ВЛИЯНИЕ ИНТЕНСИВНОСТИ ТРАНСКРАНИАЛЬНОЙ СТИМУЛЯЦИИ ПЕРЕМЕННЫМ ТОКОМ НА ВОЗБУДИМОСТЬ КОРЫ ГОЛОВНОГО МОЗГА ЧЕЛОВЕКА
ВЛИЯНИЕ ИНТЕНСИВНОСТИ ТРАНСКРАНИАЛЬНОЙ СТИМУЛЯЦИИ ПЕРЕМЕННЫМ ТОКОМ НА ВОЗБУДИМОСТЬ КОРЫ ГОЛОВНОГО МОЗГА ЧЕЛОВЕКА
Научная статья
Горина Е.О.1, *,Поздняков И.С.2, Моисеева В.В.3, Феурра М.4
1 ORCID: 0000-0002-0330-2659;
2 ORCID: 0000-0002-2450-7004;
3 ORCID: 0000-0001-8729-1610;
4 ORCID: 0000-0003-0934-6764;
1, 2, 3, 4 Центр нейроэкономики и когнитивных исследований, Институт Когнитивных Наук, Национальный исследовательский университет «Высшая школа экономики», Москва, Россия
* Корреспондирующий автор (e.o.gorina[at]gmail.com)
АннотацияДанное методологическое исследование посвящено оценке вклада параметра интенсивности (0.25-2 mA) Транскраниальной электрической стимуляции переменным током на частоте 20-Гц, в изменение активности первичной моторной коры.
В исследовании приняло участие 20 человек (10 женщин, возраст M=23.5, SD=4.16), без неврологических и психических заболеваний, травм головного мозга, правши. Испытуемые подтвердили, что не употребляли алкоголя, кофеина, и других препаратов, влияющих на работу ЦНС в дни предшествовавшие эксперименту.
Анализ данных не выявил значимых различий как между условиями с разными значениями интенсивности бета-стимуляции, так и относительно контрольных условий. Однако, были показаны накопительный эффект длительной стимуляции, и связь чувствительности к воздействию стимуляции со значением индивидуального моторного порога. Обнаруженная чувствительность к Транскраниальной электрической стимуляции переменным током в зависимости от моторного порога может внести значительный вклад в развитие когнитивных исследований и клинического применения.
Ключевые слова: ТСПеТ, ТМС, стимуляция мозга, интенсивность, электрическая стимуляция, магнитная стимуляция, электромиография, моторная кора.
INTENSITY EFFECTS OF TRANSCRANIAL ALTERNATING CURRENT STIMULATION ON HUMAN CORTICAL EXCITABILITY
Research article
Gorina E.O.1, *, Pozdniakov I.S.2, Moiseeva V.V.3 , Feurra M.4
1 ORCID: 0000-0002-0330-2659;
2 ORCID: 0000-0002-2450-7004;
3 ORCID: 0000-0001-8729-1610;
4 ORCID: 0000-0003-0934-6764;
1, 2, 3, 4 Centre for Cognition and Decision making, Institute for Cognitive Neuroscience, National Research University Higher School of Economics, Moscow, Russia
* Corresponding author (e.o.gorina[at]gmail.com)
AbstractThe present methodological study aimed to investigate the effects of Transcranial alternating current stimulation intensity (ranged 0.25-2 mA) influence of beta (20 Hz) stimulation of primary motor cortex and to test the hypothesis of these effects to be non-monotonic.
We collected data from 20 healthy subjects (age M=23.5, SD=4.16, 10 females), with no history of mental and neurological diseases or traumas, all right-handed, with no drug, alcohol or caffeine use in the days preceding the experiment.
On one hand, we observed not only the absence of any difference between all the beta intensity conditions into primary motor cortex excitation but also no difference between all beta conditions and neither alpha or sham controls nor baselines. On the other hand, our data revealed a cumulative effect of prolonged stimulation and sensitivity to this effect based on an individual motor threshold.
The finding that electrical stimulation effects can differ based on the individual motor threshold may have a great impact on clinical application and cognitive investigations.
Keywords: tACS, brain stimulation, NIBS, intensity, electrical stimulation, tES, TMS, EMG, motor cortex.
