LASER-INDUCED PROCESS OF DEFROSTING IN BIOLOGICAL TISSUES AFTER IMPREGNATION BY NANOPARTICLES WITH ABNORMAL HIGH PHOTOTHERMAL EFFECT
Гуляев П.Ю.1, Омельченко А.И.2
1ORCID: 0000-0002-4337-1253, Доктор технических наук, Профессор, Югорский государственный университет, Ханты-Мансийск, 2ORCID: 0000-0001-8546-8187, Кандидат физико-математических наук, ФНИЦ "Кристаллография и Фотоника" РАН, Москва
ЛАЗЕРНО-ИНДУЦИРОВАННЫЙ ПРОЦЕСС ОТТАИВАНИЯ БИОЛОГИЧЕСКИХ ТКАНЕЙ ПОСЛЕ ИМПРЕГНИРОВАНИЯ НАНОЧАСТИЦАМИ С АНОМАЛЬНО ВЫСОКИМ ФОТОТЕРМИЧЕСКИМ ЭФФЕКТОМ
Аннотация
Фототермический эффект лазерного излучения на замороженных биотканей имеет важное значение для целого ряда современных технологий. Например, ткань криоконсервации имеет потенциал для криохирургии и других видов медицинской обработки с использованием замораживания и лазерного нагрева. В настоящем исследовании рассмотрено применение биофункциональные наночастиц с высоким фототермическим эффектом, полученных методом самораспространяющегося высокотемпературного синтеза (СВС), для лазерного нагрева замороженных биотканей. Стабилизированные крахмалом водные растворы наночастиц KxMoO3 и HxMoO3 демонстрирующие высокий уровень поглощения лазерного излучения с длиной волны 1,56 мкм были применены для импрегнирования свиной кожи. Лазерное нагревание замороженног уха свиньи о модифицированного наночастицами оксидных бронз позволяет контролировать температуру облучаемой ткани до +1 ° С. Пропитка подкожной ткани наночастицами с высоким фототермическим эффектом может быть использована для процедуры лазерно-индуцированного оттаивания замороженной ткани уха.
Ключевые слова: Самораспространяющийся высокотемпературный синтез, бифункциональные наночастицы, замороженные биологические ткани, лазерно-индуцированный процесс оттаивания.
Gulyaev P.Yu.1, Omelchenko A.I.2
1ORCID: 0000-0002-4337-1253, PhD in Engineering, Professor, Ugra State University, Khanty-Mansiysk, 2ORCID: 0000-0001-8546-8187, PhD in Physics and Mathematics, Federal Research Center "Crystallography and Photonics", RAS, Moscow
LASER-INDUCED PROCESS OF DEFROSTING IN BIOLOGICAL TISSUES AFTER IMPREGNATION BY NANOPARTICLES WITH ABNORMAL HIGH PHOTOTHERMAL EFFECT
Abstract
Photothermal effect of laser radiation on frozen biotissues is essential for a number of modern technologies. For example, tissue cryopreservation has potentials for cryosurgery and other types of medical treatment using tissue cooling, frizzing and laser heating. In the present study the self-propagating high temperature (SHT) synthesis and application of biofunctional nanoparticles (NPs) using high photo-thermal effect for laser heating of frozen biotissues are considered. Starch stabilized aqueous solutions of KxMoO3 and HxMoO3 NPs demonstrate high absorption of laser radiation with the wavelengths of 1.56 μm being applied to pig skin containing small amounts of NPs. For hydrogen-molybdenum oxide bronze the thermal effect on pig skin is higher at 1.44 μm than at 1.56 μm. Laser heating of frozen pig’s ear previously modified by bronze NPs injection allows controlling temperature of irradiated tissue up to +1 oC. Subcutaneous tissue impregnation with NPs of the metallic oxide bronzes can be used for careful laser treatment of frozen ear tissue.
