СВОЙСТВА ШТАММА EXIGUOBACTERIUM MEXICANUM
Ерофеевская Л.А.1, Попова Л.Л.2, Салтыкова А.Л.3
1Научный сотрудник, Федеральное государственное бюджетное учреждение науки Институт проблем нефти и газа Сибирского отделения Российской академии наук, 2Генеральный директор, ООО «Зеленая лаборатория», 3Инженер, ООО «СахаНефтеБиоСорб»
СВОЙСТВА ШТАММА EXIGUOBACTERIUM MEXICANUM
Аннотация
Из нефтезагрязненных мерзлотных почв выделен штамм факультативно-анаэробных бактерий рода Exiguobacterium. Изучены его свойства и филогенетическая характеристика. Исследовано влияние штамма на активацию биологической деструкции нефти и нефтепродуктов. Установлено, что метод интродукции полученного штамма в загрязненные объекты способствует сокращению сроков деградации нефтяных углеводородов, как в почвенных, так и водных экосистемах. За 7 суток утилизация нефтепродуктов в водной среде под влиянием штамма достигает 13-81%, в зависимости от температуры окружающей среды.
Ключевые слова: микроорганизмы, углеводороды, нефть, почва, штамм, E. mexicanum.Erofeevskaia L.A.1, Popova L.L.2, Saltycova A.L.3
1Research associate, Institute of oil and gas problems Siberian branch of theRussian Academy of Sciences, 2Dierector general LLK «Green laboratory», 3Engineer, LLK «SakhaOilBioSorbents»
PROPERTIES OF STRAIN OF EXIGUOBACTERIUM MEXICANUM
Abstract
A strain of facultative anaerobic bacteria of the genus Exiguobacterium is isolated from oil-contaminated permafrost-affected soils. Its properties and phylogenetic characteristics have been studied. Also the influence of strain on the activation of the biological degradation of oil and oil products was tested. It is found that the method of introduction of the resulting strain at contaminated sites helps shorten the petroleum hydrocarbon degradation in the soil and aquatic ecosystems. Depending on the ambient temperature a 7 days recycling oil in an aqueous medium under the influence of the strain reaches 13-81%.
Keywords: microorganisms, hydrocarbons, oil, soil, strain, E. mexicanum. Currently, the problem of environmental pollution with petroleum hydrocarbons (HC) is an important area of concern. Rehabilitation of affected areas after accidental oil spills is particularly demanded in the Far North, where the main oil and gas fields are operated by the Russian Federation.Regardless of its geography and fine chemical differences oil is a unique substance in many ways, and first of all in that it degrades spontaneously very slowly, and its components often acquire high resistance to oxidation [1].
Without the use of special technologies for the rehabilitation of affected lands, self-healing process of oil-contaminated soils in the areas with certain climatic conditions may take 10-25 years, while the decomposition of oil and its derivatives in the North may last up to 50 years and beyond [2-4].
Among frequently applicable solutions for effective recovery of the soil from oil contamination in the north, biological processes based on the intensification of degradation of oil hydrocarbons by some microbes has been environmentally justified [5]. A search for highly effective, non-toxic and non-pathogenic strains of hydrocarbon-oxidizing microorganisms (LCS), has therefore become a task of an outmost importance.
The aim of the present study was to isolate and study the properties of a new non-pathogenic LCS strain, capable of cleaning up soil contamination with oil and oil products.
Objectives of research:
1) to isolate a not pathogenic strain of bacteria capable for degradation of petroleum hydrocarbons from permafrost soils;
2) to analyze the nucleotide sequence of the 16S рРНК gene fragments and to determine the phylogenetic position of the selected strain;
3) to study cultural, morphological, physiological, biochemical and chemo-taxonomic characteristics of the strain;
4) to test the resulting strain for its suitability for the environmental remediation of soils.
The materials and methods of research.
We focused on the strain E. mexicanum IPNG-ELA-5 deposited in the Russian National Collection of Industrial Microorganisms (VKPM), GosNIIgenetika FSUE (Moscow). A method of liquid enrichment cultures in the mineral medium described by Muntz was used for initial cultures of the bacteria [6]. Talakan oil, containing 0.82% wax and 12.4% of resinous substances, was used as a carbon source [7]. Evaluation of viability of the strain was carried out by the Koch method [8].
