МИКСОПЛОИДНОСТЬ СОРТА ВИНОГРАДА АВРОРА МАГАРАЧА
МИКСОПЛОИДНОСТЬ СОРТА ВИНОГРАДА АВРОРА МАГАРАЧА
Научная статья
Клименко В.П.*
ORCID: 0000-0002-7452-0776,
Всероссийский национальный научно-исследовательский институт виноградарства и виноделия «Магарач»,
Ялта, Россия
* Корреспондирующий автор (vikpaulklim[at]mail.ru)
АннотацияЦелью данной работы является оценка встречаемости клеток с отклонением от диплоидности в меристематической ткани растений сорта винограда Аврора Магарача. Всего исследовали 6812 клеток. В ткани растений сорта Аврора Магарача, в отличие от исходного сорта Совиньон зеленый, наблюдали миксоплоидию. В меристематической ткани растений встречали и учитывали триплоидные клетки с числом хромосом 3n = 57. Средняя частота триплоидных клеток в наблюдениях составила 0,64, частота варьировала в зависимости от группы наблюдения, от 22,1 до 98,2 %. Вариация объясняется встречающейся у винограда химерностью.
Ключевые слова: виноград, митоз, миксоплоидия, триплоид, химерность.
MIXOPLOIDY OF GRAPE VARIETY AURORA MAGARACH
Research article
Klimenko V.P.*
ORCID: 0000-0002-7452-0776,
All-Russian National Research Institute of Viticulture and Winemaking "Magarach", Yalta, Russia
* Corresponding author (vikpaulklim[at]mail.ru)
AbstractThe evaluation of the emergence of cells with the deviation from diploidy in the meristematic tissue of plants of the grape variety Aurora Magarach was the aim of this work. A total of 6812 cells were examined. The mixoploidy in the plant tissue of the variety Aurora Magarach was observed, in contrast to the initial variety Sauvignon Vert. Triploid cells with a chromosome number of 3n = 57 were met and taken into account in the meristematic tissue of plants. The average frequency of triploid cells in the observations was 0.64, the frequency varied depending on the group of observations, from 22.1 to 98.2%. The variation is explained by the chimera encountered in grapes.
Keywords: grape, mitosis, mixoploidy, triploid, chimera.
IntroductionFor a long time, there was the unreasonably little work on mutagenesis, polyploidy and cytogenetics of grapes. According to the development of embryo rescue technologies, modern cytogenetic and molecular genetic methods, the number of studies in this area has increased significantly; these approaches are used in breeding and implemented as new varieties. The most important results were obtained in the creation and study of triploids.
Interploid crossings have led to the appearance of many grape triploid hybrids and varieties [1]. An effective system for the breeding of triploid seedless grapes was created using embryo rescue [2]. The study of the influence of the level of ploidy on the formation of parthenocarpic berries in diploid and tetraploid grape varieties led to the assumption that the development of seedless berries can be enhanced by increasing the level of ploidy from diploid to tetraploid [3]. The results of reciprocal crosses indicated that the yield promising triploids decreased when tetraploid varieties used as the maternal forms [4]. The study of the inheritance of sugar and organic acids in berries according to the tetraploid × diploid variety crossing scheme showed that the sugar content turned out to be additive with increasing ploidy level, the total acid content decreased with increasing ploidy level [5]. Certain progress has been achieved in endosperm culture to obtain triploid plants [6]. A cytogenetic analysis of grape plants obtained from embryoids in a tissue culture made it possible to establish the presence of mixoploidy [7], [8]. Triploid plants are of great economic importance and are promising for the creation of new plant varieties. Due to the rapid progress in DNA sequencing technology, triploids may be the focus of genomic research in the future and create unprecedented opportunities for the monitoring of genomic changes in unbalanced genomes [9, P. 49].
Despite the successful results obtained, studies on polyploidy and cytogenetics of grapes in Russia have not yet reached an acceptable scale and level [10]. Some new grape varieties were obtained using induced mutagenesis and polyploidy. The known grape variety Aurora Magarach was obtained as a result of treating stratified seeds of random pollination of Sauvignon Vert by the mutagen ethyleneimine at a concentration of 0.05% [11, P. 101–106]. A cytogenetic analysis of this variety has not yet been performed.
The evaluation of the emergence of cells with the deviation from diploidy in the meristematic tissue of plants of the grape variety Aurora Magarach was the aim of this work.