Keywords: Self-propagating high-temperature synthesis, bifunctional nanoparticles, frozen biological tissues, laser-induced process of thawing. IntroductionFor numerous clinical purposes it is important to control the photothermal effect of near-IR laser irradiation of cartilage[1]. While the most of the researches are focused on fabrication of the NPs that possess photothermal effect in visible spectral range (for example, Au and Ag), the information of NPs that absorb in near IR is still limited. It is worth noting that UV-absorbance of metals has plasmon nature and is defined by free electrons weakly bonded to the metal lattice. Most of the metals absorb EM radiation in UV-range. Due to the quantum-sized effect the bathochromic shift of the plasmon resonance occurs for Au and Ag NPs and the resonance in Ag and Au NPs colloids is observed in visible spectral range. Noble metals do not absorb the near-IR radiation – thus, their photothermal effect in near-IR is poor [2].
Conversely, the absorbance of the most metal oxides and metal bronze NPs is defined by electrons bonded to the lattice. These electrons cannot abandon the allowed bands. Some oxides, for example, Fe3O4 (magnetite) and metal oxide bronzes: red (K0.33MoO3), blue (K0.3MoO3), are the narrow band semiconductors, which are close to conductors [3,18]. Spectral properties of these substances are quite similar. The considered oxides and oxide bronzes absorb radiation in near-IR range [2,9,18,19].
Transition metal oxides are of great interest for many applications due to the special chemical and optical properties. Such kind of materials is widely used in quantum electronic devices [19]. Metallic oxide bronzes AхO·BОy, where A=К, Н, Na, B=Ti, Mo, W, 0<x<2, y=2, 3 present unusual electrochemical [15], photothermal [7,11], and catalytic [2] properties. For medical purposes, the bronze NPs can be used as absorbers of EM radiation in thermal therapy, contrast agents in diagnostics, and the carriers in targeted drag delivery, as well. High absorption of the bronze NPs in the near IR spectral region [11] makes them perspective in laser diagnostics and wound healing of a damaged articular cartilage [1] and cryosurgical procedure where the controllable defrost of skin and subcutaneous tissue is an urgent problem [7].
Laser irradiation of frozen biological tissue results in gradual thawing of crystal ice [5]. But the process can be hardly controlled because of the high damage degree of the re-crystallization [7,25]. In the work [26], authors used theoretical approach for numerical study of the thawing process in biological tissue induced by laser radiation. Recently [22] showed that laser radiation of the iron oxide starch stabilized NPs embedded into the dense cartilaginous tissue does not cause additional structural alterations therein.
It is well known that new phase formation initiates near impurities, e.g. bubbles, small particles and others [16]. Thus, NPs impregnation into hydrated biological tissue should modify crystallization of interstitial water during its laser thawing. Although the fundamental problem of pure crystal ice thawing has been solved by Josef Stefan in 1889 [24], the controllable thawing of ice in biotissues is still an urgent problem. To demonstrate effectiveness for laser thawing of frozen biological tissue impregnated with NPs and safety of its laser heating we developed the new absorptive adds on the base of the metallic oxide bronzes.
The work aims at the synthesis of metallic oxide bronze NPs using SHT technique and studying the influence of high photothermal IR laser effect of these NPs on the frozen biotissue thawing.
Materials and methods
The reagents for metallic oxide bronzes synthesis were selected from commercially available powders of metal oxides: TiO2, MoO3, and WO3 (mean particles size of 100 mm, CP purity 99%, ReaChem, Russia). Transition metal oxide bronzes were obtained by three different methods: (1) SHT-synthesis, (2) mechanical and (3) wet chemical synthesis.
SHT-synthesis
The synthesis was performed according with the previously developed technique [6,8]. Stoichiometric proportion x/y=(0-2;)/(2-3;) of the chemically pure powders of Cu(OH)2, TiO2 (MoO3, WO3), NaI (KI) were mixed in agate mortar, pressed with ethanol into tablets of 5-10 mm in diameter and put into reactor. Then the mixture was burned in air flow using an incandescent wire. Combustion products have shown itself grayish crystals with metallic glint. Reaction time was approximately 2-5 s. During chemical reaction iodine vapor has been observed. After cooling the particles of the reduced metallic cooper were observed. Reaction yield was 75-90 %. The products were separated from metallic copper by multiple washing of the crystals in a 30% HNO3 aqueous solution. After drying in air, the samples were milling in a planet-type ball mill AGO-3.
