PHYSICAL-CHEMICAL PRINCIPLES OF CARDONACEOUS MATERIALS SELECTION FOR ARSENOPYRITE CONCENTRATE PROCESSING
Исабаев С.М.1, Кузгибекова Х.М.2, Жинова Е.В.3
1Доктор технических наук, профессор; 2Кандидат технических наук, доцент; 3Соискатель, Химико-металлургический институт им. Ж. Абишева
ОПРЕДЕЛЕНИЕ ФИЗИКО-ХИМИЧЕСКИХ ЗАКОНОМЕРНОСТЕЙ СЕЛЕКЦИИ УГЛИСТЫХ ВЕЩЕСТВ ПРИ ПЕРЕРАБОТКЕ АРСЕНОПИРИТНОГО КОНЦЕНТРАТА
Аннотация
Трудность извлечения золота из руд месторождения Бакырчик связана с наличием в составе элементного углерода в виде шунгитов, обладающих высокой сорбционной емкостью по отношению к флотационным реагентам и к цианистым комплексам золота, поэтому проведены исследования по селекции углистых веществ из особоупорного золотомышьяковистого сырья.
Ключевые слова: золотомышьяковистый концентрат, арсенопиритный концентрат.
Issabayev S.M.1, Kuzgibekova Kh. M.2, Zhinova E.V.3
1 Doctor of Technical Sciences, professor; 2 Candidate of techn.science, Docent; 3 Competitor, Chemical Metallurgy Institute named after Abishev
PHYSICAL-CHEMICAL PRINCIPLES OF CARDONACEOUS MATERIALS SELECTION FOR ARSENOPYRITE CONCENTRATE PROCESSING
Abstract
Extraction of gold from “Bakyrchik” deposit gold ore is complicated by the presence of elemental carbon in the form of schungite. The latter has a high absorptive capacity in respect to floatation reagent and cyanic gold complexes which require the research on carbonaceous materials selection from persistent gold-arsenic concentrate.
Keywords: gold-arsenic concentrate, arsenopyrite concentrate.
Schungite is determined as a “non-graphite” carbon and differs from graphite by the absence of crystallic structure, from bitumen and coals by the small number of contained volatile components. In Table 1 there are the results of physical-chemical properties of schungite fraction isolated from Bakyrchik ores according to the technology by Branch state establishment “Eastern scientific-research institution of non-ferrous metals” (VNIITzvetmet) [1].
Table 1 - Physical-chemical and absorptive properties of schungite fraction
| Indices | Schungite fraction |
| Poured/scattered/sprinkled density, g/cm3 | 0,650 |
| Iode activity, % | 72,4 |
| Lightening ability by methylene blue, % | 25,0 |
| Schungite fraction capacity, mg/g on gold | 14,2 |
| Schungite fraction capacity, mg/g on silver | 7,7 |
The results of schungite fraction absorptive properties, in particular the capacity in gold is 14,2 mg/g, in silver – 7,7 mg/g, show that for the effective application of traditional methods of gold lixiviation the preliminary stage of carbonaceous substances removal from the resistant gold-arsenic concentrate from Bakyrchik deposit is required.
One of the famous methods of absorptive carbonaceous substances activity reduction in the gold-contained ores is the method of absorptive activity passivation by the surface active substances. However this process depends on the converted carbonaceous substances degree, i.e. on their composition and structure. The application of the method of absorptive activity passivation to the schungite minerals group connected with the surface-active reagents did not give any positive results [2]. In this connection there were carried out experiments on the thermal processing influence on the schungite fraction removal from furnace charge of Bakyrchik gravity-floating-concentrates. The characteristic peculiarity of the nature gold-containing organic substances is their solution in the organic solvents, which contain nitrogen and arsenic compounds (functional groups), that is why 10% of tar solution was used for the carbonaceous substances removal.
The initial material after the Buechner funnel processing by 5-10% tar solution (the oil industry processing wastes) and 24-hour saturation was roasted in the muffle furnace under different temperatures and roasting length. The results are shown in fig. 1-3.