Identification of the isolated strain was carried out on the basis of morphological, cultural, physiological and biochemical properties [9, 10], and was confirmed by 16S рРНК gene nucleotide sequencing. For that, isolation of DNA for PCR was conducted by the method of Ribosomal Database Project II [11]. PCR was performed using a GeneAmp PCR System 2700 instrument [12, 13]. Phylogenetic trees were built up with the aid provided by the website software [14].
The sensitivity of the strain to antibiotics was determined by a standard agar diffusion method [15].
The presence of oil in water was determined by spectrometry using Oil Detector instrument [16]. The content of oil in the soil was determined by cold extraction by chloroform. Structural and group composition of the extracts and their fractions was studied by infrared Fourier spectroscopy. Group component composition of the extracts was determined by column chromatography [17].
The results of the study.
A strain of bacteria E. mexicanum IPNG-ELA-5 had been isolated from permafrost soils contaminated with oil. The species characteristics have been studied and are presented below.
Morphological and cultural properties.
The bacterium is a Gram (+) mobile rod, 0.8 - 1.0 μm in size. It has numerous (peritrichally organized) flagella. In a smear the rods are assembled in doublets and larger clusters. It is a facultative anaerobe which doesn’t form spores or capsules. On a meat-peptone agar, the strain forms humid dirty-yellowish colonies of round shape. The appearance of colonies change after 1-2 days: they turn dry, and their edges become uneven.
In the Saburo medium the bacteria form large colonies with yellowish-orange color and the maximum diameter reaching 0.5 cm.
In a meat-peptone broth (weight %: enzymatically treated peptone – 1.0, NaCl - 0.5, meat water extract – 98.5; pH 7.0 to 7.2) it grows forming a diffuse opacity.
In the mineral Muntz medium with oil and its derivatives (gasoline and others) (Content, wt.%: Potassium Nitrite – 0,4; Magnesium sulfite, hydrous – 0,08; sodium chloride – 0,1; Potassium Hydrophosphate K2 HPO4 -0,14; Potassium Dihydrophosphate KH2 PO4, 0.06; agar-2,0; oil or its derivatives– 1,0; distilled water – up to 100%, pH 7,2) the strain forms non-glossy, non-transparent grayish-yellow colonies 0.1-0.3 cm in diameter.
Physiological and enzymatic features.
The strain can accommodate to temperatures from +4 to +37°C. Under aerobic conditions it grows better. In completely anaerobic environment, the strain survives, but its replication slows down considerably. The best growth is observed at temperatures between of +20 and 30°C, and at pH between 6.0 and 8.0. The strain grows in a salt broth supplemented with 0.1-2.0% sodium chloride.
The bacterium is catalase positive and oxidase negative, and possesses metabolism of oxidative type. Enzymatic activity of the strain in regards of substrate fermentation is weak. It is not capable ofable to utilize polyhydric alcohols, poorly ferments glucose and sucrose, while other carbohydrates are not fermented at all. The strain does not utilize lysine and ornithine, neither does it use citrate and sodium malonate. It does not hydrolyze gelatin and starch. The strain is indole negative and behaves negative in the reaction of Voges - Proskauer. It does not contain phenylalanine desoxaminase, but has beta galactosidase.
Phylogenetic characteristics.
When the variable regions of 16Sp RNA were sequenced, the following nucleotide sequence for the tested strain was obtained:
(See Attachment)
Further analysis by RDP II 16Sр РНК database showed homology with the same bacterial species.
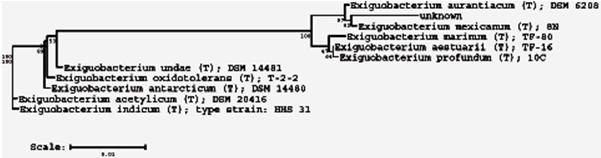
According to the sequence analysis, a phylogenetic tree with homologous strains was constructed (figure 1), where unknown is the researched strain
Fig. 1 - E. mexicanum phylogenetic tree
Sequences were aligned with the corresponding nearest sequences of bacterial species available from GenBank database. The results of processing of sequences using the software of the website RDB II (Ribosomal Database Project II), designed to determine the relationship of microorganisms and constructing phylogenetic trees, are presented in a graphic form.
