КООРДИНАЦИОННОЕ СОЕДИНЕНИЕ АЦЕТАТА МЕДИ (II) С 2-ФОРМИЛПИРИДИНОМ 4-АЛЛИЛТИОСЕМИКАРБАЗОНА ПРОЯВЛЯЕТ ИНГИБИТОРНУЮ АКТИВНОСТЬ В ОТНОШЕНИИ СУПЕРОКСИДНЫХ РАДИКАЛОВ
КООРДИНАЦИОННОЕ СОЕДИНЕНИЕ АЦЕТАТА МЕДИ (II) С 2-ФОРМИЛПИРИДИНОМ 4-АЛЛИЛТИОСЕМИКАРБАЗОНА ПРОЯВЛЯЕТ ИНГИБИТОРНУЮ АКТИВНОСТЬ В ОТНОШЕНИИ СУПЕРОКСИДНЫХ РАДИКАЛОВ
Научная статья
Пантеа В.1, *, Граур В.2, Андронаке Л.3, Гуля А.4, Цапков В.5, Сардарь В.6, Гаманюк М.7, Гудумак В.8
1 ORCID: 0000-0002-8835-6612;
2 ORCID: 0000-0001-8153-2153;
3 ORCID: 0000-0002-8781-8037;
4 ORCID: 0000-0003-2010-7959;
5 ORCID 0000-0003-1732-3116;
6 ORCID: 0000-0002-1047-9145;
7 ORCID: 0000-0002-7662-0973;
8 ORCID: 0000-0001-9773-1878;
1, 3, 6, 7, 8 Государственный университет медицины и фармации им. Н. Тестемицану, Кишинёв, Молдова;
2, 4, 5 Молдавский Государственный Университет, Кишинёв, Молдова
* Корреспондирующий автор (valeriana.pantea[at]usmf.md)
Аннотация
Получено новое биологически активное координационное соединение, относящееся к классу изотиосемикарбазидов переходных металлов - бис ((μ2-ацетато-o)-бис {[N-проп-2-ен-1-ил-N'- (пиридин -2-илметиллиден) карбамо-гидразонотиоат] меди} дигидрат. Установлено, что это соединение проявляет сильные антирадикальные свойства при взаимодействии органической молекулы с супероксидным радикалом. Благодаря этому свойству полученное соединение может найти широкое применение в медицине в качестве ингибитора супероксидных радикалов в организме человека, предотвращая, таким образом, повреждение клеток и тканей, атеросклероз и канцерогенез. Синтезируемое координационное соединение расширяет арсенал ингибиторов супероксид-радикалов, обладающих высокой биологической активностью.
Ключевые слова: ингибиторы супероксид-радикалов, координационное соединение, производные изотиосемикарбазида.
COPPER (II) ACETATE COORDINATION COMPOUND WITH 2-FORMYLPYRIDINE 4-ALLYLTHIOSEMICARBAZONE MANIFESTS INHIBITORY ACTIVITY AGAINST SUPEROXIDE RADICALS
Research article
Pantea V.1, *, Graur V.2, Andronache L.3, Gulea A.4, Tsapkov V.5, Sardari V.6, Gamaniuc M.7, Gudumac V.8
1 ORCID: 0000-0002-8835-6612;
2 ORCID: 0000-0001-8153-2153;
3 ORCID: 0000-0002-8781-8037;
4 ORCID: 0000-0003-2010-7959;
5 ORCID 0000-0003-1732-3116;
6 ORCID: 0000-0002-1047-9145;
7 ORCID: 0000-0002-7662-0973;
8 ORCID: 0000-0001-9773-1878;
1, 3, 6, 7, 8 State University of Medicine and Pharmacy “Nicolae Testemitanu”, Chisinau, Republic of Moldova;
2, 4, 5 Moldavian State University, Chisinau, Republic of Moldova
* Corresponding author (valeriana.pantea[at]usmf.md)
AbstractA new biologically active coordination compound, which belongs to the class of transition metal isotiosemicarbazides - bis (μ2-acetate-o) -bis {[N-prop-2-en-1-yl-N'- (pyridin- 2-ylmethylidene) carbamo-hydrazonothioate] copper} dihydrate was obtained. It has established that this compound exhibits strong anti-radical properties to the action of the organic molecule with the superoxide radical. Due to this property, the obtained compound can have a wide application in medicine as an inhibitor of superoxide radicals in the human body, thus preventing damage to cells and tissues, atherosclerosis and carcinogenesis. The synthesized coordination compound expands the arsenal of superoxide radical inhibitors with high biological activity.