The time and the temperature of the diffusive processes in the course of the reaction were controlled by high-speed brightness and spectral micropyrometry as described elsewhere [8,12].
Mechanical synthesis
Blending agents containing the stoichiometric proportion of TiO2 (MoO3, WO3), NaI (KI) were milling in AGO-3 ball mill during preset time of 5-50 s with the tungsten balls as grinding bodies.
Chemical (wet) synthesis
Surplus of a 2,5 M solution of HCl was added to the oxide powders of chemically pure WO3 (MoO3). Grains in amount of 2.5 g of metallic Zn were added therein. After the week, the blue-grayish powder was obtained, after the second week – the blue and deep blue colored products. Over the next weeks the violet and brown colored products were obtained (Fig.1).
Color palette (Fig.1a) corresponds to the colors of the NPs precipitates obtained in chemical synthesis. The precipitates were captured with high resolution and the pictures were used to obtain the color palette:
WO3 →H0,1WO3 →H0,1WO3 →H0,23WO3 →H0,33WO3 →H0,5WO3
Yellowish →blue-grayish→blue →deep-blue → violet →brown.

Fig.1(a,b) - 1a: Color of the Hydrogen Tungstain bronzes variation in dependence on hydrogen stoichiometry; 1b: Images of the nanoparticles solutions in dependency on hydrogen stoichiometry
In case of MoO3, a deep-blue colored crystals of HxMoO3, where 0,1< х < 0,36, were obtained [15].
Crystallinity
Crystal structure of the products of was examined previously [15] using X-ray diffraction analysis. X-ray diffraction analysis of hydrogen molybdenum bronze crystal has shown the octahedral structure with a layered ordering, which is supported by hydrogen bonds between oxide layers. Crystal structure of the tungsten bronzes is similar to that is of hydrogen molybdenum ones [11,15]. The size, morphology or structure of the particles is the key factors for the absorption of laser radiation. Characterization of the hydrogen molybdenum bronzes has been made in our papers [11, 15, 21], where NPs size distribution, optical uv-vis absorption spectra, morphology or structure of these particles were studied. The NPs of hydrogen molybdenum oxide bronzes possess the needle-like shape and they have the 9 nm-maximum in size distribution.These nanoparticles have shown [9,11] an abnormal high photothermal effect of the radiation with an Erbium fiber laser (λ=1.56 mm).
Adsorption measurements
NPs of tungsten and molybdenum bronzes were separated by sedimentation and centrifugation and then placed into the axial symmetric electromagnetic trap [20, 21] for separation of the electro-sensitive particles to obtain small neutral NPs. These NPs were used for preparation stabile colloids based on starch molecules aggregated with them [21]. Before colloid stabilization, size distribution of NPs was controlled using CPS 2400 analytical disc centrifuge. The measurements showed a narrow sized distribution of molybdenum oxide bronze NPs [11] with the maximum size about ~10 nm for the NPs, which were selected from center of the trap. These NPs were suspended with a 1% starch aqueous solution for stabilization of the colloid and functionalization of NPs for biointerface enhancement. To enhance NPs interaction with starch all the parent aqueous solutions containing particles were diluted with a 10 % ascorbic acid aqueous solution. Then, the colloids of NPs in aqueous solution were exposed to CW radiation of an Er-fiber laser (LS 2 IRE-Polus, Russia) at the wavelength (λ) of 1.56 mm during 0.5-1 min. Exposed dose was 50-100 J. Laser radiation were delivered into the solution by a 400 mm silica fiber with spherical lens at the tip. It allowed irradiation of the colloid in fragmentation mode [14]. All of the irradiated solutions were examined on transparency by spectrophotometer PE5400B (Ekros, Russia). Control of the NPs size (d) in aqueous solution was carried out using the wavelength dependency of visible light scattering (Omelchenko 2011), (Gulyaev 2012) corresponded to the Rayleigh’s criteria d << λ/20. Reduced absorption coefficients (attenuation constant) of the NPs in aqueous solutions of the different concentrations were measured at the two wavelengths: near-IR lasers emitted low intensive radiation at 1.45 mm (Diode-based laser “Lahta-Milon”, Model 1450-2.3 Quiltek manufacturing, Russia) and 1.56 mm (Er-fiber laser LS 2, IRE-Polus, Russia). Monochromatic absorption coefficient of the solutions at these wavelengths was measured using photometric technique. Measurement of attenuation of probe laser radiation with intensity I0 in the solution of a fixed optical length (l) allows obtaining value of absorption coefficient α:
α=(1/l)·ln( I0/ Ix),where Ix – intensity of transmitted light.