Methods
The plants of variety Sauvignon Vert of species Vitis vinifera Linn. and variety Aurora Magarach, the complex interspecific hybrid of the genus Vitis (Tournef.) Linn., were the material for cytogenetic research. A total of 9 groups of observations were made. Laboratory studies were carried out at the Institute "Magarach", Yalta, in 2015-2018. The cytogenetic analysis was carried out according to the recommended methods for grape [12, P. 37–42]. The 3-5 mm long roots of sprouted shortened cuttings were used as objects; the meristem of the root apex was examined. The 3-5 temporal acetocarmine preparations were made for each variant. The XSP-146TP biological microscope ("Ulab", ТМ), the Granum DC 1300 video camera ("NVL Granum", Ltd.), and the Canon Powershoot A620 digital camera ("Canon", Inc.) were used for cytogenetic analysis. A total of 6812 root meristematic cells of grape were examined. The mitotic index was calculated as the proportion of cells dividing in mitosis from the total number of analyzed cells. The proportion of cells deviating from diploidy was calculated as the ratio of the number of cells with this type of ploidy to the total number of cells in metaphase and expressed as a percentage.
The statistical processing of data was carried out according to common accepted methods [13, P. 37–45, 111–127] using the analytics software package Statistica 10 ("StatSoft", Inc.). Experiments were carried out in 3 replicates. The mean (M) and standard deviations (± SD) were calculated and presented in the table. The significance of the difference between the variants was evaluated using Student's t-test. Differences between variants were considered statistically significant at a confidence level of p ≤ 0.05.
Results
All stages of mitosis were observed during cytogenetic experiments. A total of 2473 cells were found in the division state (see table 1). The mitotic index varied significantly.
The mixoploidy in the meristematic plant tissue of the variety Aurora Magarach was observed, in contrast to the initial variety Sauvignon Vert. Triploid cells with a chromosome number of 3n = 57 were met and taken into account in the meristematic tissue of plants (see figure 1).
A total of 2217 cases of deviations from diploidy were observed, the frequency of deviations varied depending on the observation group. The variation is explained by the chimera encountered in grapes. Most triploid cells were observed in the observation group 2.
Table 1 – The results of evaluation of the deviation from diploidy in grape varieties
Sauvignon Vert and Aurora Magarach (М±SD, Laboratory of Institute "Magarach", Yalta, 2015-2018)
| Observation group | Deviations from diploidy in variety Sauvignon Vert | Deviations from diploidy in variety Aurora Magarach | ||||
| number of cells, PC. | mitotic index,% | triploid cells,% | number of cells, PC. | mitotic index,% | triploid cells,% | |
| 1 | 343 | 36.2±0.4 | 0 | 308 | 33.4±0.5 | 49.8±2.4 |
| 2 | 379 | 18.8±0.4 | 0 | 301 | 49.7±0.5 | 98.2±2.0 |
| 3 | 330 | 34.3±0.4 | 0 | 342 | 41.7±0.4 | 86.7±2.1 |
| 4 | 365 | 22.5±0.4 | 0 | 323 | 48.5±0.4 | 58.8±2.0 |
| 5 | 374 | 28.7±0.4 | 0 | 304 | 46.5±0.5 | 22.1±2.1 |
| 6 | 331 | 41.1±0.4 | 0 | 317 | 47.2±0.4 | 81.3±2.0 |
| 7 | 301 | 36.4±0.5 | 0 | 513 | 46.8±0.3 | 48.2±1.6 |
| 8 | 431 | 26.9±0.3 | 0 | 361 | 31.6±0.4 | 66.7±2.3 |
| 9 | 486 | 32.7±0.3 | 0 | 703 | 29.7±0.5 | 68.2±2.4 |
Earlier studies of the morphological features of tetraploid and triploid grape hybrids showed that the leaves of them, compared with diploid plants, are more solid, very large and dense, intense green color, with pointed teeth [12, P. 180–198]. Triploidy increases the size of somatic cells and the number of chloroplasts, which leads to increased photosynthesis and resistance to stress [6], [9, P. 43]. Therefore, many triploid plants are more vigorous than their diploid prototypes, and have wide, thick, dark green leaves. Studies have shown that the thickness of the leaf spongy tissue of the mutant is greater than that of the original variety, and the differences are significant [14]. Seeds obtained from triploids are generally not viable [12, P. 215-226]. In cases where some seeds germinated, the set of chromosomes in seedlings was diploid.