Keywords: superoxide radical inhibitors, coordination compounds, isothiosemicarbazide derivates.
IntroductionStudies in recent years have provided increasing evidence that damage caused to cells by oxygen free radicals and nitrogen free radicals are the most important factors leading to aging and degenerative diseases such as cancer, cardiovascular diseases, cataracts, chronic inflammatory processes, renal failure, etc. [1], [2], [3]. The identification, study and testing of new remedies to correct disorders that occur as a result of the imbalance between oxidants and antioxidants, in favor of oxidants, having destructive and pathogenic potential in acute and chronic degenerative diseases (of the most common types of diseases), is of interest especially due to the increased incidence and severity of these pathologies.
However, there is a clear necessity for the development of new compounds, which could serve as a base for the development of medical preparations for the prevention and treatment of the mentioned above diseases.
The particular interest in this regard are thiosemicarbazide derivatives, which could exert a significant influence on the multiple free radical processes, which are forming in large quantities in many diseases and pathological processes.
Research over the last decade has revealed their therapeutic efficacy and their prospects for recovery [4], [5], [6].
At the same time, their mechanisms of biochemical action, especially on peroxidative processes, are not known in detail.
The aim of the study is to investigate the influence of new coordination compounds of copper, thiosemicarbazide derivatives on free radical oxidation processes, estimating and selecting the most effective compounds to combat diseases caused by excess of free radicals, and which can be used for prevention and treatment of acute and chronic degenerative diseases.
Respectively, one of the priority directions of modern applied chemistry is the synthesis of new compounds, which capture and neutralize superoxide radicals, thus preventing the development of cell and tissue damage, including inflammatory processes in the human body, atherosclerosis and carcinogenesis.
Material and methods
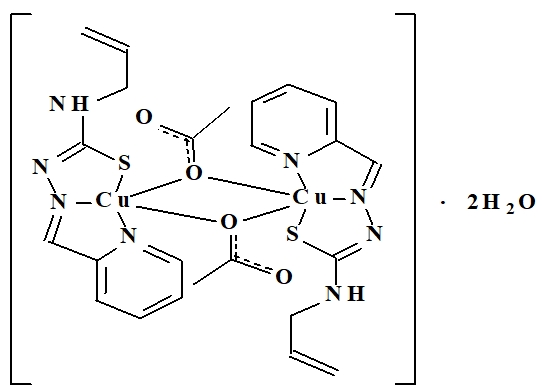
A new, biologically active, copper-coordination compound in the class of isothiosemicarbazides of transition metals - bis (μ2-acetate-o)-bis {[N-prop-2-en-1-yl-N'- (pyridin- 2-ylmethylidene) carbamo-hydrazonothioate] copper} dihydrate, of formula (Scheme 1) has been investigated. The structure of the investigated complex was established using X-ray diffraction studies. This compound has been synthesized [5], at the State University of Moldova in the Laboratory "Advanced materials in biopharmaceutics and technics" but its influence on oxidative processes with ROS, such as superoxide radical, has not been studied.
The scavenging activity of the superoxide radical was determined by the spectrophotometric method, described in [7], [8] with some modifications. This method is based on the generation of superoxide radicals by the reduced phenazine methosulfate / nicotinamide adenine dinucleotide (PMS / NADH) system by oxidation of NADH, and the superoxide radicals reduce the tetrazolium salt - Nitro Blue Tetrazolium (NBT) in blue-purple formazane.