Initial screening of GenBank database and RDP-II showed that the strain belongs to the following systematic groups of Bacteria; Firmicutes; Bacillales; Bacillaceae; Exiguobacterium, and homology to some species of the genus Exiguobacterium is 99%.
S000417444 0.938 0.711 1403 Exiguobacterium indicum (T); HHS 31; AJ846291 S000439502 0.940 0.723 1423 Exiguobacterium oxidotolerans (T); AB105164 S000466589 0.975 0.870 1405 Exiguobacterium profundum (T); 10C; AY818050 S000504076 0.977 0.898 1422 Exiguobacterium aestuarii (T); TF-16; AY594264 S000504078 0.973 0.872 1420 Exiguobacterium marinum (T); TF-80; AY594266 S000571530 0.990 0.972 1321 Exiguobacterium mexicanum (T); 8N; AM072764 S000650873 0.942 0.716 1458 Exiguobacterium antarcticum (T); DSM 14480; S000650874 0.943 0.725 1458 Exiguobacterium undae (T); DSM 14481; DQ019165 S000650875 0.990 0.930 1448 Exiguobacterium aurantiacum (T); DSM 6208; S000650876 0.936 0.709 1451 Exiguobacterium acetylicum (T); DSM 20416.
Allocation of a microbe to a particular species is based on homology of at least 97%. According to this criterion, the investigated strain can be any of several species of the genus Exiguobacterium. Analysis of phylogenetic relationships, built using standard strains of closely related bacteria showed that the best fit to the test strain is E. mexicanum.
Toxicology tests performed in white mice and seeds of the higher plants demonstrated that the strain is not virulent, non-toxic, non-toxigenic, and is not phytotoxic.
A study of oil-oxidizing activity of the E. mexicanum IPNG-ELA-5 strain found that, during 7 days culture in the mineral medium at +4°C the bacteria utilize 6,38 - 13,34% of oil and its derivatives; at +20°C this activity increased up to 24,62 - 54,60%; at +30°C reached 51,9 – 69,25%; at the temperature of +37°C could become as high as 57,78 - 81,46 %, depending on the xenobiotic type (table 1).
The ability of E. mexicanum, to recycle oil in the soil has been proved in a separate experiment set in an open ecosystem. The experiment was set on a heavy loam type of soil. Mean temperature of the soil during the whole period of the experiment was +8°C at the depth of 20 cm, and +14°C at the depth of 10 cm.
A suspension of E. mexicanum, with a titer of at least 109 cells / cm3 was added into the contaminated soil at the amount of 1 liter of the product per 1 m3 of soil.
The soil was mixed thoroughly using shovels and left for 60 days. Degradation of oil by the bacteria determined at the end of the incubation was of 86.9% (table 2).
Table 1 - The degree of utilization of oil and oil products
| Option experience | t°С | ||||
| Xenobiotic, mg/dm3 | The term | +4 | +20 | +30 | +37 |
| Oil | Before experience | 1000,0 | 1000,0 | 1000,0 | 1000,0 |
| after experience | 936,2 | 502,4 | 481,0 | 422,2 | |
| % degradation | 6,38 | 49,76 | 51,9 | 57,78 | |
| Diesel fuel | before experience | 1000,0 | 1000,0 | 1000,0 | 1000,0 |
| after experience | 902,0 | 454,8 | 333,3 | 185,4 | |
| % degradation | 9,8 | 54,60 | 66,67 | 81,46 | |
| Oil motor | before experience | 1000,0 | 1000,0 | 1000,0 | 1000,0 |
| after experience | 866,6 | 753,8 | 400,6 | 398,2 | |
| % degradation | 13,34 | 24,62 | 59,94 | 60,18 | |
| Gas condensate | before experience | 1000,0 | 1000,0 | 1000,0 | 1000,0 |
| after experience | 923,6 | 533,9 | 307,5 | 296,6 | |
| % degradation | 7,64 | 46,61 | 69,25 | 70,36 | |
Table 2 - The dynamics of degradation of oil in soil
| Variant of the experiment | Oil content before the experiment, mg/kg | Oil content, after the experiment, mg/kg | Destruction of oil, % |
| Soil + oil + cells of strain E. mexicanum | 34675 | 4517 | 86,97 |
| Soil + oil | 13771 | 12762 | 7,32 |
Conclusions: Our study demonstrates that the isolated procariote species E. mexicanum IPNG-ELA-5 rapidly (in 7 to 60 days) degrades oil and its derivatives. The bacterium can be used for cleaning of soils and for purification of water contaminated with oil and its derivatives in a wide temperature range from +4 to +37°C, making it a promising candidate microbe for biotechnological scale production.
