1.8%) of sperm cells had certain changes, the most significant ones were head defects (Fig. 1).
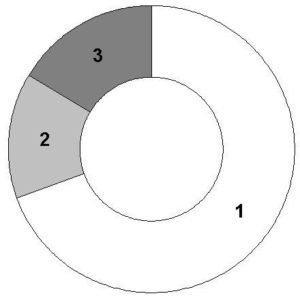
Fig. 1 – Spectrum of the recorded morphological changes of sperm of men with asymptomatic bacteriospermia (1 – head defects, 2 – mid piece defects, 3 – tail defects)
In general, despite the identified defects the indicators of sperm cells condition matched the values regulated by WHO (2010) as "normal".
The researchers believe that microbial contamination of sperm could be one of the reasons of reproductive failure [13]. According to our observations the total bacterial number of seminal plasma of surveyed group was 4.7±0.2 CFU/ml (Fig. 2). Ejaculate contamination by Gram-positive cocci was 5.2±0.1 CFU/ml, and by Gram-negative rods was 4.4±0.6 CFU/ml. Depending on gram status of plated bacteria, tested samples were nominally divided into 2 groups (Fig. 3). The number of sperm cells and their viability in two groups were not significantly different (p>0.05).
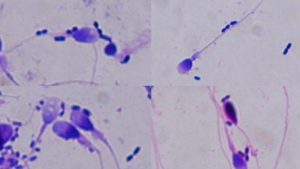
Fig. 2 – Micrograph of sperm cells with adherent microorganisms (Gram stain, ×100, oil immersion)
7.7% of samples contaminated with Gram-positive cocci had sperm cells agglutination, which can be explained by the presence of sperm agglutination factor in coccal microflora [5]. At the same time the agglutination phenomenon wasn’t found in patients with Gram-negative rods in the ejaculate. Microbial contamination didn’t have a significant impact on the aggregation of sperm cells.
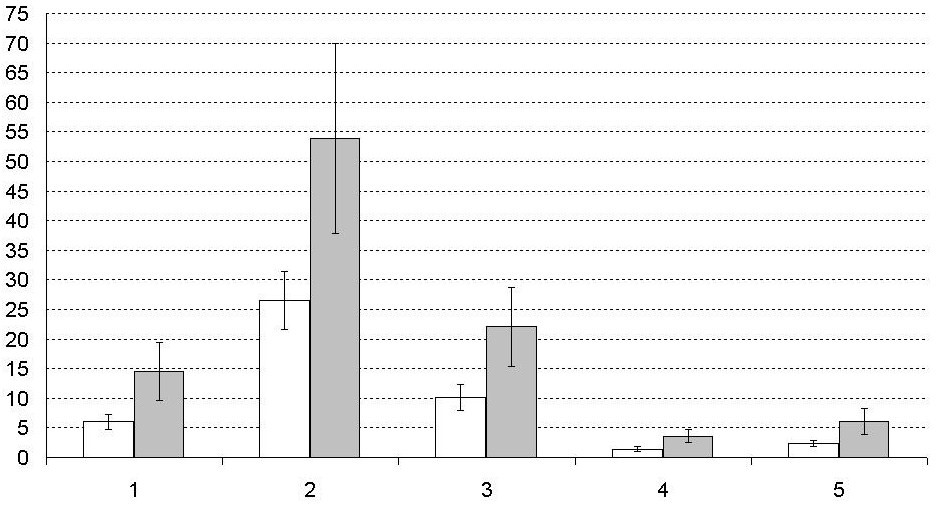
Fig. 3 – The incidence of disorders of sperm cells morphological and functional characteristics depending on the dominant microbiota (million/ml). White bars - samples containing Gram-positive cocci, grey bars - samples containing Gram-negative rods. 1 - number of sperm cells with nonforward motility, 2 - total number of defective sperm cells, 3 - number of sperm cells with head defects, 4 - number of sperm cells with mid piece defects, 5 - number of sperm cells with tail defects
It is known that while persistence of bacteria on sperm cells there is a formation of antibodies to bacterial antigens, as well as to human cells antigens [9]. ASA were found in 8.5% of men. Total microbial count of the ejaculate of men with ASA was 5.3±0.3 CFU/ml, in the absence of autoantibodies it was 3.9±0.4 CFU/ml (p<0.05). Detection of ASA in the seminal fluid was associated mainly with the release of the representatives of the Enterobacteriaceae family; in the context of gram-positive cocci detection the autoantibodies were recorded in isolated cases. According to our data the presence of sperm antibodies was associated with decreased sperm count. Thus, the number of gametes in the absence of ASA was 91.4±12.5 mln/ml, and in their presence it was 47.9±9.2 mln/ml (p<0.05).