The method was carried out as follows: the working dilutions of the tested substances in DMSO solution in concentrations 0.1; 1.0; 10.0; 100 µM / l were prepared. Then 20 µl of each working dilution of the tested substances into the wells of the 96-well microplate was pipetted. Each dilution was poured into duplicate. Then 180 µl of medium (working mixture) containing 20 mM phosphate buffer (pH 7.4), NADH (0.1 mM) and NBT (0.09 mM) was added. The control sample was mounted in the same way as the test sample, but instead of dilutions of the tested substances, an equivalent amount of 20 mM phosphate buffer solution (pH 7.4) was poured. It has been prepared into duplicate. After mixing, the absorbance at 560 nm [Ao] was measured. Then, in all wells, 20 μl of 8.0 μM phenazine methosulphate (FMS) solution was added, was stired for 10-15 s and was incubated at room temperature for 5 min, after which the absorbance [A1] was remeasured at 560 nm. The percentage of superoxide radical scavenging was calculated using the following formula:
Superoxide radical scavenging activity (%) = [(A0 –A1)/A0 x 100];
where A0 is the absorbance of the control and A1 is the absorbance of the tested compounds or the standard and / or reference substances.
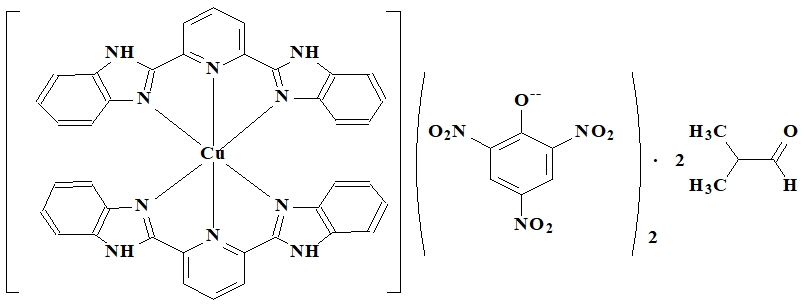
Quercetin (3,3', 4,5,6-pentahydroxyflavone) and coordination compound bis (2,4,6-trinitrophenolate) bis (2,2'-pyridin-2,6-diyl-kN)-bis-1H-benzimidazole] - copper (II) bis (N, N-dimethylformamide) solvates were used as a standard and reference substances for the determination of superoxide radical inhibitory activity.
Quercetin (3,3',4,5,6-pentahidroxiflavone) with the formula:
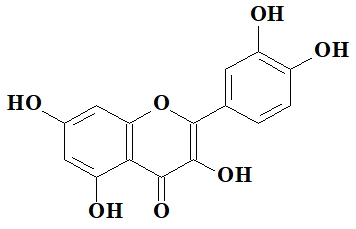
is a natural flavonol which possesses antioxidant and antiinflammatory effects, destroys cancer cells, prevents cardiovascular disease [9], [10], [11].
Coordination compound bis (2,4,6-trinitrophenolate) bis (2,2'-pyridin-2,6-diyl-kN)-bis-1H-benzimidazole] - copper (II) bis (N, N-dimethylformamide) solvates of the formula (Figure 1) is one of the synthetic chemical compounds with the highest antiradical activity described in the literature [12]:
Fig. 1 – The chemical structure of the compound bis (2,4,6-trinitrophenolate) bis (2,2'-pyridin-2,6-diyl-kN)-bis-1H-benzimidazole] - copper (II) bis (N, N-dimethylformamide) solvate [12]
Results and discussions
The compound bis (μ2-acetate-o)-bis {[N-prop-2-en-1-yl-N'-(pyridin-2-ylme thyl-idene) carbamo-hydrazonothioate] copper} dihydrate has anti-superoxide radical activity, with an IC50 of 0.35 ± 0.07 μM, which is 176.7 times higher than the activity of quercetin, used as a standard for the determination of superoxide radical inhibition activity and is 2.8 times more effective than prototype (Table 1).