IntroductionTranscranial alternating current stimulation (tACS), Transcranial alternating current stimulation (tACS), though a rather novel non-invasive brain stimulation (NIBS) technique, was widely used in basic and clinical research. It allows influence cortical excitability and endogenous neural activity by application of external oscillating electrical current usually of sinusoidal waveform [1], [2]. The follow-up changes in intrinsic neuronal activity are shown to relate to changes in the motor, perception, and higher-level functioning [3], [4], [5]. Electrical current implementation results in only minor changes in neurons' resting potential, therefore tACS is a neuromodulation technique, which allows establishing a causal relationship between neurophysiology and behavioral performance.
Though tACS being an advantageous technique as it is easy to use, portable and rather safe, adverse events should be considered [6]. Depending on experimental design and individual characteristics of a subject, tACS has been shown to induce: retinal phosphenes or flashes, cutaneous perceptions, dizziness, pressure perception, mild headache, vertigo, back pain, stiff neck and others [7].
So far, tACS is assumed to affect intrinsic brain oscillations amplitude, coherence or phase via entrainment, plasticity, or stochastic resonance, thus either synchronizing and enhancing the efficacy of endogenous neuronal activity or inducing interfering noise [6], [13], [14]. Several studies suggest AC effects be due to changes in neurotransmitters (GABA, serotonin, norepinephrine) or neuropeptides release, but it is not clear whether those changes are directly related to stimulation [15], [17], [18]. It was also shown that tACS leads to some changes in neuronal plasticity, for example, 20 Hz applied over M1 induces NMDAR-mediated plasticity [20]. Interestingly, neural plasticity changes are attributed to as an after-effect of tACS, while online changes are associated with neural entrainment [21], [22].
A variety of studies showed that application of tACS protocols can effect (bidirectionally) motor, perception, and higher cognitive processes [23]. Along with that, tACS was widely studied as a potential treatment for different neurological and psychiatric disorders (e.g. stroke, Parkinson’s disease, schizophrenia, depression, ADHD, sleep disorders, OCD, etc.), but most of these studies are preliminary with low sample size [24], [25], [26].
Contradiction and low replicability in tACS experiments could be caused by differences in tACS protocols [24]. Indeed, some studies showed that varying frequency, phase, etc. we can achieve different results of tACS application. However, these parameters still haven't been studied systematically [10]. Since tES is used for rehabilitation and neuroenhancement purposes, it is mandatory to study any possible confounding effects and manipulation of stimulation parameters in order to define an optimal tACS protocol that may pave the way for rehabilitation in patients with motor and cognitive impairment.
Intensity is a crucial parameter to consider in tACS studies, cause we must be sure that current penetrates scalp and reaches brain tissue. In its turn, within- and between-subjects differences in neural tissue state and it's sensitivity to external current are to be considered. Finally, as there is still no clear explanation of the physiology of oscillating current implementation, more might not mean better, meaning that higher intensities not necessarily result in higher excitation of neural activity.
As we can see by reviewing existing literature, intensity impact into tACS effects of EEG frequency bands is underexplored unlike for other tES methods, that exhibit a non-linear effect for varied intensity levels [25], [26]. However, tACS will not necessarily show a similar non-linear pattern, as AC has a different mechanism.
Though tACS intensity effects have been marginally explored, yet one study reveals a non-monotonic dependency for frequencies outside the convenient range. Stimulation at 140 Hz over M1 at a lower intensity (0.2 mA) led to cortical inhibition, while at a higher intensity (1 mA) to excitation [27].
Given the aforementioned, in our project, we explored if tACS delivered at different intensities will affect motor cortex activity non-linearly. We used a combined tACS/TMS method for measuring tACS effects via TMS-induced motor evoked potentials (MEPs), proved to be a reliable technique for basic research [28], [28], [30]. Specifically, we varied the intensity of 20 Hz over M1 (in respect to sham and 10 Hz controls) and recorded MEPs with EMG from the right first dorsal interosseous muscle (FDI). We used beta frequency since it was shown that it effectively modulates M1 [30], [31].
Methods, subjects
To test our hypothesis we recruited 20 healthy subjects (age M=23.55, SD=4.16 years, 10 females, all right-handed), reporting no history of neurological and mental illness, traumas, or substance use for at least 24 hours preceding experiment. All subjects signed an informed consent form and received payment for participating. The study was accepted by the Higher School of Economics ethics committee at the National Research University, Moscow. During the experiment, subjects sat in a customizable chair with the relaxed right hand on the arm-pad. TMS-tACS We utilized a combined tACS/TMS method to mensurate M1 excitability changes.
