СРАВНЕНИЕ МАЦЕРИРУЮЩЕЙ СПОСОБНОСТИ РАСТИТЕЛЬНЫХ МАСЕЛ НА ПРИМЕРЕ ВОЗДУШНО-СУХОГО СЫРЬЯ ЛАМИНАРИИ САХАРИСТОЙ (LAMINARIA SACCHARINA, L., LAMINARIACEAE)
Пампуха А.Г.
Студент 4 курса стоматологического факультета, ГБОУ ВПО «Северный государственный медицинский университет», г. Архангельск
СРАВНЕНИЕ МАЦЕРИРУЮЩЕЙ СПОСОБНОСТИ РАСТИТЕЛЬНЫХ МАСЕЛ НА ПРИМЕРЕ ВОЗДУШНО-СУХОГО СЫРЬЯ ЛАМИНАРИИ САХАРИСТОЙ (LAMINARIA SACCHARINA, L., LAMINARIACEAE)
Аннотация
Целью настоящего исследования явилось изучение мацерирующей способности растительных масел на примере воздушно-сухого сырья (Laminaria saccharina, L., Laminariaceae). Для определения суммы хлорофиллов в мацератах использовали метод УФ-спектроскопии. В качестве растворителя применяли ацетон. В результате проведенных исследований установлено, что наиболее перспективным для получения суммы хлорофиллов из воздушно-сухого сырья ламинарии сахаристой является рыжиковое масло.
Ключевые слова: Laminaria saccharina, хлорофилл, растительное масло, мацерация
Pampukha A.G.
4th year student of the dentistry department, Northern State medical university, Arkhangelsk
THE COMPARISON OF VEGETABLE OILS MACERATION OF LAMINARIA SACCHARINA (LAMINARIACEAE L.)
Abstract
The aim of this study was to investigate the substantial differences of edible vegetable oils maceration of brown algae Laminaria saccharina (Laminariaceae L.) (L. saccharina) dry species. The method of UV-spectroscopy was used to quantify the extracted sum of chlorophylls. Pure acetone was used as a solvent. As a result it was established that using of the Camelina oil is the most perspective for the chlorophylls extraction from the dried L. saccharina.
Keywords: Laminaria saccharina, chlorophyll, vegetable oil, maceration
Algae and consequently their extracts is the treasure trove of biologically active compounds. Their beneficial properties for humans, animals and plants were recognized in the past and are appreciated nowadays, in the development of new biotechnological products. Products with functional properties containing organic compounds derived from natural sources, rather than being a product of heavy organic synthesis are increasingly demanded by consumers.
Seaweeds contain many different biologically active compounds, which chemical structure relates to the corresponding taxonomic classification of algae and their cell structure [1].
Three main types of seaweed are distinguished: brown, red and green algae. Laminariales, which belong to brown algae, are the most abundant macro-algae of White Sea. The brown colour of these algae results from the dominance of the xanthophyll pigment fucoxanthin, which masks other pigments – chlorophylls a and c because of which the brown algae differ from land plants and many other types of algae which contain clorophylls a and b. More than 50% of chlorophylls a and c sum, and 80% of fucoxanthin present in thylakoids of brown algae [2].
The pigments of Laminariales have various pharmacological activities. For example, fucoxanthin has anti-cancer activity [3]. It is known also that fucoxanthin has antioxidant, anti-inflammatory, anti-obese, antidiabetic, antiangiogenic and antimalarial activities. It has protective effects on the liver, blood vessels of the brain, bones, skin, and eyes. In addition, many studies have suggested that a combination of fucoxanthin and edible oil or lipid increases the absorption rate of fucoxanthin, and thus it might be a promising marine drug [4]. Сhlorophyll has anti-inflammatory [5], antibacterial, antioxidant and anticancer activity [6].
Chlorophylls, as well as carotenoids, are lipid soluble compounds of L. saccharina chemical composition so it is fully logical to use organic solvents for their extraction from plant raw material. It is known about usage of acetone, ethanol, ethylacetate [7], dimethylsulphoxide [8], dimethylformamide, methanol [9] and so on as solvents.
It is known that extraction of pigments from brown algae, particularly the larger macrophytes, can be difficult because of the rubbery nature of the thalli, and the large amount of polysaccharides in the tissue. Also it is known that among several extraction methods, maceration is the most traditional and the simplest extraction procedure. Some drawbacks of this method include long extraction time and low mass transfer, but this method excludes presence of organic solvents and their rests in oil pigments extract. This fact is important if this extract is planned to use for preparing different medicines for using in children, in people in ages and in people with different allergic reactions.
