EXPERIMENTAL INVESTIGATIONS OF THE INFLUENCE OF THE ELECTRIC FIELD ON THE FORMATION OF THE ICE PHASE ON ZINC OXIDE NANOTUBE CLUSTERS
EXPERIMENTAL INVESTIGATIONS OF THE INFLUENCE OF THE ELECTRIC FIELD ON THE FORMATION OF THE ICE PHASE ON ZINC OXIDE NANOTUBE CLUSTERS
Abstract
The paper presents the results of studying the effect of an electric field on the ice-forming properties of a new ice-forming reagent based on clusters consisting of zinc oxide nanotubes. In the course of laboratory experiments, it was found that the mechanism of the effect of an electric field on the specific yield of ice-forming zinc oxide clusters depends on the field strength. At field strengths up to 1.1∙104 V/m, the impact is associated with fragmentation of clusters into several parts, at field strengths over 1.1∙104 V/m, with a change in the structure of water in clusters. Both of these processes lead to a change in the specific yield of ice-forming particles. When crushing clusters, both an increase in the specific yield of ice-forming particles and its decrease is possible. The specific yield is determined by the number of particles whose size is larger than the critical one. The critical particle size decreases at lower temperatures. A change in the structure of water at high electric field strengths leads to an increase in the specific yield of ice-forming particles.
1. Introduction
Of great importance for a successful impact on cloud processes is the correct dosage of the introduced regent, for which it is necessary to study the effect of various cloud parameters on the ice-forming properties of the reagents. One of these cloud parameters is the presence of an electric field in it. The influence of the electric field can be divided into two parts: on the formation of reagent particles and on the structure of water in the cloud. The influence of the electric field on the formation of reagent particles is associated with the processes of coagulation and fragmentation of particles in an electric field. When the reagent is sublimated, a spectrum of particles is formed, the left part of which, ranging from zero to a certain size R, does not exhibit ice-forming properties. The value of the particle size R depends on the temperature. There is an optimal particle size of the reagent for each temperature, where the specific yield of ice-forming particles is maximum. An increase or decrease in temperature leads to a decrease in the specific yield of ice-forming particles. The electric field breaks the reagent particles, increasing the number of particles. Currently, experimental works are known to study the effect of an electric field on the phase transition of supercooled water from a liquid thermodynamically unstable state to a crystalline one , , . Studies carried out in this direction show that the growth of seed particles from the vapor phase depends on the strength of the electric field, the charge of the crystallizing core and on the implementation of one or another growth mechanism , . An interesting result on the influence of an electric field on the process of ice formation was obtained in . The authors, conducting experiments on freezing a water drop at the tip of an electron microscope cantilever, found that the drop crystallizes at room temperature at an electric field strength of 106 V/m. As is known, the electric field strength increases by three orders of magnitude at the tip of the cantilever. Consequently, structural changes in water occur at electric field strengths of the order of 109 V/m; there is no such strength in clouds. Before creating a complex of special equipment for the study of the influence of the electric field on the processes of formation of the ice phase in clouds, the authors of the work studied a huge scientific material , , , .
In the early works of the authors , the results of studying the effect of an electric field on the ice-forming properties of zinc oxide nanotubes at an electric field strength of 105 V/m were presented. This paper presents the results of further research. The purpose of this work is to study the effect of an electric field of different electric field strengths on the ice-forming properties of a reagent based on clusters of zinc oxide nanotubes. The authors of this article have empirically obtained that zinc oxide nanotube clusters are formed during the sublimation of zinc. The study of clusters under an electron microscope shows that they have a loose structure and are divided into several parts under the influence of an electric field. Based on the foregoing, the effect of the electric field on the ice-forming properties of zinc oxide nanotube clusters should be significant.
2. Equipment for studying the effect of an electric field on the ice-forming properties of reagents
As part of the study, the dependence of the ice-forming efficiency of zinc oxide nanotube clusters on the electric field strength was studied. To conduct the study, a complex of special equipment was created, the scheme of which is shown in Figure 1. The complex of equipment includes a large cloud chamber, plates of a flat capacitor, a high-voltage rectifier, a reagent sublimation device, an ultrasonic steam generator, a balance, an optical microscope (Figure 1).