Table 1 – The anti-superoxide radical activity of the researched compound compared to quercetin and prototype
| Compound | IC50, μM/L |
| Quercetin (3,3',4,5,6-pentahydroxyflavon) | 61,86±2,51 |
| Bis (2,4,6-trinitrophenolate) bis [2,2'-pyridin-2,6-diyl-kN)-bis-1H-benzimidazole]-copper (II) bis (N, N-dimethylformamide) solvate (prototype) | 0,99±0,09 |
| Bis (μ2-acetate-O)-bis {[N-prop-2-en-1-yl-N'- (pyridin-2-ylmethylidene) carbamo-hydrazonothioate] copper} dihydrate | 0,35±0,07 |
The disadvantage of quercetin is that, it does not have a high antiradical activity [half maximal inhibitory concentration (IC50) is only 61.86 ± 2.51μM / L], as well as causing toxic side effects [11].
From known synthetic chemicals, it has been established that in the case of bis (2,4,6-trinitrophenolate) bis [2,2'-pyridin-2,6-diyl-kN) -bis-1H-benzimidazole] -copper (II) bis (N, N-dimethylformamide) solvates (prototype) the half maximal superoxide radical inhibitory concentration (IC50) is = 0.99 ± 0.09 μM / L, which is 62.5 times higher than of quercetin. However, the given compound does not possess a sufficiently high superoxide radical inhibiting activity and has not been found wide application in medicine so far.
Established property of the compound bis (μ2-acetate-o)-bis {[N-prop-2-en-1-yl-N '- (pyridin-2-ylme thyl-idene) carbamo-hydrazonothioate] copper} dihydrate is new, because its use as an inhibitor of superoxide radicals has not been described so far.
Comparative analysis of bis (μ2-acetate-o)-bis {[N-prop-2-en-1-yl-N'- (pyridin-2-ylme thyl-idene) carbamo-hydrazonothioate] copper} dihydrate with prototype demonstrates that they differ, and they belong to different classes of copper (II) coordination compounds and a new combination of already known chemical bonds have been made in the investigated compound.
The process of obtaining of the above-mentioned compound is simple in execution, the starting substances are accessible [5]. The investigated complex is stable in contact with air, slightly soluble in water and aliphatic alcohols, is soluble in dimethylformamide and dimethylsulfoxide, practically insoluble in ether. The сonducted researches established that the investigated compound represents a coordination dimer, in which the elementary fragment represents a copper (II) complex with a distorted tetragonal-pyramidal structure (Figure 2).
Fig. 2 – Chemical structure of the compound bis (μ2-acetate-o)-bis {[N-prop-2-en-1-yl-N'- (pyridin-2-ylme thylidene) carbamo-hydrazonothioate] copper} dihydrate
In the inner sphere of the central atom is a tridentate thiosemicarbazone molecule, which coordinates to the copper atom through the pyridine nitrogen atoms [d (Cu-N) = 2.059 Å], azomethine [d (Cu-N) = 1.970 Å] and sulfur atom in deprotonated thiol form [d (Cu-S) = 1,736 Å], forming two metallocycles of five atoms. The double bond in the coordinated isothiosemicarbazone molecule is delocalized between the carbon and nitrogen atoms N4 [d(C3 - N4) = 1,337Å] and carbon and nitrogen azot N3 [d(C3 – N3) = 1,334Å]. The fourth and fifth places in the inner sphere of the copper atom are occupied by two oxygen atoms of acetate ions with distances d1 (Cu-O) = 1,954Å and d2 (Cu-O) = 2,429Å. In the outer sphere of the complex dimer there are two molecules of water of crystallization, which form hydrogen bonds with the coordination dimer. Other interatomic distances and valence angles are standard for compounds in this class.