Moreover once extracted, the chlorophyll pigments are very unstable and easily oxidized. Purified chlorophylls are especially unstable in the presence of dilute acids and in light. Under even slightly acidic conditions the magnesium ion at the centre of the porphyrin ring is readily lost. This process is accompanied by characteristic, distinct and readily visible color change.
In order to 'regenerate' the bright blue-green color of chlorophyll, usually a copper salt is added to chlorophyll extract. The resulting blue-green copper chlorophyllins (as they are commonly but confusingly called) are relatively resistant to both light and acidic conditions.
In this case a preferable alternative is to prepare an oil-soluble plant extract in such a controlled manner that the original (magnesium) chlorophyll is retained, providing that a method can be found to preserve its original structure and therefore its original color during its subsequent processing, storage and use.
Such a carefully made extract will have the additional benefit of helping to preserve other labile components such as carotenoids, tocopherols, etc [10].
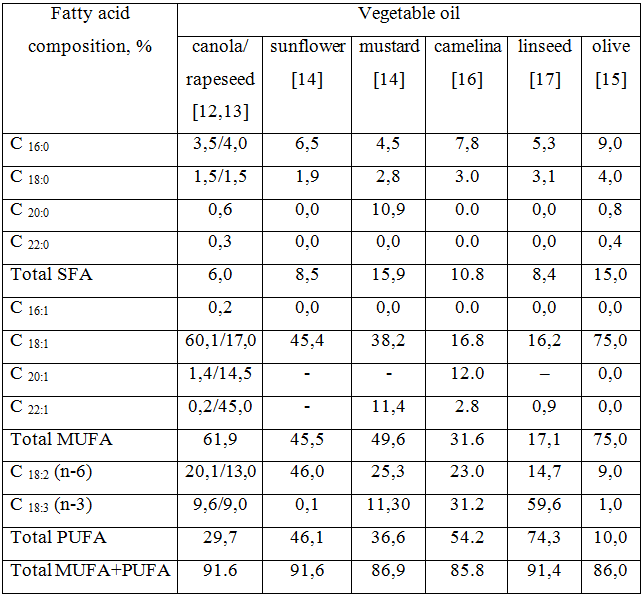
Edible oil includes different fatty acids, which are classified as saturated (SFA), monounsaturated (MUFA) and poly-unsaturated (PUFA) fatty acids. A high intake of saturated fatty acids may lead to hypercholesterolaemia. On the contrary, polyunsaturated fatty acids have a hypocholesterolaemic effect in human. As a result of deficiency of essential fatty acids such as linoleic, linolenic and arachidonic acid growth retards and dermal symptoms appear.
After comparison of constituents of major fatty acids in base oils commonly used for maceration (Tab. 1) we have chosen linseed, sunflower and camelina oils for extraction of pigments because of the sum of SFA, MUFA+PUFA and maximum level of erucic acid which must not exceed 5% of the total level of fatty acids in the fat component [11]. As chlorophyll is a thermolabile constituent, we have used cold maceration as extraction method.
The examples of wild L. saccharina were collected in aquatoria of White Sea near Solovetsky Islands shores between July 2014 and August 2014. Plant size and age of collected species were not determined. The harvested algae was washed from salt, dried in the open air in the shade and then stored in a dry, cool place, protected from light. Loss on drying of dry algae was counted after oven-drying at 105 °C. Plant material was powdered, passed through sieves with 1 mm size of the holes. To 10.000 g of powdered plant material placed into light protected plastic containers with plugs, 100.0 ml of each kind of oils was added and it was let stand in dark place at room temperature, shaken periodically for 10 days.
Table 1 – the comparison of major fatty acids in some vegetable oils (w/w%)
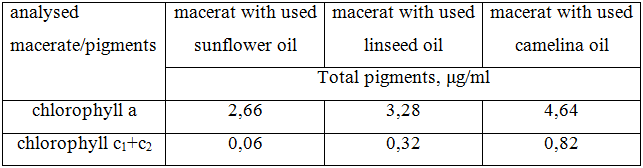
After that liquid was strained off, solid residue (mark) was pressed filtered using cheese cloth. The obtained filtrate was centrifuged at 5000 rpm for 10 minutes. The supernatant was separated and absorbance was read at 350 – 750 nm on UV spectrophotometer. At the same time the UV-specters of respective oil in the same conditions were obtained. The amount of pigments, with account of chlorophyll a, presented in respective sample of vegetable oil, was calculated according to Jeffrey and Humphrey formula [18]. The obtained results are presented in tabl. 2.
Table 2 – the results of quantification of pigments, presented in analyzed macerates
* half value of 6 quantification results
Thereby, in the course of carried investigations it was established that Camelina oil usage is the most perspective for L. saccharina macerate obtainment as the highest content of algae chlorophylls was identified in it.