We delivered TMS (MagPro x100 stimulator, MagVenture) through the 75-mm eight-shaped biphasic coil Butterfly C-B60 (MagVenture), placed tangential to the surface of the scalp. The coil handle was backward oriented, angled 45 oC to the midline sagittal axis of the participant’s head. For navigation, we used individual MRI scans (T1) and TMS Navigator (Localite GmbH). We induced MEPs by the series of single TMS pulses. We delivered TMS pulses at the individual level of intensity, set during the preparatory part of the experiment. TMS was delivered through the coil placed over the tACS electrode previously positioned over the M1 area on the scalp(«hot-spot»), associated with the subject's right FDI muscle.
To measure the target muscle (FDI) output, we used Electromyography (EMG) and Brain Vision Amplifier (Brain Products GmbH) at the 5 kHz sampling rate. We placed three EMG surface bipolar adhesive electrodes on the right hand in a bipolar belly-tendon montage: (1) one target electrode in the middle of FDI, (2) one reference - on the index finger joint, (3) and one ground - on the forearm.
We adopted tACS through the BrainStim (EMS Medical) at specific frequencies & intensities, with the maximum current density of 14.3 A/cm2 per electrode. To eliminate skin sensations, we kept impedance under 10 k (without DC offset). We used two tACS 5x7 cm2 electrodes (covered sponges soaked in saline) that were placed: (1) one above the "hot-spot", fixed with tapes and rubber head strips, (2) one on the ipsilateral shoulder as a reference.
During the preparatory part, we set the target stimulation point (“hot-spot”) coordinates at the left M1, which is the position of the coil at which TMS pulses elicited reliable MEPs from FDI. Further, we determined the resting motor threshold (MT) at this point using a staircase procedure to mete the minimal intensity of TMS stimulation that provides MEPs of 50 mV (peak-to-peak) in point 50% of cases (5 out of 10 pulses).
Further, due to the on-scalp tACS electrode refraction, we determined coordinates of the TMS coil now placed above the tACS electrode such that TMS pulses over it show a "hot-spot" behavior for FDI muscle. Following we measured the new (with tACS electrode) MT, again as the lowest TMS intensity value at which 5 out of 10 TMS pulses (from the new coil position) elicited MEPs > 50 mV.
Next, we found individual TMS intensity that we further used during the experimental part: the lowest intensity which provides 10 out of 10 MEPs with the amplitude ranged 600-1000 V peak-to-peak for TMS delivered over on-scalp tACS electrode that provides no electrical current.
In each experimental block either with or without tACS, we delivered a series of TMS pulses. After the experiment, subjects filled in a questionnaire on the sensations potentially felt from tACS (Fertonani, Ferrari, & Miniussi (2015) translated to Russian) [32].
Experimental design
The experiment was a within-subject with 2 control conditions (no-stimulation, 10 Hz), 2 baselines, and one session per subject. Each participant performed 12 blocks (of tACS+TMS, or TMS only) of 5 min 20 s each separated by 3 min breaks (to avoid carry-over effects).
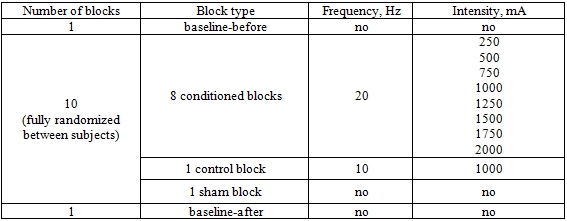
During each of 12 blocks, we delivered ≈30 TMS pulses separated by small time intervals (5-10 s). On behalf of data normalization and MEPs stability checking, the first and the last blocks were baselines («baseline-before» and «baseline-after» respectively) with only TMS applied. 10 blocks in-between baselines were conditioned and fully randomized in order: 8 tACS+TMS blocks (20 Hz at different intensities ranged from 0.25 to 2 mA with a step of 0.25 mA); one tACS+TMS control block of 10 Hz at 1 mA, and one sham block with no tACS (see Table 1).