Thus, based on the results of the element analysis, the physico-chemical research and the X-ray analysis, the composition and structure of the investigated compound has been established.
Use of a novel coordination compound - bis (μ2-acetate-o)-bis {[N-prop-2-en-1-yl-N'- (pyridin-2-ylme thyl-idene) carbamo-hydrazonothioate] copper} dihydrate expands the arsenal of compounds with high superoxide radical inhibitory activity.
Direct quantitative measurement of the superoxide radical (O2-.) is difficult due to its exceptional reactivity and short half-life. The most commonly used in biological and chemical systems are the classical methods that use the PMS / NADH NBT system and that allow to determine the activity of superoxide radical scavenging.
As mentioned above, repeated exposure to these radicals is considered a major cause of aging, neurodegenerative and inflammatory diseases due to the gradual deterioration of major cellular components, such as DNA and proteins. In pathogenic process of acute and chronic degenerative diseases (the most common diseases) an important role is being attributed to reactive oxygen and nitrogen species (ROS / RNS), in particular, the superoxide radical, which from a biological view, can be generated from two major sources: mitochondrial respiratory chain and NADPH oxidase (nicotinamide adenine dinucleotide phosphate oxidase) - an enzyme complex found in the plasma membrane, as well as in the membranes of phagosomes of nucleate polymorphic leukocytes of the blood to destroy microorganisms [13].
However, the superoxide radical (O2-.) is the product of the mitochondrial respiratory chain and a crucial component of the immune system. Due to high reactivity of superoxide radicals (O2-.) - through their potential to oxidize nucleic acids, proteins, lipids or carbohydrates, they are responsible for multiple harmful actions in the body, such as inflammation, cancer, cardiovascular disease, hypertension, ischemia /reperfusion, diabetes mellitus, neurodegenerative diseases (Alzheimer's and Parkinson's disease), rheumatoid arthritis, alcohol-induced liver disease, ulcerative colitis, senescence and atherosclerosis. Antioxidants through their ability to eliminate free radicals (FR) present in biological systems from a wide variety of endogenous and / or exogenous sources, limit the harmful effects of FR, allowing the body to fight efficiently in various pathological situations, limiting the injuries, and do not allow their spread [14], [15], [16].
Therefore, the therapeutic inhibition of the superoxide radical is a new contribution, because the compounds with antiradical activity show a strong curative effect, thus preventing multiple harmful actions in the body.
Further studies have to confirm the therapeutic utility of this bioactive compound under investigation.
| Финансирование Это исследование проведено в рамках Государственной программы (2020-2023) Республики Молдова (исследовательский грант № 20.80009.5007.10). | Funding This study has been supported by the State Program (2020-2023) of the Republic of Moldova (research grant No. 20.80009.5007.10). |
| Конфликт интересов Не указан. | Conflict of Interest None declared. |
Список литературы / References
- Lien Ai Pham-Huy. Free Radicals, Antioxidants in Disease and Health / Lien Ai Pham-Huy, Hua He, Chuong Pham-Huy // Int J Biomed Sci. 2008 Jun, 4(2): pp. 89–96.
- Babizhayev M.A. Reactive Oxygen Species and the Aging Eye: Specific Role of Metabolically Active Mitochondria in Maintaining Lens Function and in the Initiation of the Oxidation-Induced Maturity Onset Cataract - A Novel Platform of Mitochondria-Targeted Antioxidants With Broad Therapeutic Potential for Redox Regulation and Detoxification of Oxidants in Eye Diseases / M.A. Babizhayev, Y.E. Yegorov // Am J Ther. 2016 Jan-Feb;23(1):e98-117. DOI: 1097/MJT.0b013e3181ea31ff. PMID: 21048433.
- Dhaliwal J.S. Free Radicals and Anti-oxidants in Health and Disease / J.S. Dhaliwal, H. Singh // Int J Oral Health Med Res 2015;2(3):97-99.
