РЕНТГЕНОСПЕКТРАЛЬНАЯ НАНОДИАГНОСТИКА
Мулла Али Махер Н.
Магистр физики нанотехнологий
Дияла / Ирак
РЕНТГЕНОСПЕКТРАЛЬНАЯ НАНОДИАГНОСТИКА
Аннотация
Рентгеновское излучение представляет собой электромагнитное излучение. Абсорбционной спектроскопии рентгеновских лучей является методом характеристики и используется для определения структурной информации и / или электронной структуры соединения. Поглощения рентгеновских лучей вблизи края структуры (XANES) является типом абсорбционной спектроскопии, что указывает на особенности в спектрах поглощения рентгеновского (XAS) конденсированного вещества. Расширенная рентгеновских спектров поглощения тонкая структура отображается в виде графиков коэффициента поглощения данного материала против энергии. Ключевые слова: XAS, XANES, EXAFS.Mula ALI M. N.
Master Physics Nanotechnology
Governorate Diyala / Iraq
X-RAY SPECTRAL NANODIAGNOSTIC
Abstract
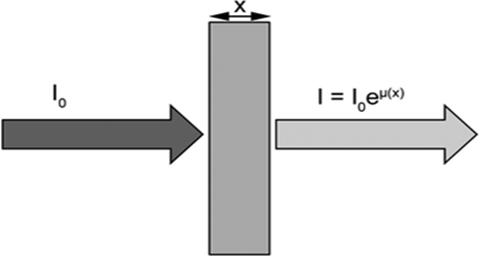
X-radiation (X-rays) is an electromagnetic radiation. X-ray absorption spectroscopy is a characterization technique used to determine structural information and/or electronic structure of a compound. X-ray absorption near edge structure (XANES) is a type of absorption spectroscopy that indicates the features in the X-ray absorption spectra (XAS) of condensed matter. Extended X-Ray Absorption Fine Structure spectra are displayed as graphs of the absorption coefficient of a given material versus energy. Keywords: XAS, XANES, EXAFS. Introduction X-radiation (X-rays) is an electromagnetic radiation having much higher energy and much shorter wavelengths than ultraviolet light. X-rays have very small wavelengths, in the range of 0.01 and 10 nanometers, so small that some x-rays are no bigger than a single atom of many elements, X-rays have frequencies in the range (3×1016 Hz to 3×1019 Hz) and energies in the range 100 eV to 100 KeV. X-rays were first observed and documented in 1895 by German scientist Wilhelm Conrad Roentgen, so X-radiation is referred to with terms meaning Rontgen radiation. X-ray absorption spectroscopy (XAS) is a characterization technique used to determine structural information and/or electronic structure of a compound. When the x-rays incident and hit a sample in Figure 1 , the oscillating electric field of the electromagnetic radiation interacts with the electrons bound in an atom. Either the radiation will be scattered by these electrons, or absorbed and excite the electrons[1,2]. A narrow parallel monochromatic x-ray beam of intensity ( I0) passing through a sample of thickness (x) will get a reduced intensity I according to the expression:ln (I0 /I) =µ . x (1)
Fig. 1– X-ray beam intensity ( I0) passing through a sample of thickness (x)
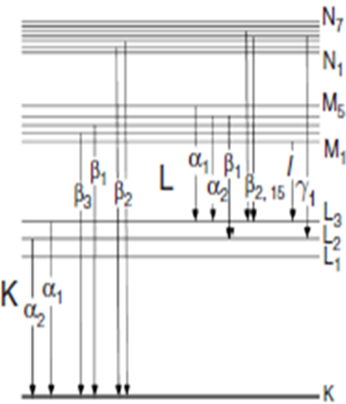
X-ray absorption spectroscopy (XAS) data are obtained by tuning the photon energy using a crystalline monochromator to a range where core electrons can be excited (0.1-100 keV photon energy). The edges depends upon the core electron which is excited. The principal quantum numbers n=1, 2, and 3, correspond to the K-, L-, and M-edges, respectively. For instance, excitation of a 1s electron occurs at the K-edge, while excitation of a 2s or 2p electron occurs at an L-edge.Fig. 2 –Transitions of XAS edges
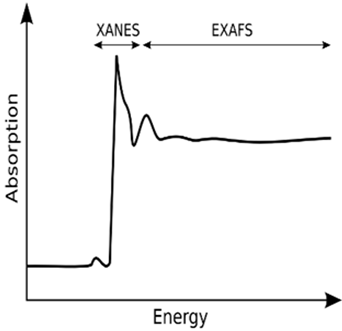
In X-ray spectroscopy, transitions are involved in absorption (XAS, X-ray absorption spectroscopy) or emission (XES, X-ray emission spectroscopy) of X-rays, where the former probes the ground state to the excited state transitions, while the latter probes the decay process from the excited state. Both methods characterize the chemical nature and environment of atoms in molecules . X-ray absorption spectroscopy (XAS) is the measurement of transitions from core electronic states of the metal to the excited electronic states (LUMO) and the continuum; the former is known as X-ray absorption near-edge structure (XANES), and the latter as extended X-ray absorption fine structure (EXAFS) which studies the fine structure