Table 1 – Experimental conditions
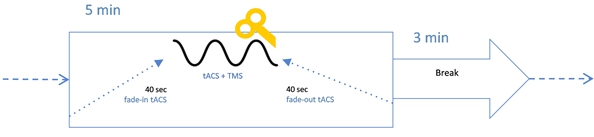
During each of the conditioned blocks we delivered: firstly 40 seconds of only tACS, with intensity rising from 0 to the value set for this block ("fade-in"); then 4 min 20 sec of full intensity tACS accompanied with 30 TMS pulses (full-on phase); then 40 seconds of only tACS , with intensity falling to 0 ("fade-out") (see Fig. 1).
Fig. 1 – Structure of a tACS+TMS block
The experiment duration was around 3.5 hours: preparatory part of 1.5 hours and experimental part of 2 hours.
Data analysis and results
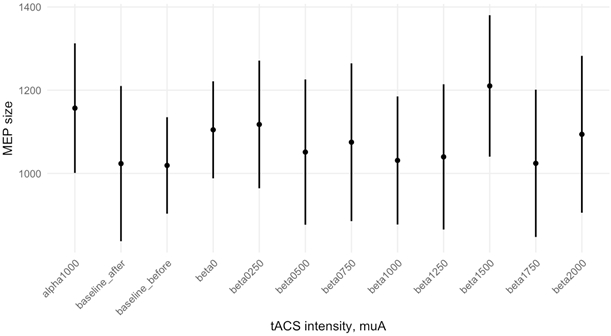
For data preprocessing and statistics, we used MATLAB 2014-a (The MathWorks Inc., Natick, MA, USA) and R-Studio 1.1.463 (R Foundation, Vienna, Austria). We firstly segmented and filtered EMG data with high-pass and notch filter (50 Hz) to remove power-line noise, then manually rejected artifacts and muscular activity (in a blind manner), and measured the peak-to-peak amplitude of each MEP. We excluded from further analysis all the MEPs that were: less than 50 mV (noise artifacts); higher or lower than 3 SD from the mean for the condition (outliers); recorded inside 2 seconds after the previous TMS pulse (to avoid charge accumulation effects). In total, we had from 20 subjects for analysis, on average, 28.75 (SD=3.81) MEPs per condition. For each subject, we averaged MEPs for each condition. We used ANOVA for statistical analysis, with Levene's Test for Homogeneity of Variance, Mauchly's Test for Sphericity, and Greenhouse-Geisser Sphericity Corrections. The significance level was set at 0.05. For Posthoc we used Bonferroni correction. The t-test between baselines (t = 2.734, df = 19, p = .013), revealed a significant difference which reflects that on average the MEP's size increase from the beginning to the end of the experiment. So, at first, we normalized data to the starting baseline (“baseline-before”). One-way repeated-measures ANOVA with block type as a factor showed no significant difference between conditions (F=0.68, p=.662), which contradicted our expectations on beta to effect MEPs compared to controls (Fig. 2).
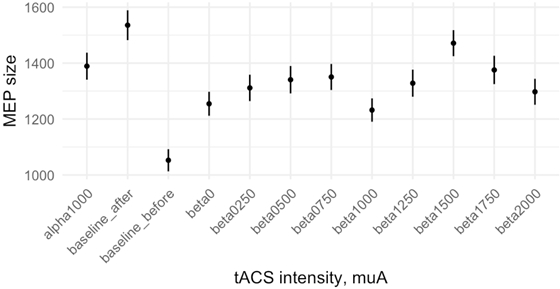
Fig. 2 – Relative changes of average MEP size in different conditions, calculated for raw data (error bar represents SEM)
Taking order effect into account (Fig. 3), we further decided to normalize MEP amplitudes (A) on the individual change between two baselines.
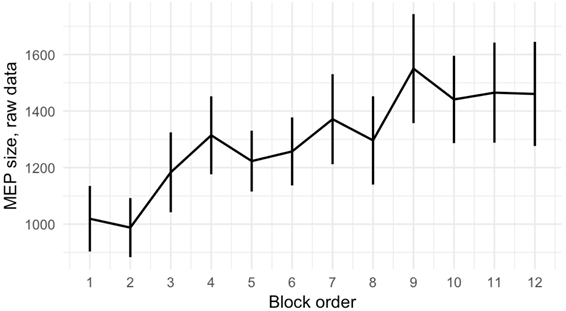
Fig. 3 – Average MEP size values for different order positions of a block, calculated for raw data (error bar represents SEM)
To do so, we firstly calculated individual “step-coefficient” (si) as the difference between last block (baseline-after) mean MEP amplitude (Āba) and first block (baseline-before) mean MEP (Ābb) divided by the number of blocks minus 1 (n-1). Then, for each subject, we subtracted from each block mean MEP value step-coefficient value multiplied by this block order number minus 1 (k-1).