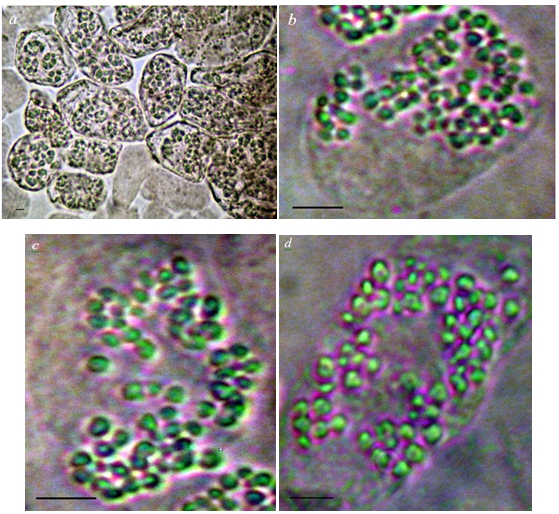
Fig. 1 – Mixoploidy in the meristematic tissue of plants of the grape variety Aurora Magarach: a – a group of mixoploid cells; b, c, d – triploidy (3n =57)
Notes: scale – 5 μM
Variety Aurora Magarach differs from the initial variety Sauvignon Vert by vigor vines, larger clusters and larger berries, as well as resistance to diseases and pests. These properties could be used in the grape breeding, but the viability of the offspring of mutants and polyploids is low [15, P. 49]. The leaf of this variety is rather large, very dense, weakly dissected, lateral clippings are small and medium, open, barely marked. The teeth on the edge of the leaf are small and pointed. The lamina is smooth, shiny and very dense, dark green color persists until defoliation. Therefore, the characteristic of the variety Aurora Magarach largely corresponds to the features of triploid forms of grapes.
It is not always possible to determine whether the plant is triploid or chimera. Mixoploid chimeras are one of the common results of interploid crosses or endosperm culture [9, P. 49]. In most cases, somatic mutations do not affect the whole plant; rather, they act on only one cell layer, leading to periclinal chimeras [16]. These structures are stable during vegetative propagation.
One of the features of the variety Aurora Magarach is the presence of a well-defined yellowish leaf edging, what is typical for metabolic disorders of magnesium in some grape varieties and may be due to genetic reasons. Variety Welschriesling is known for similar symptoms of inefficient assimilation of magnesium. Based on the analysis of the offspring from the Welschriesling × Sirius crossing, a genetic linkage map was constructed, which made it possible to identify the loci of signs associated with the visual manifestation of magnesium deficiency and its content in leaves [17]. Genetic factor is one of the most important reasons for the lack of chlorophyll pigments and chloroplast membranes in plants [18]. Advances in the study of the fundamental principles of magnesium metabolism are necessary to improve the diagnosis of stress and selection using genetic markers.
Conclusion
Thus, the mixoploidy in the meristematic plant tissue of the variety Aurora Magarach was observed, in contrast to the parental variety Sauvignon Vert. The average frequency of triploid cells in the observations was 0.64. The results obtained can be used in the grape practical breeding for creating new varieties. In the future, the possibility of overcoming the lethality of generative offspring of the variety Aurora Magarach related to mixoploidy will be studied, using biotechnological methods.
| Финансирование | Funding |
| Работа выполнена как поисковое исследование. | This work was carried out as exploratory research. |
| Благодарности | Acknowledgement |
| Автор выражает искреннюю признательность к.б.н., доценту В. Ф. Безрукову за инспирацию данной работы. | The author thanks V. F. Bezrukov, Ph.D., associate professor, for the inspiration of this work. |
| Конфликт интересов | Conflict of Interest |
| Не указан. | None declared. |
Список литературы / References
- Yamada M. Advances in table grape breeding in Japan / M. Yamada, A. Sato // Breeding Science. – 2016). – Vol. 66 (1). – pp. 34–45. doi: 10.1270/jsbbs.66.34.
- Ji W. In vitro embryo rescue culture of F1 progenies from crosses between different ploidy grapes / W. Ji, G. R. Li, Y. X. Luo and others // Genetics and Molecular Research. – – Vol. 14 (4). – pp. 18616–18622. doi: 10.4238/2015.
- Sarikhani H. Effect of ploidy on parthenocarpy in grape cultivars / H. Sarikhani, A. Wakana // Acta Horticulturae. – 2009. – Vol. 827. – pp. 433–438. doi: 10.17660/ActaHortic. 2009. 827.74.
- Sun L. The study of triploid progenies crossed between different ploidy grapes / L. Sun, G. Zhang, A. Yan and others // African Journal of Biotechnology. – 2011. – Vol. 10 (32). – pp. 5967–5971. doi: 10.5897/AJB10.1850.