Laser radiation power was measured by power meter Fieldmaster (Coherent, USA). Spatial distribution of laser radiation was recorded by laser beam profile imager based on CCD (VGA 640x480) with the visualization of IR-radiation by an up-converting fluorescent film (Polyronik, Russia). Laser beam size (r0) was measured using a gauss approximation of intensity distribution I(r)=I0 exp(–r2/r02).
Tissue impregnation testing
NPs of oxide bronzes in starch stabilized aqueous solutions were used for biotissues impregnation to change their optical properties. Fresh pig’s ear tissues were used in our experiments for tissue modification by NPs. For this purpose we used tattoo making up to the pig’s ear skin. A scarification machine filled with pigment based on the synthesized NPs has been used to impregnate ear tissue in shape of the word “ALT’15”. The diameter of the tattoo was 0.3-0.5 mm on full depth of the ear skin. Adhesion of NPs of the oxide bronzes to pig’s ear tissue was evaluated by washing of tattoo with a 0.1% sodium dodecyl sulfate aqueous solution.
Then the tissue samples of pig’s ear were placed in freezer for 2-3 hours. After that they were taken from freezer and kept at an ambient temperature until their temperature reached -2 - 0 0C. Then the samples were irradiated by laser beam with Gaussian intensity distribution of radiation. Various regimes of laser radiation of 1.56 and 1.44 μm wavelengths were examined: power of laser radiation of 5 W and 2 W in repetitive pulse regime (pulse duration 400 ms, repetition rate 0.7 Hz) were used for laser heating of the frozen tissue samples. Temperature of the skin and subcutaneous tissues was controlled by Testo 875 thermal vision system.
Results and Discussion
SNT-synthesis
At the combustion of reacting mixture, we observed a rapid propagating of high temperature along the tablet after its ignition (Fig.2).
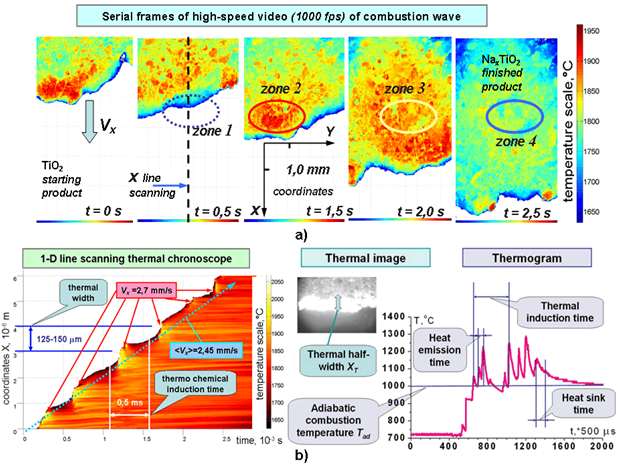
Fig.2 - SHT-synthesis of metal oxides: a) serial frames of records of the solid state TiO2 burning process; b) thermogram of the process of solid state burning
Fig.2a demonstrates serial frames of records of the solid state TiO2 burning process. The basic stages of the process can be observed (Fig.2b). The warming up of the lowest cold layer is presented in zone 1. Here we do not observe any chemical reactions. In zone 2 the rapid ignition and exothermic combustion reaction as local thermal explosion take place [10,13]. The effective thermal width XT varies from 0.15 to 1 mm. However the necessary structural and phase changes of the crystal lattice still do not have time to occur. Zone 3 presents the disintegration process of high-temperature site to the small ones due to internal heat outflow. The new sites form the extensive “thermal cloud” where the temperature approaches the adiabatic value and promotes the intercalation of Na atoms into the octahedral lattice of TiO2 crystals. The next stage is presented in zone 4. The endothermic process of the bronze NaxTiO2 formation decrease the temperature of the final product and the needed stoichiometric proportion established. The micropyrometry measurements are presented in Fig.2b as the 1-D scanning thermal chronoscope along the dashed line of the heat monitoring photo-matrix. The thermal emission time in zone 2, the time of heat induction in zone 3 and the constant of the heat outflow time in zone 4 can be measured. The method of technological control of the values is the preliminary mechanical activation and grinding of the initial metal oxide powders in the mill under the controllable energy stress loading of the mill, time of activation and the specific surface of the powder [6]. Thus, the high reproducibility of the synthesized metal oxide bronzes can be achieved.