When studying the parameters characterized the mobility of sperm cells, we found a significant change in them under the influence of sperm antibodies (Fig. 4).
Despite the change of characteristics reflecting the mobility of sperm cells ASA in the semen had no significant effect on the morphological properties of cells. The number of defective cells in the seminal plasma with ASA was 22.0±3.6 mln/ml (30.9±4.1 mln/ml - without ASA; p>0.05).
The number of leucocytes in men with different contents of ASA was not significantly different. Attention is drawn to the established connection of increasing number of germ cells (from 0.5±0.1% to 0.7±0.1%) with the presence of ASA (p<0.05). However, the explanation of mechanism and causes of this phenomenon requires the further study.
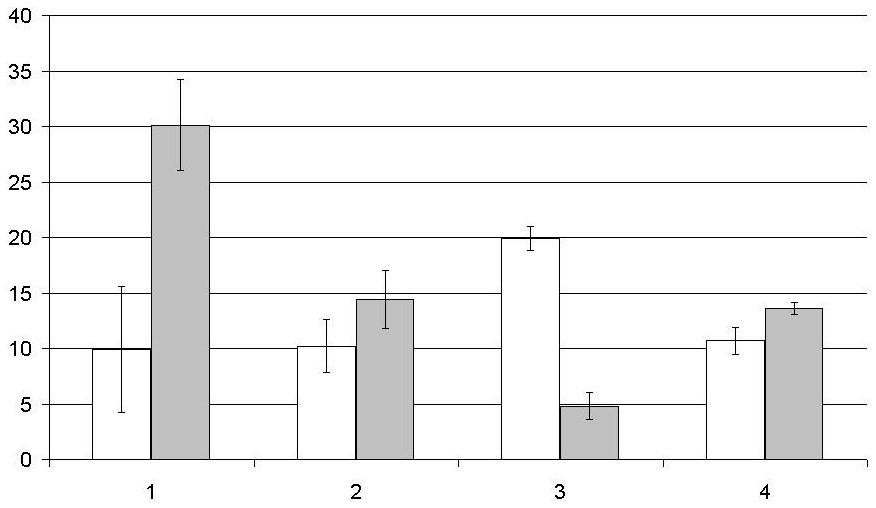
Fig. 4 – Effect of sperm antibodies on the performance of sperm motility (million/ml). White bars - group of men with ASA, grey bars - group of men without ASA, sperm cells motility: 1 - Fast forward, 2 - slow forward, 3 – none, 4 - the average line speed
Discussion. In accordance with the literature data, bacteriospermia occurs in 10-100% of cases [6] in fertile and subfertile men which may be a reflection of the diversity of used sampling protocols. According to WHO, the microorganisms are found in the ejaculate in 54% of cases. Typically, the microorganisms are isolated from the first or midstream urine and semen samples which are usually contaminated with a greater degree.
Microorganisms which are often present in seminal plasma, even if they are conditionally pathogenic commensals, may be of importance during assisted reproductive technology [7]. Such contamination often leads to disruption of fertilization and/or embryo development [2]. It is known that microorganisms have a unique ability to adhere, including adhesion to sperm promoting infection, as well as to oocyte affecting the process of fertilization and division of the zygote [8, 11].
The present study found that most of the samples collected from men with asymptomatic bacteriospermia, was contaminated with microorganisms of different species, most of which was present in amount of more than 103 CFU/ml, and the studies with use of molecular genetic techniques [7] detected microorganisms in amount of more than 104 CFU/ml in 66% of men. In addition, a significant part of bacteria passes from plankton to adhered state, including adhesion on sperm cells with altering their characteristics. According to our observations, one of the causes of transition from active generation to biofilm formation and, consequently, virtually asymptomatic infection, may be semen pH with parameters irrelevant for optimal growth of most bacteria.
As a rule, the seminal plasma infection results in impaired fertility in different ways. We also traced a tendency of increasing the content of sperm antibodies in men with asymptomatic bacteriospermia. In addition, a certain part of our observations noted the association of ejaculate contamination and sperm functional characteristics defect. The presence of microorganisms in the seminal plasma damages spermatogenesis [4] and may cause the obstructive changes of excretory ducts [6]. Microorganisms negatively affect sperm cells function both directly and indirectly through the activation of leucocytes which produce reactive oxygen species and cytokines. Reactive oxygen species contribute to the lipid peroxidation of the plasma membrane of gametes [12], and to enhancement of DNA fragmentation [10]. A significant part of the surveyed male germ cells had changes in morphology, more than half of the sperm cells was with head defects.