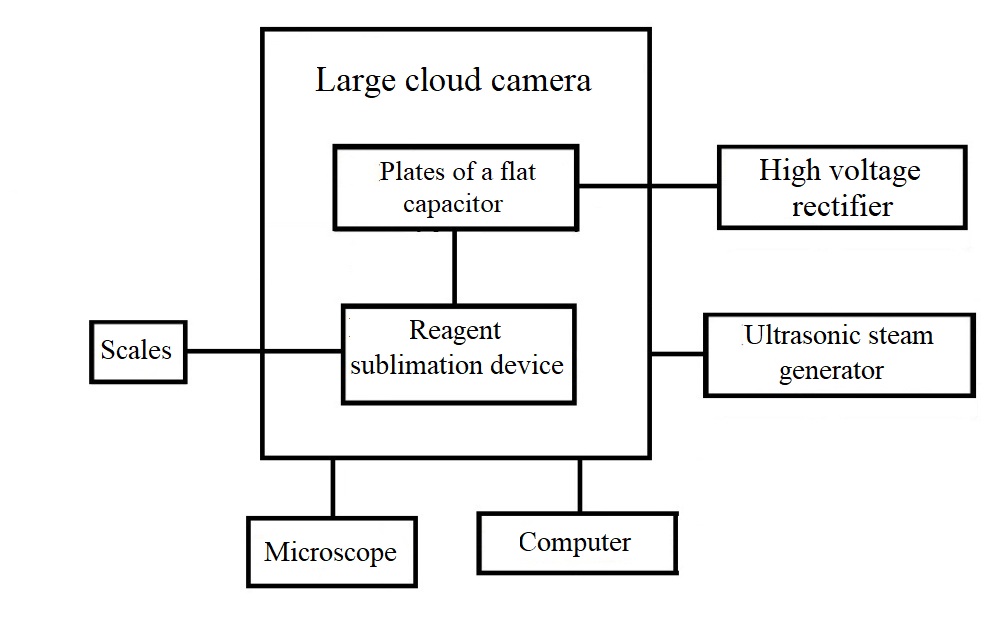
Figure 1 - A set of equipment for studying the effect of electric field strength on the ice-forming properties of zinc oxide nanotube clusters
3. Method for studying the effect of electric field strength on the ice-forming efficiency of the reagent
A certain amount of zinc is weighed on electronic scales. It is loaded onto the graphite substrate of the reagent sublimation device. Thermostated substrates covered with lids are installed at the bottom of a large cloud chamber. An artificial cloud environment is created in the chamber using an ultrasonic steam generator. After that, a high voltage is applied to the capacitor plates from a high-voltage rectifier. A direct current is supplied to the sublimation device, high-temperature sublimation of zinc occurs. After sublimation, the air in the chamber is mixed with a fan. With the appearance of the first ice crystals in the field of view, the substrates are opened in turn and crystals are deposited on them. Each substrate is examined in the field of an optical microscope, the number of crystals in the frame is counted, and then the specific yield is calculated on a computer.
The specific yield of ice-forming nuclei is determined by the formula:
where Scham is the area of the reagent inlet chamber, µm2; Sframe is the frame area in the field of view of the microscope, µm2; nsub is the mass of the injected reagent, g; mreag is the number of crystals in the setting, m-2.
4. Materials and methods of research
The results of the conducted studies are presented in Tables 1 and 2, and are illustrated in Figures 2-3.