References
- Chojnacka, K. Biologically active compounds in seaweed extracts - the prospects for the application / K. Chojnacka, A. Saeid, Z. Witkowska, T. Łukasz // The Open Conf. Proce. J. – Vol. 3. – p. 20–28.
- Douady, D. Fucoxanthin-chlorophyll a/c light-harvesting complexes of Laminaria saccharina: partial amino acid sequences and arrangement in thylakoid membranes / D. Douady, B. Rousseau, L. Caron // – 1994. – Vol. 33. – N 11. – p. 3165–3170.
- Chung, T.W. Marine algal fucoxanthin inhibits the metastatic potential of cancer cells / T. W. Chung, H. J. Choi, S. Jeong, C. H. Kim and all // Biochem. Biophys. Res. Commun. – 2013. – Vol. 439. – N 4. – p. 580–585.
- Peng, J. Fucoxanthin, a marine carotenoid present in brown seaweeds and diatoms: metabolism and bioactivities relevant to human health / J. Peng, J-P. Yuan, C-F. Wu, J-H. Wang // Drugs. – 2011, N. 9. – p. 1806–1828.
- Islam, M. N. Anti-inflammatory activity of edible brown alga Saccharina japonica and its constituents pheophorbide a and pheophytin a in LPS-stimulated RAW 264.7 macrophage cells / M. N. Islam, I. J. Ishita, S. E. Jin, R. J. Choi and all // Food Chem Toxicol. – 2013. – Vol. 55. – p. 541–548.
- Naimushinaa, L. V. Spectrophotometric study of chlorophyll and its derivatives accumulation in Melissa officinalis extracts using two-phase solvent system / L. V. Naimushinaa, A. Y. Karasevaa, N. V.Chesnokov // of Siber. Federal Univ. Chem.– 2012. – Vol. 5. – N 3. – p. 281–288.
- Kumar, P. Solvent extraction and spectrophotometric determination of pigments of some algal species from the shore of Puthumadam, southeast coast of India / P. Kumar, C. M. Ramakritinan, A. K. Kumaraguru // Jour. of Oceans and Oceanography. – 2010. – Vol. 4. – N. 1. – p. 29–34.
- Pompelli, M. F. Spectrophotometric determinations of chloroplastidic pigments in acetone, ethanol and dimethylsulphoxide / F. Pompelli, S. C. França1, R. C. Tigre, M. Teixeira de Oliveira and all // Braz. Journ. of Biosci. – 2013. – Vol. 11. – N 1. – p. 52–58.
- Wiltshire, K. H. Extraction of pigments and fatty acids from the green alga Scenedesmus obliquus (chlorophyceae) / K. H. Wiltshire, M. Boersma, A. Möller. H. Buhtz // Aquatic Ecology. – 2000. – Vol. 34. – p. 119–126.
- Kalinowski, P. M. Stabilisation of chlorophyll using medium chain triglyceride oils / Р. М. Kalinowski // Pat. WO/2003/002133 EP. A61K 47/44, A61K 47/14. – № 1 ; prior. date 29.06.01 ; publ. date 21.01.06.
- Relating to the fixing of the maximum level of erucic acid in oils and fats intended as such for human consumption and in foodstuffs containing added oils or fats // Council directive (76/621/EEC). – 1976. – 35 p.
- Canola oil. Physical and chemical properties. – http://www.canolacouncil.org/media/515239/canola_oil_physical_chemical_properties_1.pdf.
- Sahasrabudhe, M. R. Crismer values and erucic acid contents of rapeseed oils / M. R. Sahasrabudhe // J. Amer. Oil Chem. Soc. – 1977. – Vol. 54. – N 8. – p. 323–324.
- Chowdhury, K. Studies on the Fatty Acid Composition of Edible Oil / K. Chowdhury, L. A. Banu, S. Khan, A. Latif // Bangladesh J. Sci. Ind. Res. – – Vol. 42. – N 3. – p. 311–316.
- Zambiazi, R. C. Fatty acid composition of vegetable oils and fats / C. Zambiazi, R. Przybylski, M. W. Zambiazi, C. B. Mendonça // B.Ceppa. – 2007. – Vol. 25. – N. 1. – p. 111-120.
- Camelina oil. – http://en.wikipedia.org/wiki/Camelina_sativa.
- Linseed oil. – http://en.wikipedia.org/wiki/Linseed_oil.
- Jeffrey, S. W. New Spectrophotometric Equations for Determining Chlorophylls a, b, c1 and c2 in Higher Plants, Algae, and Natural Phytoplankton / W. Jeffrey, G. F. Humphrey // Biochem. und physiol. der pflan. – 1975. – Vol. 167. – p. 191-194.