- Iakovidis I. Copper and Its Complexes in Medicine: A Biochemical Approach / I. Iakovidis, I. Delimaris, M. Piperakis // Molecular Biology International. 2011, p. 13. DOI: 10.4061/2011/594529.
- Graur V. Designul şi sinteza compuşilor biologic activi ai metalelor 3d cu 4-alilcalcogensemicarbazone şi derivaţii lor / Graur. Autoreferat al tezei de doctor în științe chimice. Chișinău 2017, 30 p.
- Balan G. Novel 2-formylpyridine 4-allyl-S-methylisothiosemicarbazone and Zn(II), Cu(II), Ni(II) and Co(III) complexes: Synthesis, characterization, crystal structure, antioxidant, antimicrobial and antiproliferative activity / G. Balan, O. Burduniuc, I. Usataia et al. // Appl Organometal Chem. 2019; e5423.
- Robak J. Flavonoids are scavengers of superoxides anions /Robak, R.J. Gryglewski // Biochem Pharmacol, 37 (1988), pp. 837-841.
- Fontana M. Interaction of enkephalines with oxyradicals / M. Fontana, L. Mosca, M.A. Rosei // Biochemical Pharmacology, 2001; Vol.61. pp 1253-1257.
- David A. Overviews of Biological Importance of Quercetin: A Bioactive Flavonoid / David A., Arulmoli R., Parasuraman S. // Pharmacogn. 2016, Jul-Dec; 10(20), pp. 84–89.
- Srivastava S. Quercetin, a Natural Flavonoid Interacts with DNA, Arrests Cell Cycle and Causes Tumor Regression by Activating Mitochondrial Pathway of Apoptosis / Srivastava, S., Somasagara, R., Hegde, M. et al.// Sci Rep6, 24049 (2016).
- Andres S. Safety Aspects of the Use of Quercetin as a Dietary Supplement / Andres S, Pevny S, Ziegenhagen R. et al. // Mol Nutr Food Res. 2018 Jan;62(1). DOI: 10.1002/mnfr.201700447. Epub 2017 Dec 19. PMID: 29127724.
- Hui-Lu Wu. Copper(II) complex based on a V-shaped ligand, 2,6- bis (2-benzimidazolyl)-pyridine: synthesis, crystal structure, DNA-binding properties, and antioxidant activities / Hui-Lu Wu, Xingcai Huang, Bin Liu et al. // Journal of Coordination Chemistry, 64:24, 4383-4396, DOI: 10.1080/00958972.2011.640934
- Hayyan M. Superoxide Ion: Generation and Chemical Implications / Hayyan M., Hashim M.A., AlNashef I.M. // Chem Rev. 2016 Mar 09;116(5):3029-3085.
- Buetler T. Role of Superoxide as a Signaling Molecule / Buetler, Timo & Krauskopf, Alexandra & Ruegg, Urs // News Physiol Sci 19: 120-123, 2004;
- Varela-Chinchilla C.D. Biochemistry, Superoxides / Varela-Chinchilla C.D., Farhana A. // In: StatPearls. [Electronic resource]. URL: https://www.ncbi.nlm.nih.gov/books/NBK555982/. (accessed 12/07/2021)
- Sies H. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents / Helmut Sies, Dean P. Jones. // Nature Reviews Molecular Cell Biology (2020), volume21, pages363–383.
Список литературы на английском языке / References in English
- Lien Ai Pham-Huy. Free Radicals, Antioxidants in Disease and Health / Lien Ai Pham-Huy, Hua He, Chuong Pham-Huy // Int J Biomed Sci. 2008 Jun, 4(2): pp. 89–96.
- Babizhayev M.A. Reactive Oxygen Species and the Aging Eye: Specific Role of Metabolically Active Mitochondria in Maintaining Lens Function and in the Initiation of the Oxidation-Induced Maturity Onset Cataract - A Novel Platform of Mitochondria-Targeted Antioxidants With Broad Therapeutic Potential for Redox Regulation and Detoxification of Oxidants in Eye Diseases / M.A. Babizhayev, Y.E. Yegorov // Am J Ther. 2016 Jan-Feb;23(1):e98-117. DOI: 1097/MJT.0b013e3181ea31ff. PMID: 21048433.