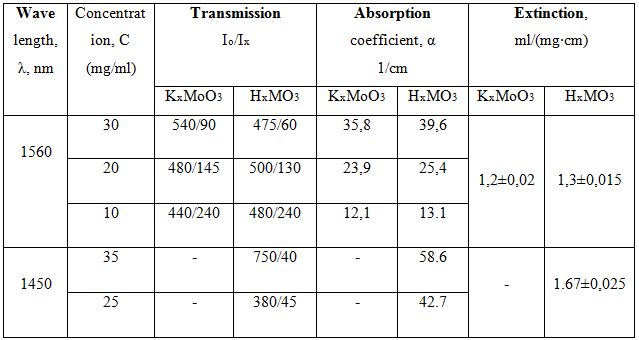
Optical measurementsThe absorption coefficient measurements of the metal oxide bronze colloids have shown that absorption of laser radiation increases with the growth of NPs concentration in aqueous solution. The solutions of molybdenum oxide bronzes at the concentration of 30 mg/ml demonstrate a high value of absorption coefficient of approx. in four times higher than water absorption (Table 1). The monochromatic absorption coefficient of the colloids at the wavelength of 1.45mm was higher than that coefficient measured at 1.56 μm. Measurement of absorption coefficient of the colloids based on the titanium oxide and tungsten oxide bronzes has shown the lower values of absorption than for the molybdenum. Also, absorption coefficient of the hydrogen molybdenum oxide bronze colloid was two times higher than aqueous solution of iron oxide NPs of the same concentration [15]. Therefore, further experiments with biotissues and NPs were carried out with molybdenum oxide bronze, which demonstrates higher photo-thermal effect.
Table 1 - Reduced absorption coefficient (attenuation constant) of Potassium and Hydrogen Molybdenum oxide bronze in aqueous solutions of the different concentrations at the wavelengths of a near-IR laser
Ex-vivo testing of the molybdenum oxide bronzes
Starch stabilized aqueous solutions of the KxMoO3 and HxMoO3 NPs have demonstrated high absorption of laser radiation at the wavelengths of 1.44 μm and 1.56 μm. NPs of molybdenum oxide bronzes also have shown high adsorption in the pig’s skin after tattoo making, moreover adsorption to the tissue of the HxMoO3 NPs was higher than other type of the used bronzes. It was proved by washing tests.
Photograph of pig’s ear with tattoo is shown in Fig.3a. Fig.3b shows setup for laser irradiation of the samples of ear tissue. Ahead of the silent rod supported the fiber and the sample, there is Er- fiber laser used for sample irradiation. Thermal vision system is shown behind.
Thermograms obtained with Testo 875 thermal vision system are shown in Fig.3c and Fig.3d.
Fig.3c presents IR- thermograms of non-radiated pig’s ear frozen to -2-00C. The word ALT’15 is hardly distinguishable. As it can be seen from IR- thermograms (Fig.3d), laser irradiation of tattoo modified skin of pig’s ear demonstrate a high photo-thermal effect of pulse periodic laser radiation with power of 2W. The word made up with tattoo is well distinguishable. Besides, the hot spot due to thermal effect of Gaussian laser beam on pig’s ear skin can be observed. Measurements of the distribution of intensity of laser radiation in the limit of this hot spot has shown that it’s satisfy to Gaussian I(r)=I0 exp(–r2/r02), where r0 (beam radius) is 2 cm.