One-way repeated-measures ANOVA with block type as a factor revealed no significant difference between conditions for data normalized this way (F= 0.486, p=. 843) (Fig. 4).
Fig. 4 – Relative changes of averaged for all subjects MEP size in different tACS conditions, calculated for data normalized to order effect (error bar represents SEM)Note: no significant difference between conditions (p=. 8426)
We further decided to check whether effects of different conditions depend on MT value, and divided subjects into two groups ("lower threshold", "higher threshold") according to their MT (with electrode) values, separated by median threshold value 50.5. Two-way ANOVA with MT and block type as factors showed a significant difference between threshold conditions (F=11.347, p=.003), with higher MEP values for subjects with higher MT, and insignificant interaction (F= 0.718, p= .721) (Fig. 5).
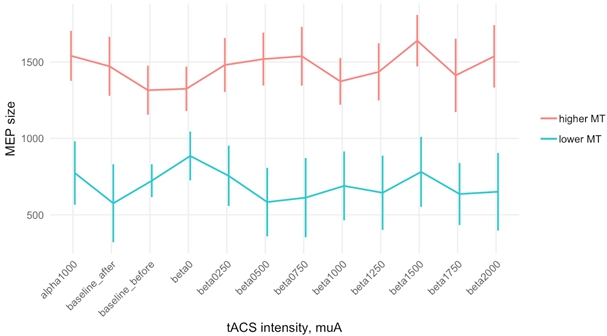
Fig. 5 – Average MEP size values for MT groups in different tACS conditions, calculated for data normalized to order effect (error bar represents SEM)
Note: a significant difference between threshold groups (p=.0034)
DiscussionIn our study, we aimed to investigate whether beta tACS intensity effects on M1 excitability would behave non-monotonically. Our data neither supports nor rejects this hypothesis, as soon as we have actually observed no significant difference neither between all the beta intensity conditions nor between any intensity of beta and controls (alpha, sham, both baselines). However, a block order dependent increase in MEP intensity has been found, along with different sensitivity to it depending on MT value (the lower it is, the lower is sensitivity).
Such results might be caused by the accumulation of modulatory effect either due to the too-short breaks between tACS blocks or due to the number of TMS pulses which could have both induced a linear increase of the MEPs size or lengthened the experiment thus making the experiment too tiring for the subjects, and tiredness affects cortical excitability [33], [34]. Another interpretation could be that for normalization to the order effect we used a rather rough linear approximation of the observed curve.
Our finding of MT-dependent sensitivity to tACS is counterintuitive, as we would rather expect subjects with lower MTs to be more responsive. The problem is that tACS is a transcranial method, so it is never clear how much of a charge penetrates the skull and reaches brain tissue, as it depends on skull and skin parameters.
Other parameters, on which individual MT value depends, are important to be considered, such as, for example, individual neuronal thresholds, neurotransmitters metabolism, membrane permeability, or difference between an individual intrinsic oscillation in M1 and an applied one [35].
Furthermore, in stroke patients, smaller MEPs size accompanied cortical-motoneurons loss, membrane excitability and charge conduction alterations [36]. From this perspective, the application of tACS might influence neuronal recruitment or membrane excitability, therefore, those who have lower MT seem to be less susceptible to benefits of tACS since their system is already optimized.
Limitations
Our results are limited as we do not measure any physiological parameters that could affect MEP amplitude. Moreover, TMS-EMG experiments have a rather long preparation part, during which the target brain area receives many TMS pulses. Also, the breaks between the blocks might be too short, thus not allowing wash out previous block effects. Thereby there may be a cumulative effect of electrical stimulation as shown by the serial order effects (Fig. 5). This research field requires a higher sample size and further replication studies by considering prominent individual differences shown in other tACS studies.
Conclusion
Our study revealed no significant effect of any intensity of beta tACS, however, we revealed accumulation of modulatory effect in case of prolonged stimulation. In addition, we found that sensitivity to tACS depends on individual MT. In order to disentangle the controversial effects observed in the current study, further studies are needed.
| Финансирование Работа выполнена при поддержке гранта Российского научного фонда (№ 17-11-01273). | Funding This work was supported by the Russian Science Foundation grant (No: 17-11-01273). |
| Конфликт интересов Не указан. | Conflict of Interest None declared. |
Список литературы / References
- Antal, A. Transcranial alternating current stimulation (tACS) [Electronic resource] / A. Antal, W. Paulus // Frontiers in Human Neuroscience. — 2013. — V. 7. — DOI: 10.3389/fnhum.2013.00317.