Attachment: GACGGAACCCTTCGGGGGGAAGTCGACGGAATGAGCGGCGGACGGGTGAGTAACACGTAA AGAACCTGCCCTCAGGTCTGGGATAACCACGAGAAATCGGGGCTAATACCGGATGGGTCA TCGGACCGCATGGTCCGAGGATGAAAGGCGCTTCGGCGTCGCCTGGGGATGGCTTTGCGG TGCATTAGCTAGTTGGTGGGGTAATGGCCCACCAAGGCGACGATGCATAGCCGACCTGAG AGGGTGATCGGCCACACTGGGACTGAGACACGGCCCAGACTCCTACGGGAGGCAGCAGTA GGGAATCTTCCACAATGGACGAAAGTCTGATGGAGCAACGCCGCGTGAACGATGAAGGCC TTCGGGTCGTAAAGTTCTGTTGTAAGGGAAGAACAAGTGCCGCAGGCAATGGCGGCACCT TGACGGTACCTTGCGAGAAAGCCACGGCTAACTACGTGCCAGCAGCCGCGGTAATACGTA GGTGGCAAGCGTTGTCCGGAATTATTGGGCGTAAAGCGCGCGCAGGCGGCCTCTTAAGTC TGATGTGAAAGCCCCCGGCTCAACCGGGGAAGGCNTTGAACGGGAAGCTGAGTATAСписок литературы / References
- Гриценко А.И., Акопова Г.С., Максимов В.М. Экология. Нефть и газ. – М.: Наука, 1997. – 557 с.
- Пиковский Ю.И. Трансформация техногенных потоков нефти в почвенных экосистемах // Восстановление нефтезагрязненных почвенных экосистем. – М.: Наука, 1988. – С. 7-31.
- Оборин A.A., Калачникова И.Г., Масливец Т.А., Базенкова Е.И., Плещева О.В., Оглоблина А.И. // Восстановление нефтезагрязнённых почвенных экосистем. — М.: Наука, 1988. С. 140-159.
- Foght J.M., Westlike D.W.S. Bioremediation of oil spills // Spill Technol. Newsiett., 1992. – V. 17. – P. 1-10.
- Коронелли Т.В., Комарова Т.И., Ильинский В.В. и др. Интродукция бактерий рода Rhodococcus в тундровую почву, загрязненную нефтью // Прикладная биохимия и микробиология. 1997. Т. 33. №2. С. 198-201.
- Керстен Д.К. Морфологические и культуральные свойства индикаторных микроорганизмов нефтегазовой съемки // Микробиология, 1963,№5, С.1024-1030.
- Чалая О.Н., Зуева И.Н., Лифшиц С.Х., Трущелева Г.С., Иванова И.К. Состав и свойства нефти Талаканского месторождения //Малотоннажная переработка нефти и газа в Республике Саха (Якутия): Материалы конференции (26-27 июля 2001 г., г. Якутск). – Якутск, Изд-во ЯЧНЦ СО РАН, 2001. – С.165-170.
- Сакович Г.С., М.А. Безматерных Физиология и количественный учет микроорганизмов: Методические указания. - Екатеринбург: ГОУ ВПО УГТУ-УПИ, 2005. - 40 с.
- Красильников Н. А. Определитель бактерий и актиномицетов, М. — Л., Изд. АН СССР,1949. - 830 с.
- Определитель бактерий Берджи / под Ред. Д. Хоулта, Н. Крига, П. Снута и др. М.: Мир, 1997, т. 1-2. - 800 с.