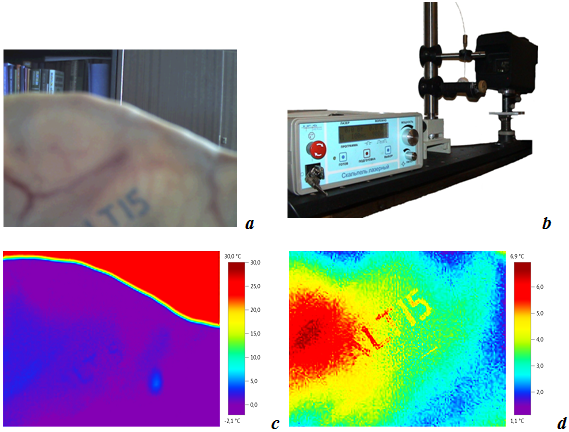
Fig.3 - Measurements of the temperature distributions at surface of frozen tissue: a-View of pig’s ear in visible light (fragment of Tattoo bottom); b- setup for IR temperature measurements; c-Thermogram of skin of the frozen pig’s ear (non-radiated); d- Thermogram of the tissue irradiated by laser beam with intensity distribution: I(r)=I0 exp(–r2/r02) in pulse periodic regime: power of 2W, pulse duration 400 ms, repetition rate 0.7 Hz
Laser radiation with this intensity distribution should generate heat produced stationary temperature distribution along radial coordinate at surface of irradiated tissue (Carslaw, Jaeger, 1993):
T(r)=√π/2∙ Io r0/ κ ∙exp(–r2/r02)∙ Jo(–r2/r02),
where Io –intensity of laser radiation in center of beam. Jo(–r2/r02)- Bessel’s function, r0-beam radius, r- radial coordinate.Thermal conductivity of frozen tissue (κ) contains two summands: κic - ice thermal conductivity and κht -convective part of thermal conductivity that corresponds to the heat transition in the tissue. Using the presented equation, we determined optimal intensity of laser radiation which is needed to heat frozen pig’s ear up to +10C. In dependency of initial temperature of frozen tissue, it amounted from: Iopt = 0.05-0.1 W/cm2. Estimation of thermal conductivity of frozen tissue was made for laser heating of semi-infinite porous medium accounting for heat-mass transfer in ice of the tissue at laser-induced phase transition. It was experimentally revealed that κ is higher than κic. Details for calculation of thermal conductivity of frozen tissue are presented in [23]. Calculation of heat transition in frozen tissue according to reference [24, 25] gives κht~10 W/(moC). It shows that convective part κht of heat transition in the tissue is significant.
Results of the measurements of the temperature field’s distribution at the surface of frozen pig’s ear tissue with a hydrogen molybdenum oxide bronze NPs have shown that it is dependent on the NPs absorption, the wavelength and intensity of laser radiation, and the temporal regimes of laser irradiation of tissue, as well. It has been shown that pulse periodic regime of irradiation of the frozen biotissue allows localizing temperature field near tattoo picture drawn by NPs.
It is known [26] that laser radiation of ice changes the thawing process. This could be used not only for cell’s cryopreservation, but it may be successfully used in the different application of the laser treatment of frozen biotissue modified by NPs with high photothermal effect (e.g. cryosurgery, laser-induced Tattoo removal and others). Temperature control in the limit of 0 oC to +1 oC of the frozen tissue impregnated with NPs allows laser heating of ice confined in the matrix only in the fixed place of tissue. It is important for safe treatment of live tissues and cells.
Conclusion
SHT-synthesis of metal oxide bronzes can be used for generation of biofunctional NPs with high photo-thermal effect on frozen biotissue. Laser irradiation of frozen pig ear with molybdenum oxide bronze NPs allows a temperature control of biotissue thawing. Photo-thermal effect of laser radiation on frozen pig skin impregnated with NPs depends on absorption of the NPs, laser wavelength and intensity of radiation, and the temporal regimes of laser irradiation of tissue, as well. Photothermal effect of laser radiation on frozen pig skin impregnated with NPs of hydrogen-molybdenum oxide bronze is higher at the wavelength of 1.45 µm than at 1.56 µm.