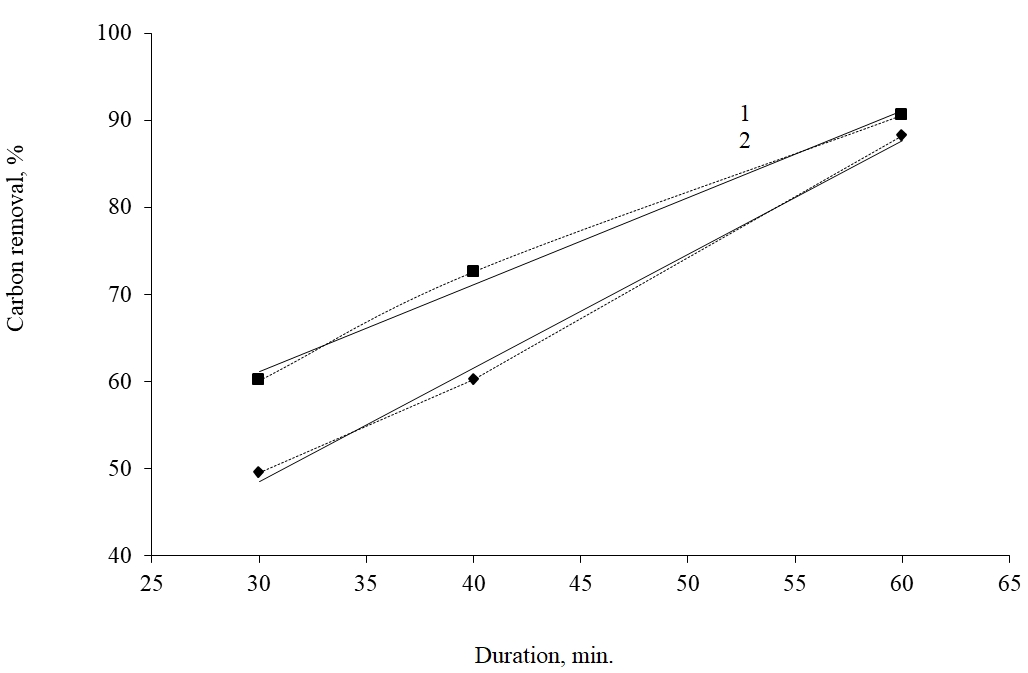
1 – 7000С, is described by the equation y = x + 31,1, R2 = 0,9926; 2 - 6000С, has an equation y = 1,3057x + 9,3857, R2 = 0,9961
Fig. 1. Dependence of the degree of carbonaceous substances removal from Bakyrchik concentrate on the temperature and roasting length
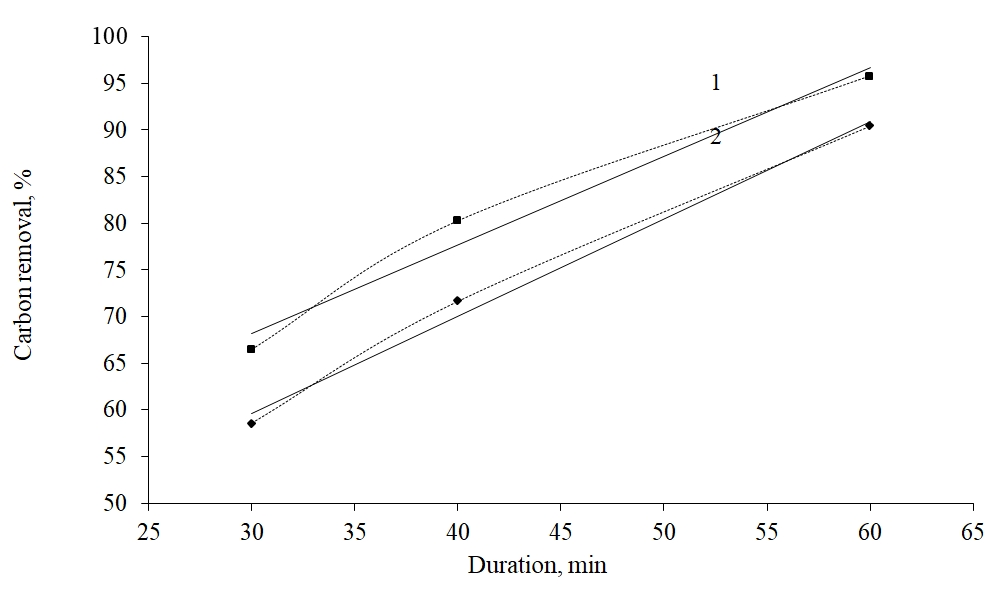
1 – 7000С, is described by the equation y = 0,9481x + 39,774, R2 = 0,976; 2 - 6000С, has an equation y = 1,0426x + 28,362, R2 = 0,9926
Fig. 2. Dependence of the degree of carbonaceous substances removal from Bakyrchik concentrate processed by 5% tar on the temperature and roasting length
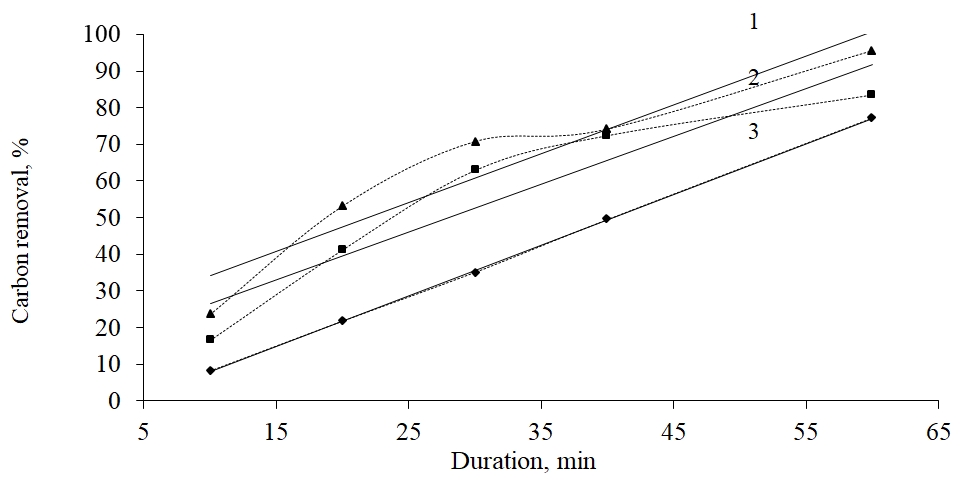
1 – 7000С, is equated y = 1,3319x + 20,859, R2 = 0,9065; 2 - 6000С, has an equation y = 1,3068x + 13,484, R2 = 0,8869; 3 - 5000С, has an equation y = 1,382x - 5,8649, R2 = 0,9999
Fig. 3. Dependence of the degree of carbonaceoussubstances removal on Bakyrchik concentrate processed by 10% tar on the temperature and roasting length
In Table 2 there are shown the meanings of activation energy of removal process of carbonaceous substances from furnace charge of gravity-floating-concentrate in proportion 1:1 based on the results, shown in fig. 1-3.
Table 2 - Meanings of activation energy of removal process of carbonaceous substances
| Initial material | Activation energy meanings, kJ/molar | ||
| without tar processing | processed by 5% tar solution | processed by 10% tar solution | |
| Furnace charge (gravity-floating-concentrate =1:1) | 12,7 | 22,0 | 50,1 |
The carbonaceous substances removal process during Bakyrchik concentrates roasting under our testing conditions can be referred to the reactions (1) of the type:
solid I + liquid → solid II + gas (1)
In our tests the induction period was observed with difficulties; after surface formation of phase section – porous iron oxides - diffusion did not influence the process of carbon removal that could be seen from the activation energy meanings (Table 2).
In Table 3 there are shown the results of carbonaceous substances removal from thermal-processed residue of Bakyrchik concentrates by hydrometallurgical methods.