- PCR Protocols. A Guide to methods and applications. Innis M, Gelfand D., Sninsky J. - Р.14-15.
- Каталог MBI Fermentas 1998\1999. – Р. 146-157.
- Pavlicek A et al "Fre-Tree-freeware program for construction of phylogenetic trees on the basis of distance data and bootstrap\jackknife analysis of tree robustness. " Application in the RAPD analysis of genus Frenkelia Folia Biol (Praha) 1999 45(3). – Р. 97-99. 14. Ribosomal Database Project II (http://rdp.cme.msu.edu).
- Егоров Н.С. Основы учения об антибиотиках М.: Высшая школа, 1986. - 448 с.
- ФР.1.31.2007.03234 МВИ 01.02.117 Методика выполнения измерений массовой концентрации нефтепродуктов в питьевых, природных и сточных водах ИК-спектрометрическим методом с использованием концентратомера «ИКН-025».
- РД 52.18.647-2003. Методические указания определения массовой доли нефтепродуктов в почвах. Методика измерений гравиметрическим методом.
Список литературы на английском языке / References in English
- Gritsenko A. I., Akopova G. S., Maksimov, V. M. Ecology. Oil and gas. – M.: Nauka, 1997. – 557 p.
- Pikovsky, Y. I. Transformation of technogenic flows of petroleum in soil ecosystems // Restore oil-polluted soil ecosystems. – M.: Nauka, 1988. – P. 7-31.
- Oborin A. A., Kalashnikova I. G., maslivets A. T., Bazankova E. I., Plesea O. V., Ogloblina, A. I. // Restore oil-polluted soil ecosystems. — M.: Nauka, 1988. - P. 140-159.
- J. M. Foght, D. W. S. Westlike Bioremediation of oil spills // Spill Technol. Newsiett., 1992. – V. 17. – P. 1-10.
- Koronelli T. V., Komarova T. I., Ilyinsky V. V., Introduction bacteria of the genus Rhodococcus in tundra soil contaminated with oil // Applied biochemistry and Microbiology. 1997. T. 33. No. 2. - P. 198-201.
- Kersten, D. K. Morphological and cultural properties of indicator microorganisms in the oil and gas shooting // Microbiology, 1963, No. 5, - P. 1024-1030.
- Chalaya O. N., Zueva I. N., Lifschitz C. H., Trushelev G. S., Ivanov K. I. the Composition and properties of the Talakan oil Deposit //small-scale processing of oil and gas in the Republic of Sakha (Yakutia): Materials of the conference (July 26-27, 2001, Yakutsk). – Yakutsk, Publishing house of SB RAS ACNC, 2001. – P. 165-170.
- Sakovich G. S., Bezmaternyh M. A. Physiology and quantitative accounting of microorganisms: a manual. - Yekaterinburg: GOU VPO UGTU-UPI, 2005. - 40 p.
- Krasilnikov N. A. the Determinant of bacteria and actinomycetes, M. — L., Ed. AN SSSR,1949. - 830 p.
- The determinant of bacteria Berge / Ed. D. Holt, N. Krieg, P. Sota, etc. M.: Mir, 1997, vol. 1-2. - 800 P.
- PCR Protocols. A Guide to methods and applications. Innis M, Gelfand D., Sninsky J. - R. 14-15.
- Directory MBI Fermentas 1998\1999. – P. 146-157.
- Pavlicek A, et al "Fre-Tree-freeware program for construction of phylogenetic trees on the basis of distance data and bootstrap\jackknife analysis of tree robustness. Application in the RAPD analysis of genus Frenkelia. Folia Biol (Praha) 1999 45(3). – P. 97-99.
- Ribosomal Database Project II (http://rdp.cme.msu.edu).
- Egorov N. Basic principles of antibiotics, Moscow: Higher school, 1986. - 448 p.
- FR.1.31.2007.03234 MIM 01.02.117 procedure of measurement of mass concentration of petroleum products in drinking, natural and wastewater. IR-spectrometric method using concentratometer "IRH-025".
- RD 52.18.647-2003. How to determine the mass fraction of oil products in soil. Measurement technique gravimetric method.