- Liang Z. Inheritance of sugar and acid contents in the ripe berries of a tetraploid × diploid grape cross population / Z. Liang, M. Sang, A. Ma and others // Euphytica. – 2011. – Vol. 182. – pp. 251–259. doi: 10.1007/s10681-011-0487-x.
- Thomas Th.D. Endosperm culture: a novel method for triploid plant production / Th. D. Thomas, R. Chaturvedi // Plant Cell, Tissue and Organ Culture. – 2008. – Vol. 93. – pp. 1–14. doi: 10.1007/s11240-008-9336-6.
- Марченко А.О. Соматический эмбриоидогенез в культуре ткани винограда / А.О. Марченко, П. Я. Голодрига, В. П. Клименко и др. // Физиология и биохимия культурных растений. – 1987. – Т. 19, № 4. – С. 408–411.
- Klimenko V. P. Pathological Mitosis and Mixoploidy in the Meristematic Tissues of Grape Plant / V. P. Klimenko // Russian Journal of Developmental Biology. – 2019. – 50(2). – pp. 31–38. doi: 10.1134/S1062360419020024.
- Wang X. Breeding Triploid Plants: A Review / X. Wang, Z.-M. Cheng, Sh. Zhi and others // Czech Journal of Genetics and Plant Breeding. – 2016. – Vol. 52(2). – pp. 41–54. doi: 10.17221/151/2015-CJGPB.
- Зленко В.А. Оптимизация методологии получения полиплоидных растений из почек винограда в культуре тканей in vitro / В.А. Зленко, В. В. Лиховской, В. А. Волынкин и др. // «Магарач». Виноградарство и виноделие. – 2017. – №1.– С. 3–5.
- Киреева Л.К. Новые методы в селекции винограда / Л.К. Киреева. – Ялта: ВНИИВиПП «Магарач», 1991. – 134 с.
- Топале Ш.Г. Кариология, полиплоидия и отдаленная гибридизация винограда / Ш.Г. Топале. – Кишинев: Ботанический сад АНМ, 2008. – 507 с.
- Лакин Г.Ф. Биометрия / Г.Ф. Лакин. – М.: Высшая школа, 1990. –352 с.
- Guo D. Histological and Molecular Characterization of Grape Early Ripening Bud Mutant / D. Guo, Y. Yu, F. Xi and others // International Journal of Genomics. – 2016. – Vol. 2016(4). – pp. 1–7. doi: 10.1155/2016/5620106.
- Клименко В.П. Генетические основы создания сортов винограда при участии источников ценных признаков с низкой фертильностью / В. П. Клименко, И. А. Павлова // «Магарач». Виноградарство и виноделие. – 2015. – № 3. – С. 47–49.
- Pelsy F. Molecular and cellular mechanisms of diversity within grapevine varieties / F. Pelsy // Heredity. – 2010. – Vol. 104(4). – pp. 331–340. doi: 10.1038/hdy.2009.161.
- Mandl K. A genetic map of Welschriesling × Sirius for the identification of magnesium-deficiency by QTL analysis / K. Mandl, J. L. Santiago, R. Hack and others // Euphytica. – 2006. – Vol. 149(1-2). – pp. 133–144. doi: 10.1007/s10681-005-9061-8.
- Kumari M. Albinism in plants: A major bottleneck in wide hybridization, androgenesis and doubled haploid culture. Critical Review / M. Kumari, J. C. Heather, S. Ian and others // Critical Reviews in Plant Science. – 2009. – Vol. 28. – pp. 393–409. doi: 10.1080/07352680903133252.
Список литературы на английском языке / References in English
- Yamada M. Advances in table grape breeding in Japan / M. Yamada, A. Sato // Breeding Science. – 2016). – Vol. 66 (1). – pp. 34–45. doi: 10.1270/jsbbs.66.34.
- Ji W. In vitro embryo rescue culture of F1 progenies from crosses between different ploidy grapes / W. Ji, G. R. Li, Y. X. Luo and others // Genetics and Molecular Research. – – Vol. 14 (4). – pp. 18616–18622. doi: 10.4238/2015.
- Sarikhani H. Effect of ploidy on parthenocarpy in grape cultivars / H. Sarikhani, A. Wakana // Acta Horticulturae. – 2009. – Vol. 827. – pp. 433–438. doi: 10.17660/ActaHortic. 2009. 827.74.
- Sun L. The study of triploid progenies crossed between different ploidy grapes / L. Sun, G. Zhang, A. Yan and others // African Journal of Biotechnology. – 2011. – Vol. 10 (32). – pp. 5967–5971. doi: 10.5897/AJB10.1850.