Table 3 - Results of carbonaceous substances removal from (arsenic – 0,1%, gold – 24 g/t, iron – 12,88%, sulphur – 5,24%, carbon – 2,45%)
| Testing conditions | Oxidizer’s expense, in % on the furnace charge weight | Carbon removal degree, % |
| Hydrochlorination:700С, 1 hour | Cl2 | 1,9 |
| Sulphur-oxidizing lixiviation: 900С, 1 hour | 2% MnO2 | 7,2 |
| Nitrate lixiviation: 900С, 1 hour | 50% KNO3 | 0 |
| Lixiviation with ammonium persulphate: 900С, 30 minutes | 10% (NH4)2S2O8 | 13,8 |
From the analysis of Table 3 it is seen that liquid-phase carbonaceous substances selection from Bakyrchik concentrate does not give any positive results, that is why the choice is made in favor of pyrometallurgical methods of carbon removal.
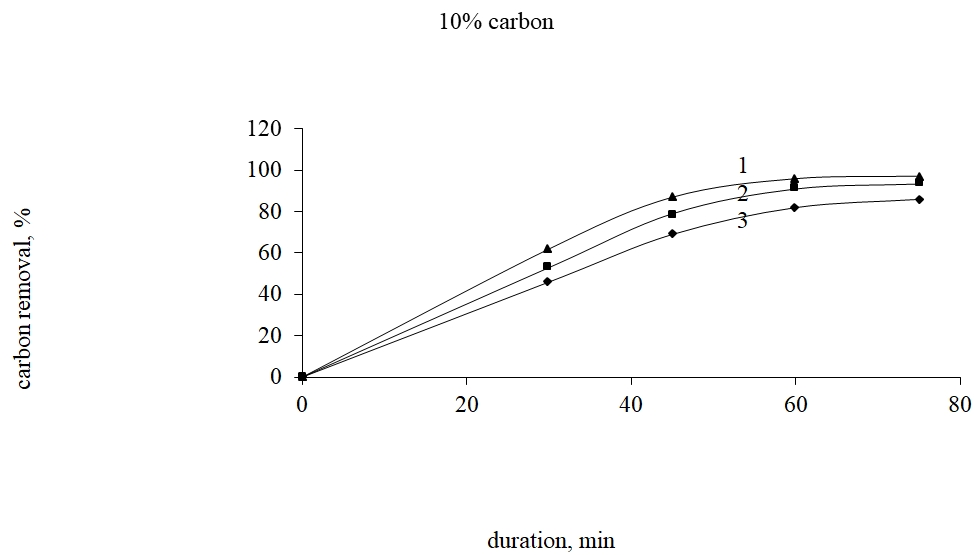
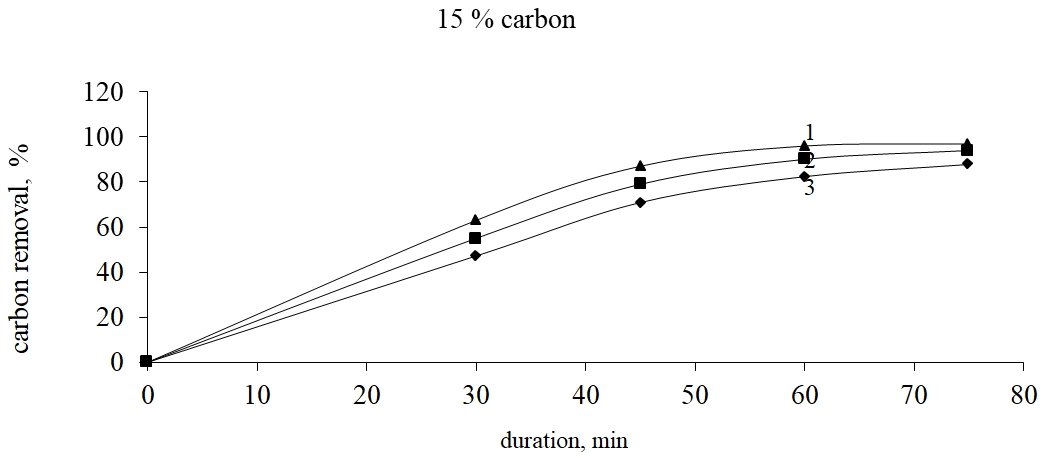
Analysis of thermal and liquid-phase method of carbonaceous substances removal from resistant raw material shows that the most acceptable method is the thermal selection under the temperature lower than 7000C which is connected with porous material formation used in traditional methods of lixiviation of gold by cyanide, tiourea, tiosulphate and others. The research in carbonaceous substances removal from dearsenized thermal-processed residue from Bakyrchik concentrate was made by the roasting in the oxidized surroundings with Shubarkul carbon addition because the tar processing is complex for its technical support. In fig. 4-6 there are shown the results of the carbon removal from dearsenized thermal-processed residue by the oxidized roasting in the dependence on the temperature and the carbon amount taking into account the initial material weight.