in the absorption at energies greater than the threshold for electron release. These two methods give complementary structural information in Figure 3, the XANES spectra reporting electronic structure and symmetry of the metal site, and the EXAFS reporting numbers, types, and distances to ligands and neighbouring atoms from the absorption elements. X-ray absorption near edge structure (XANES) is a type of absorption spectroscopy that indicates the features in the X-ray absorption spectra (XAS) of condensed matter due to the photo absorption cross section for electronic transitions from an atomic core level to final states in the energy region of 50–100 eV above the selected atomic core level ionization energy, where the wavelength of the photoelectron is larger than the interatomic distance between the absorbing atom and its first neighbour atoms. The fundamental processes which contribute to XANES spectra.Fig. 3 – XANES & EXAFS Spectra
Extended X-Ray Absorption Fine Structure (EXAFS) spectra are displayed as graphs of the absorption coefficient of a given material versus energy, typically in a 500 – 1000eV [3] range beginning before an absorption edge of an element in the sample. In the EXAFS region, the kinetic energy of the electron is large enough that small variations in the interstitial potential do not make much difference, and the muffin-tin approximation works well. Single scattering The EXAFS region is typically taken as starting 20–30 eV [2,3] above the edge jump. At these energies, the X-ray excited photoelectron has significant energy, and thus has a de Broglie wavelength that is comparable to the interatomic distances. Multiple scattering Multiple scattering is particularly important at low k where the photoelectron has a very low energy and consequently a long mean-free path, allowing it to undergo extensive multiple scattering. Multiple scattering is particularly strong if the two scattering atoms are nearly collinear since the photoelectron is strongly scattered in the forward direction. Programs for Calculating and Analysing EXAFS The analysis of EXAFS data can be divided into two stages: reduction of the measured absorption spectra to EXAFS (i.e., application of EXAFS Equation and analysis of the x(k) data to obtain structural parameters. In order to fit EXAFS data, it is first necessary to determine the parameters that define the scattering. This can be done using ab initio calculations or from model compounds of known structure. In recent years, the available theoretical methods for quickly and accurately calculating these parameters have improved dramatically. Ab initio calculations are now relatively straight forward, with main program that is in wide use: FEFF . Although these differ in the particulars of their approach to EXAFS, all give approximately the same structural parameters. Conclusion XANES analysis based on linear combinations of known spectra from “model compounds” is sufficient to tell ratios of valence states and/or phases. More sophisticated linear algebra techniques such as Principle Component Analysis and Factor Analysis can (and are) also be applied to XANES spectra. XANES is considerably harder to fully interpret than EXAFS. Precise and accurate calculations of all spectral features are still difficult, time-consuming, and not always reliable. This situation is improving, but at this point, quantitative analyses of XANES using ab initio calculations are very rare. Still, such calculations can help explain which bonding orbitals and/or structural characteristics give rise to certain spectral features.References
- Rehr, J. J. Theoretical approaches to x-ray absorption fine structure / J. J. Rehr, R. C. Albers // Reviews of Modern Physics - July 2000.- Vol. 72.- No. 3. - P.261.
- Penner -Hahn, J. E. X-ray Absorption Spectroscopy . eLS. / J. E. PENNER-HAHN. - Michigan, USA: John Wiley & Sons, Ltd. , 2005.
- Bunker, G. INTRODUCTION TO XAFS / G. Bunker.- Cambridge CB2 8RU, UK: Cambridge University Press – 2010. – 268 p.