- Liang Z. Inheritance of sugar and acid contents in the ripe berries of a tetraploid × diploid grape cross population / Z. Liang, M. Sang, A. Ma and others // Euphytica. – 2011. – Vol. 182. – pp. 251–259. doi: 10.1007/s10681-011-0487-x.
- Thomas Th.D. Endosperm culture: a novel method for triploid plant production / Th. D. Thomas, R. Chaturvedi // Plant Cell, Tissue and Organ Culture. – 2008. – Vol. 93. – pp.1–14. doi: 10.1007/s11240-008-9336-6.
- Marchenko A. O. Somaticheskij jembrioidogenez v kul'ture tkani vinograda [Somatic embryoidogenesis in grape tissue culture] / A. O. Marchenko, P. Ya. Golodriga, V.P. Klimenko and others // Fiziologiya i biohimiya kulturnyh rastenij [Physiology and biochemistry of cultivated plants]. – 1987. – Vol. 19 (4). – pp. 408–411. [in Russian]
- Klimenko V. P. Pathological Mitosis and Mixoploidy in the Meristematic Tissues of Grape Plant / V. P. Klimenko // Russian Journal of Developmental Biology. – 2019. – Vol. 50(2). – pp. 31–38. doi: 10.1134/S1062360419020024.
- Wang X. Breeding Triploid Plants: A Review / X. Wang, Z.-M. Cheng, Sh. Zhi and others // Czech Journal of Genetics and Plant Breeding. –2016. – Vol. 52(2). – pp. 41–54. doi: 10.17221/151/2015-CJGPB.
- Zlenko V.A. Optimizacija metodologii poluchenija poliploidnyh rastenij iz pochek vinograda v kul'ture tkanej in vitro [Optimization of the methodology for producing polyploid plants from grape buds in an in vitro tissue culture] / Zlenko V. A., Lihovskoj V. V., Volynkin V. A. and others // "Magarach". Vinogradarstvo i vinodelie ["Magarach". Viticulture and winemaking]. – 2017. – № 1. – pp. 3–5. [in Russian]
- Kireeva L. K. Novye metody v selekcii vinograda [New methods in the grape breeding] / L. K. Kireeva. – Yalta: VNIIViPP "Magarach" [ASIIVaPP "Magarach"], 1991. – 134 p. [in Russian]
- Topale Sh.G. Kariologiya, poliploidiya i otdalennaya gibridizaciya vinograda [Karyology, Polyploidy, and Distant Hybridization in Grapevine] / Sh. G. Topale. – Chisinau: Botanicheskij sad ANM [Botanical Garden of the ASM], 2008. – 507 p. [in Russian]
- Lakin G. F. Biometriya [Biometry] / G. F. Lakin. – Moscow: Vysshaja shkola [Graduate School], 1990. – 352 p. [in Russian]
- Guo D. Histological and Molecular Characterization of Grape Early Ripening Bud Mutant / D. Guo, Y. Yu, F. Xi and others // International Journal of Genomics. – 2016. – Vol. 2016(4). – pp. 1–7. doi: 10.1155/2016/5620106.
- Klimenko V. P. Geneticheskie osnovy sozdanija sortov vinograda pri uchastii istochnikov cennyh priznakov s nizkoj fertil'nost'ju [Genetic basis for the creation of grape varieties with the participation of sources of valuable traits with low fertility] / V. P. Klimenko, I. A. Pavlova // "Magarach". Vinogradarstvo i vinodelie ["Magarach". Viticulture and winemaking]. – 2015. – № 3. – pp. 47–49. [in Russian]
- Pelsy F. Molecular and cellular mechanisms of diversity within grapevine varieties / F. Pelsy // Heredity. – 2010. – Vol. 104(4). – pp. 331–340. doi: 10.1038/hdy.2009.161.
- Mandl K. A genetic map of Welschriesling × Sirius for the identification of magnesium-deficiency by QTL analysis / K. Mandl, J. L. Santiago, R. Hack and others // Euphytica. – 2006. – Vol. 149(1-2). – pp. 133–144. doi: 10.1007/s10681-005-9061-8.
- Kumari M. Albinism in plants: A major bottleneck in wide hybridization, androgenesis and doubled haploid culture. Critical Review / M. Kumari, J. C. Heather, S. Ian and others // Critical Reviews in Plant Science. – 2009. – Vol. 28. – pp. 393–409. doi: 10.1080/07352680903133252.