Acknowledgement: Authors thanks Russian Foundation for Basic Research for financial aid in this work. Grant RFBR- 15-42-00106.
Список литературы/ References
- Baum O.I. Nanoparticles for diagnostics and laser medical treatment of cartilage in orthopaedics / O. I. Baum, Y. M. Soshnikova, A. I. Omelchenko et al // Proc. SPIE 8595, Colloidal Nanocrystals for Biomedical Applications VIII.- 85951K. doi: 10.1117/12.2008536
- Song X.-J. Microstructure and evolution of (TiB2+Al2O3)/NiAl composites prepared by self-propagation hightemperature synthesis / Xiao-jie Song,·Hong-zhi Cui,·Li-li Cao,·P.Y. Gulyaev // Transactions of Nonferrous Metals Society of China.- 2016.- Volume 26.- Issue 7.- Pages 1878-1884. doi: 10.1016/S1003-6326(16)64265-6
- Brus L.E. Magnetite Fe3O4 nanocrystals: Spectroscopic observation of aqueous oxidation kinetics / L.E. Brus, K.A. Bosnick, M. Myers, J. Tang// J.Phys. Chem. B.- 2003.- Volume 107.- P. 7501-7506.
- Gulyaev P.Yu. Plasma spraying of protective coatings from ferromagnetic SHS-materials / P.Yu. Gulyaev //Research Journal of International Studies.- 2013.- № 12-1 (19).- P. 74 -77.
- Comini G. Thermal aspects of cryosurgery/ G. Comini, S. Del Giudice // J. Heat Trans.- 1976.-Volume 98.- P. 543-549.
- Evstigneev V.V. Development Prospects of SHS Technologies in Altai State Technical University/ V.V. Evstigneev, P.J. Guljaev, I.V. Miljukova, V. D. Goncharov, V.A.Vagner and A.A. Gladkih// International Journal of Self-Propagating High-Temperature Synthesis.- 2006.- Т. 15. № 1.- P. 99-104.
- Gage A.A. Mechanisms of tissue injury in cryosurgery / A.A. Gage, J. Baust//Cryobiology.- 1998.- Volume 37, P. 171-186
- Garkol’ D.A. A new high-speed brightness pyrometry method to investigate self-propagating high-temperature synthesis/ D.A. Garkol’, P.Y. Gulyaev, V.V. Evstigneev, A.B. Mukhachev // Combustion, Explosion, and Shock Waves.- 1994.- Volume 30, Issue 1.- P. 72-76. doi: 10.1007/BF00787888
- Gulyaev P.Yu. SHT-Synthesis and application of biofunctional nanoparticals used high photo-thermal effect for laser heating of biotissues / P.Yu. Gulyaev, M.K. Kotvanova, A.I. Omelchenko, E.N. Sobol // The 23th Annual International Conference on Advanced Laser Technologies. ALT'15 Book of Abstracts.- 2015.- P. 46.
- Boronenko M.P. Increasing accuracy of high temperature and speed processes micropyrometry / M. P. Boronenko, P. Yu. Gulyaev, A. E. Seregin and А. G Bebiya // IOP Conference Series: Materials Science and Engineering.- 2015.- Vol. 93.- № 1.- 012021. DOI:10.1088/1757-899X/93/1/012021
- Gulyaev P.Y. Photothermal effects of laser heating iron oxide and oxide bronze nanoparticles in cartilaginous tissues/ P.Y. Gulyaev, M.K. Kotvanova, S.S. Pavlova, E.N. Sobol', A.I. Omel'chenko // Nanotechnologies in Russia.- 2012.- Т. 7. № 3- 4.- P. 127-131. doi: 10.1134/S1995078012020097
- Gulyaev I.P. New High-Speed Combination of Spectroscopic And Brightness Pyrometry For Studying Particles Temperature Distribution In Plasma Jets/ I.P. Gulyaev, K.A. Ermakov, P.Yu. Gulyaev//European researcher. Series A.- 2014.- № 3-2 (71).- P. 564-570.