Table 1 - The results of the study of changes in the specific yield of ice-forming particles at different temperatures and electric field strength
Specific yield, 1013, g-1 | ||||
t, °С | 0 V/m | 1500 V/m | 15000 V/m | 150000 V/m |
-13 | 3.4 | - | - | 10.5 |
-12 | 2.8 | - | 7.1 | 12 |
-11 | 4.4 | 4.9 | 5.6 | 8.6 |
-10 | 3.5 | 4.4 | 4.9 | 11.4 |
-9 | 1.5 | 3.3 | 4.5 | 7.3 |
-8 | 2.9 | 4.6 | 3.9 | 9.6 |
-7 | 1.3 | 2.3 | 2 | 8.3 |
-6 | 1.9 | 3.3 | 1.7 | 5.4 |
-5 | 2.5 | 1.3 | 1.6 | 3.3 |
-4 | 1.2 | 2.2 | 1.4 | 4.8 |
-3 | 2.3 | 0.8 | 0.2 | 2.7 |
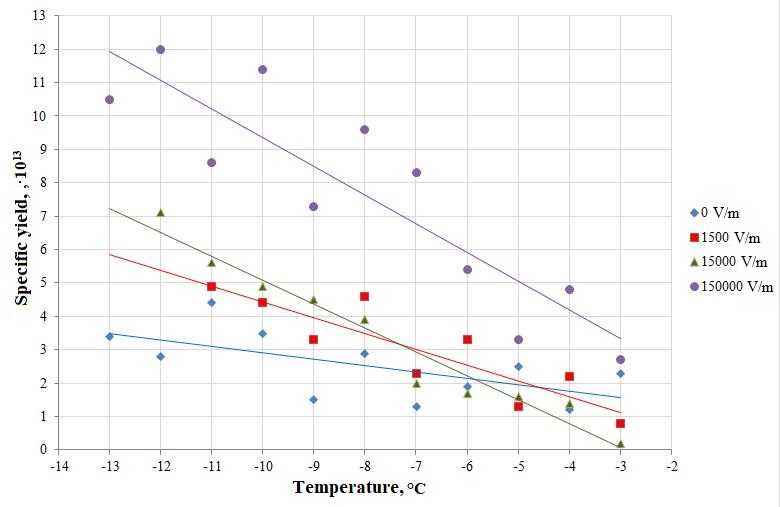
Figure 2 - Temperature dependence of the specific yield of zinc oxide nanotube clusters at different electric field strength
Table 2 - The results of the study of changes in the specific yield of ice-forming clusters at different electric field strengths
Specific yield, 1013, g-1 | ||||
Е, V/m | -3…-4°С | -5…-7°С | -8…-10°С | -11…-13°С |
0 | 1.8 | 1.9 | 2.6 | 3.5 |
1500 | 1.5 | 2.3 | 4.1 | 4.9 |
15000 | 0.8 | 1.8 | 4.4 | 6.4 |
150000 | 3.8 | 5.7 | 9.4 | 10.4 |
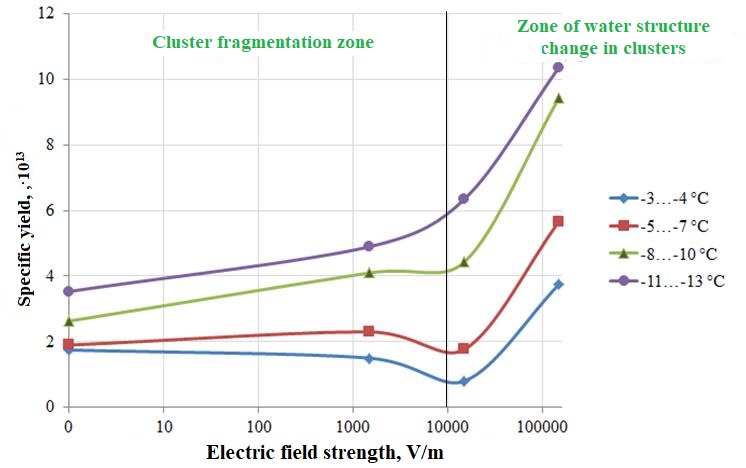
Figure 3 - Dependence of the specific yield of zinc oxide nanotube clusters on the electric field strength at different temperature ranges
The research results presented in the paper show that with an increase in the electric field strength from 0 to 1.0∙105 V/m, the specific yield of ice-forming zinc oxide nanotube clusters varies depending on temperature. In the temperature range from -3 to -4°C at an electric field strength of up to 1.0∙104V/m, the specific output decreases, a more significant decrease is observed at electric field strengths from 1.0∙103V/m to 1.0∙104V/m. A further increase in the electric field strength leads to an increase in the specific yield of ice-forming particles. At an electric field strength of up to 1.0∙103V/m, there is a slight increase in the specific yield of ice-forming particles in the range from -5 to -7°C. A further increase in the electric field strength to 1.0∙104V/m leads to a decrease in the specific yield of ice-forming particles. The higher the temperature, the greater the decline in the specific yield of ice-forming particles. Sublimation of zinc results in the formation of clusters of oxide nanotubes. Studies show that clusters have a very loose structure and, under the influence of an electric field, are divided into several clusters. Clusters, depending on their size, can be ice-forming cores or passive impurities. If the particle size is larger than the critical one, then the cluster is an ice-forming particle; if it is smaller, then it is a passive impurity. The lower the ambient temperature, the smaller the critical diameter and the more ice-forming centers, and, accordingly, the higher the specific yield of ice-forming nuclei. The mechanism of the effect of an electric field on the specific yield of ice-forming zinc oxide clusters depends on the strength of the electric field. Up to electric field strengths of 1.0∙104V/m, the impact is associated with fragmentation of clusters into several parts; at strengths over 