The importance of treating symptomatic genital infections in infertile men is clear, however, the majority of patients with culturable bacteria within the semen are asymptomatic.
Conclusion. Thus, the surveyed men who have reproductive problems had no diagnostically significant deviations in terms of morphology and motility detected. At the same time, a significant microbial contamination of the ejaculate was accompanied not only by changing the qualitative and quantitative characteristics of male germ cells, but also caused the production of autoantibodies. Therefore, asymptomatic bacteriospermia can be considered as a risk factor for impaired fertility, as well as be taken into account when planning a pregnancy and using the assisted reproductive technologies.
References
- Carr B.R. Optimal diagnosis and medical treatment of male infertility // Semin. Reprod. Med. – 2013. – Vol. 31. – P. 231–232.
- Cottell E., Lennon B., McMorrow J., Barry-Kinsella C., Harrison R.F. Processing of semen in an antibiotic-rich culture medium to minimize microbial presence during in vitro fertilization // Fertil. Steril. – 1997. – Vol. 67. – P. 98–103.
- Domes T., Lo K.C., Grober E.D., Mullen J.B., Mazzulli T., Jarvi K. The incidence and effect of bacteriospermia and elevated seminal leukocytes on semen parameters // Fertil. Steril. – 2012. – Vol. 97. – P. 1050–1055.
- Fowler J.E. Infections of the male reproductive tract and infertility: a selected review // J. Androl. – 1983. – Vol. 3. – P. 121–131.
- Kaur S., Prabha V., Sarwal A. Receptor mediated agglutination of human spermatozoa by spermagglutinating factor isolated from Staphylococcus aureus // J. Urol. – 2010. – Vol. 184(6). – P. 2586-2590.
- Keck C., Gerber-Schafer C., Clad A., Wilhelm C., Breckwoldt M. Seminal tract infections: impact on male fertility and treatment options // Hum. Reprod. Update. – 1998. – Vol. 4. – P. 891–903.
- Kiessling A.A., Desmarais B.M., Yin H.Z., Loverde J., Eyre R.C. Detection and identification of bacterial DNA in semen // Fertil. Steril. – 2008. – Vol. 90. – P. 1744–1756.
- Lang T., Dechant M., Sanchez V., Wistuba J., Boiani M., Pilatz A., Stammler A., Middendorff R., Schuler G., Bhushan S., Tchatalbachev S., Wübbeling F., Burger M., Chakraborty T., Mallidis C., Meinhardt A. Structural and functional integrity of spermatozoa is compromised as a consequence of acute uropathogenic E. coli-associated epididymitis // Biol. Reprod. – 2013. – Vol. 89(3). – P. 1-10.
- Mazumdar S., Levine A.S. Antisperm antibodies: etiology, pathogenesis, diagnosis, and treatment // Fertil. Steril. – 1998. – Vol. 70(5). – P. 799-810.
- Moustafa M.H., Sharma R.K., Thornton J., Mascha E., Abdel-Hafez M.A., Thomas A.J. Jr., Agarwal A. Relationship between ROS production, apoptosis and DNA denaturation in spermatozoa from patients examined for infertility // Hum. Reprod. – 2004. – Vol. 19. – P. 129–138.
- Rennemeier C., Frambach T., Hennicke F., Dietl J., Staib P. Microbial quorum-sensing molecules induce acrosome loss and cell death in human spermatozoa // Infection and immunity. – 2009. – Vol. 77(11). – P. 4990–4997.
- Sharma R.K., Agarwal A. Role of reactive oxygen species in male infertility // Urology. – 1996. – Vol. 48. – P. 835–850.
- Weng S.L., Chiu C.M., Lin F.M., Huang W.C., Liang C., Yang T., Yang T.L., Liu C.Y., Wu W.Y., Chang Y.A., Chang T.H., Huang H.D. Bacterial communities in semen from men of infertile couples: metagenomic sequencing reveals relationships of seminal microbiota to semen quality // PLoS One. – 2014. – Vol. 9(10). – P. e110152.
- WHO laboratory manual for the examination and processing of human semen. 5th ed. – WHO (Geneva), 2010. – Vol. 270. – 437 P.