- Gulyaev P.Yu. Temperature measurements for Ni-Al and Ti-Al phase control in SHS Synthesis and plasma spray processes / P.Yu. Gulyaev, I.P. Gulyaev, I.V. Milyukova, H.-Z. Cui // High Temperatures - High Pressures.- 2015.- Т. 44. № 2.- P. 83-92.
- Omelchenko A.I. Biofunctional magnetic “core-shell” nanoparticles generation via laser ablation of metallic iron in liquids/A.I. Omelchenko, E.N. Sobol, A.V. Simakin, A.A. Serkov, I.A. Sukhov, G.A. Shafeev //Laser Physics. -2015. - Vol. 25.- No.2. - 025607.
- Kotvanova M. Evoluation of combustion temperature and combustion speed of process of SH-Synthesis of titanium oxide bronze / M. Kotvanova, N. Blinova, P. Gulyaev, A. Dolmatov, S. Pavlova// International symposium on self-propagating high temperature synthesis SHS XIII.- 2015.- P. 160-161.
- Borodina K.A. Per-pixel adjustment of the afterglow effect EOC screen / K.A. Borodina, S.A. Sorokina, A. G. Bebiya, M.P. Boronenko, P.Yu. Gulyaev // Journal of Physics: Conference Series.- 2016.- Volume 741.- Number 1.- 012139. DOI:10.1088/1742-6596/741/1/012139
- Mitrovic S. Mobile small polarons and the Peierls transition in the quasi-one-dimensional conductor K0.3MoO3 / S. Mitrovic, L. Perfetti, G. Margaritondo, M. Grioni, L. Forro, L. Degiorgi, H. Hochst, // Phys. Rev. B.- 2002.-Volume 66.- 075107.
- Gulyaev I.P. Anomalous high-velocity outbursts ejected from the surface of tungsten microdroplets in a flow of argon-air plasma / I.P. Gulyaev, A.V. Dolmatov, P.Yu. Gulyaev, V.I. Iordan, M.Yu. Kharlamov, I.V. Krivtsun // IOP Conference Series: Materials Science and Engineering.- 2016.- Volume 110.- conference 1.- 012057 doi:10.1088/1757- 899X/110/1/012057
- Boronenko M.P. Phase formation time evaluation in NiAl combustion systems by the thermal fields visualization method/M.P. Boronenko, A.E. Seregin, P.Yu. Gulyaev, I.V. Milyukova //Scientific Visualization.- 2015.- Т. 7.- № 5.- P. 102-108.
- Sobol E.N. Study of cartilage shaping with holmium laser/ E.N. Sobol, V. Bagratashvili, A. Sviridov, A. Omel'chenko // Progress in Biomedical Optics and Imaging.- 1996.- Т. 2623.- P. 544-552.
- Sobol E.N. Laser reshaping and regeneration of cartilage/ E.N. Sobol, T.E. Milner, A.I. Omelchenko et al // Laser Phys. Lett.- 2007.- Volume 4(7).- P. 488-502. doi: 10.1002/lapl.200710019
- Soshnikova Y.M. Laser radiation effect on chondrocytes and intercellular matrix of costal and articular cartilage impregnated with magnetite NPs / Y.M. Soshnikova, A.B. Shekhter, A.I. Omelchenko et al // Lasers in Surgery and Medicine. 2015. Т. 47. № 3. P. 243-251. doi:10.1002/lsm.22331
- Pham Q.T. Food Freezing and Thawing Calculations/ Q.T. Pham.- Springer, 2014.- 152 p. doi: 10.1007/978-1-4939-0557-7_1
- Stefan, J.: Uber einiger probleme der theorie der warmeleitung. Sitzungsberichte der kaiserliche akademie der wissenschaften in Wien. XCVIII. 473-484 (1889) (in German).
- Vorotilin A.M. Cell injury at the stage of thawing / A.M. Vorotilin, A.V. Zinchenko, V.A. Moiseyev, // Cryo-Lett..- 1991.- Volume 12.- P. 77-86.
- Zhu J. Numerical study on the thawing process of biological tissue induced by laser radiation/J. Zhu, J. Lu, A. Yu // J. Biomech. Eng.-2005.- Volume 127, P. 416-431.