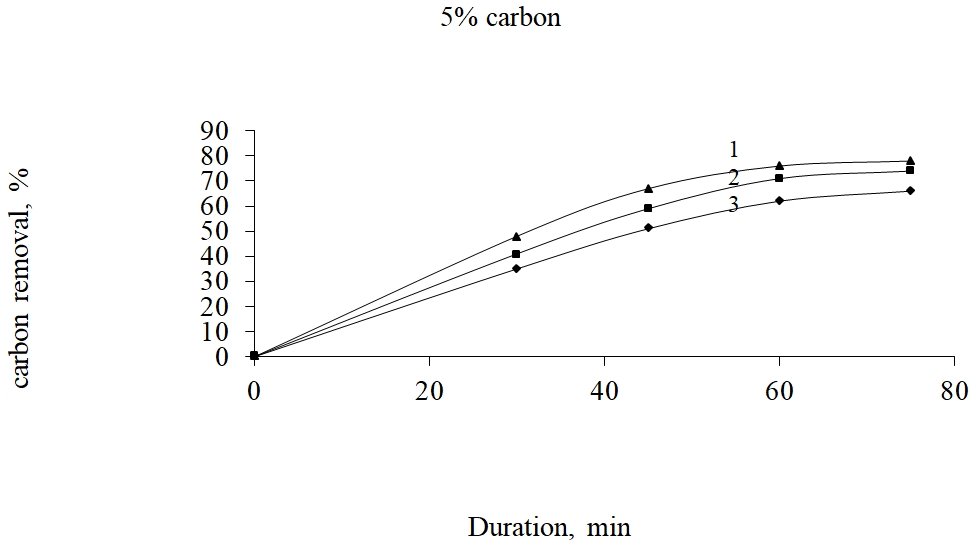
1 – 6000С; 2 - 5000С; 3 - 4000С
Fig. 4. Dependence of the degree of carbonaceous substances removal on the roasting temperature at carbon expense 5% considering the initial material weight
1 – 6000С; 2 - 5000С; 3 - 4000С
Fig. 5. Dependence of the degree of carbonaceous substances removal on the roasting temperature at carbon expense 10% considering the initial material weight
1 – 6000С; 2 - 5000С; 3 - 4000С
Fig. 6. Dependence of the degree of carbonaceous substances removal on the roasting temperature at carbon expense 15% considering the initial material weight
From fig. 4-6 it is seen that at 15% of Shubarkul carbon addition by the oxidizing roasting there is achieved 99,0% of carbon extraction at 6000C and 75 min. duration.
Consequently, these parameters of thermal schungite selection will serve as starting results in working out the technology of Bakyrchik concentrates agglomerated roasting aiming at absorptive-active carbonaceous substances removal.
References
- Golikov A.A. An experience of resistant gold-containing ores. In the book: Collection of scientific articles of Eastern scientific-research institution of non-ferrous metals. Rational use of mineral resources in Republic of Kazakhstan and environment improvement. Ust-Kamenogorsk: 1996. P. 87-93.
- Fridman I.D., Savari Ye.Ye., Dyomina N.N. Influence of carbonaceous substances contained in ores on the cyanization process // Non-ferrous metals. - 1979. - № 9. - P. 104-106.