1.0∙104V/m, the structure of water in clusters changes. Both processes lead to a change in the specific yield of ice-forming particles. When crushing clusters, both an increase in the specific yield and its decrease are possible. The specific yield is determined by the number of particles whose size is larger than the critical size, in turn, the critical size is smaller at lower temperatures. A change in the structure of water at high values of the electric field leads to an increase in the specific yield of ice-forming nuclei. The results of the experiments show that in the temperature range from -3 to -4°C at an electric field strength of 0 to 1.0∙104V/m, the number of particles with sizes larger than the critical one decreases, and particles with sizes smaller than the critical one are observed at electric field strengths from 1.0∙104 to 1.0∙105V/m in the temperature range from -3 to -13°С. At electric field strengths greater than 1.0∙105V/m, the influence of the electric field on the structure of water is observed. It should be noted here that external electric fields are amplified by three orders of magnitude by nanotubes, therefore, the effect on the structure of water begins at electric field strengths from 1.0∙108V/m.
5. Conclusion
The results of the conducted studies show that the mechanism of the effect of an electric field on the specific yield of ice-forming zinc oxide clusters depends on the electric field strength. Up to an electric field strength of 1.0∙104V/m, the impact is associated with the fragmentation of clusters into several parts, at a strength of more than 1.0∙104V/m, the impact is associated with a change in the structure of water in clusters. Both processes lead to a change in the specific yield of ice-forming particles. When crushing clusters, both an increase and a decrease in ice-forming particles are possible. The specific yield is determined by the number of particles whose size is larger than the critical one. In turn, the critical size is smaller at lower temperatures. A change in the structure of water at high values of the electric field leads to an increase in the specific yield of ice-forming nuclei. It was found that with an increase in the electric field strength from 0 to 1.0•105 V/m, the specific yield of ice-forming clusters of zinc oxide nanotubes varies depending on the temperature. In the temperature range -3…-4°C up to electric field strengths of 1.0∙104V/m, the specific output decreases, a more significant decrease is observed at electric field strengths from 1.0∙103V/m to 1.0∙104V/m. A further increase in the electric field strength leads to an increase in the specific yield of ice-forming particles. In the temperature range -5..-7°C up to the value of the electric field strength of 1.0∙103V/m, there is a slight increase in the specific yield of ice-forming particles, and a further increase in the electric field strength to 1.0∙104V/m leads to a decrease in the specific yield ice forming particles. The higher the temperature, the greater the decline in the specific yield of ice-forming particles.