- Dhaliwal J.S. Free Radicals and Anti-oxidants in Health and Disease / J.S. Dhaliwal, H. Singh // Int J Oral Health Med Res 2015;2(3):97-99.
- Iakovidis I. Copper and Its Complexes in Medicine: A Biochemical Approach / I. Iakovidis, I. Delimaris, M. Piperakis // Molecular Biology International. 2011, p. 13. DOI: 10.4061/2011/594529.
- Graur V. Designul şi sinteza compuşilor biologic activi ai metalelor 3d cu 4-alilcalcogensemicarbazone şi derivaţii lor [Design and synthesis of biologically active compounds of 3d metals with 4-allylalkogensemicarbazone and their derivatives] / Graur. Autoreferat al tezei de doctor în științe chimice. Chișinău 2017, 30 p. [in Romanian].
- Balan G. Novel 2-formylpyridine 4-allyl-S-methylisothiosemicarbazone and Zn(II), Cu(II), Ni(II) and Co(III) complexes: Synthesis, characterization, crystal structure, antioxidant, antimicrobial and antiproliferative activity / G. Balan, O. Burduniuc, I. Usataia et al. // Appl Organometal Chem. 2019; e5423.
- Robak J. Flavonoids are scavengers of superoxides anions /Robak, R.J. Gryglewski // Biochem Pharmacol, 37 (1988), pp. 837-841.
- Fontana M. Interaction of enkephalines with oxyradicals / M. Fontana, L. Mosca, M.A. Rosei // Biochemical Pharmacology, 2001; Vol.61. pp 1253-1257.
- David A. Overviews of Biological Importance of Quercetin: A Bioactive Flavonoid / David A., Arulmoli R., Parasuraman S. // Pharmacogn. 2016, Jul-Dec; 10(20), pp. 84–89.
- Srivastava S. Quercetin, a Natural Flavonoid Interacts with DNA, Arrests Cell Cycle and Causes Tumor Regression by Activating Mitochondrial Pathway of Apoptosis / Srivastava, S., Somasagara, R., Hegde, M. et al.// Sci Rep6, 24049 (2016).
- Andres S. Safety Aspects of the Use of Quercetin as a Dietary Supplement / Andres S, Pevny S, Ziegenhagen R. et al. // Mol Nutr Food Res. 2018 Jan;62(1). DOI: 10.1002/mnfr.201700447. Epub 2017 Dec 19. PMID: 29127724.
- Hui-Lu Wu. Copper(II) complex based on a V-shaped ligand, 2,6- bis (2-benzimidazolyl)-pyridine: synthesis, crystal structure, DNA-binding properties, and antioxidant activities / Hui-Lu Wu, Xingcai Huang, Bin Liu et al. // Journal of Coordination Chemistry, 64:24, 4383-4396, DOI: 10.1080/00958972.2011.640934
- Hayyan M. Superoxide Ion: Generation and Chemical Implications / Hayyan M., Hashim M.A., AlNashef I.M. // Chem Rev. 2016 Mar 09;116(5):3029-3085.
- Buetler T. Role of Superoxide as a Signaling Molecule / Buetler, Timo & Krauskopf, Alexandra & Ruegg, Urs // News Physiol Sci 19: 120-123, 2004;
- Varela-Chinchilla C.D. Biochemistry, Superoxides / Varela-Chinchilla C.D., Farhana A. // In: StatPearls. [Electronic resource]. URL: https://www.ncbi.nlm.nih.gov/books/NBK555982/. (accessed 12/07/2021)
- Sies H. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents / Helmut Sies, Dean P. Jones. // Nature Reviews Molecular Cell Biology (2020), volume21, pages363–383.