- Dowsett, J. Transcranial Alternating Current Stimulation with Sawtooth Waves: Simultaneous Stimulation and EEG Recording [Electronic resource] / J. Dowsett, C.S. Herrmann // Frontiers in Human Neuroscience. — 2016. — V. 10. — DOI: 10.3389/fnhum.2016.00135.
- Pogosyan, A. Boosting Cortical Activity at Beta-Band Frequencies Slows Movement in Humans / A. Pogosyan, L.D. Gaynor, A. Eusebio, P. Brown // Current Biology. — 2009. — V. 19, № 19. — P. 1637–1641.
- Helfrich, R.F. Entrainment of Brain Oscillations by Transcranial Alternating Current Stimulation / R.F. Helfrich, T.R. Schneider, S. Rach, S.A. Trautmann-Lengsfeld, A.K. Engel, C.S. Herrmann // Current Biology. — 2014. — V. 24, № 3. — P. 333–339.
- Cohen Kadosh, R. Modulating and enhancing cognition using brain stimulation: Science and fiction / R. Cohen Kadosh // Journal of Cognitive Psychology. — 2015. — V. 27, № 2. — P. 141–163.
- Santarnecchi, E. Enhancing cognition using transcranial electrical stimulation / E. Santarnecchi, A.-K. Brem, E. Levenbaum, T. Thompson, R.C. Kadosh, A. Pascual-Leone // Current Opinion in Behavioral Sciences. — 2015. — V. 4. — P. 171–178.
- Matsumoto, H. Adverse events of tDCS and tACS: A review / H. Matsumoto, Y. Ugawa // Clinical Neurophysiology Practice. — 2017. — V. 2. — P. 19–25.
- Alekseichuk, I. Spatial Working Memory in Humans Depends on Theta and High Gamma Synchronization in the Prefrontal Cortex / I. Alekseichuk, Z. Turi, G. Amador de Lara, A. Antal, W. Paulus // Current Biology. — 2016. — V. 26, № 12. — P. 1513–1521.
- Polanía, R. The precision of value-based choices depends causally on fronto-parietal phase coupling / R. Polanía, M. Moisa, A. Opitz, M. Grueschow, C.C. Ruff // Nature Communications. — 2015. — V. 6, № 1. — P. 8090.
- Antal, A. Transcranial Alternating Current and Random Noise Stimulation: Possible Mechanisms / A. Antal, C.S. Herrmann // Neural Plasticity. — 2016. — V. 2016. — P. 1–12.
- Liu, A. Immediate neurophysiological effects of transcranial electrical stimulation / A. Liu, M. Vöröslakos, G. Kronberg, S. Henin, M.R. Krause, Y. Huang, A. Opitz, A. Mehta, C.C. Pack, B. Krekelberg, A. Berényi, L.C. Parra, L. Melloni, O. Devinsky, G. Buzsáki // Nature Communications. — 2018. — V. 9, № 1. — P. 5092.
- Zaehle, T. Transcranial Alternating Current Stimulation Enhances Individual Alpha Activity in Human EEG / T. Zaehle, S. Rach, C.S. Herrmann // PLoS ONE. — 2010. — V. 5, № 11. — P. e13766.
- Vosskuhl, J. Non-invasive Brain Stimulation: A Paradigm Shift in Understanding Brain Oscillations / J. Vosskuhl, D. Strüber, C.S. Herrmann // Frontiers in Human Neuroscience. — 2018. — V. 12. — P. 211.
- Zaghi, S. Noninvasive Brain Stimulation with Low-Intensity Electrical Currents: Putative Mechanisms of Action for Direct and Alternating Current Stimulation / S. Zaghi, M. Acar, B. Hultgren, P.S. Boggio, F. Fregni // The Neuroscientist. — 2010. — V. 16, № 3. — P. 285–307.
- Nowak, M. Driving Human Motor Cortical Oscillations Leads to Behaviorally Relevant Changes in Local GABA A Inhibition: A tACS-TMS Study / M. Nowak, E. Hinson, F. van Ede, A. Pogosyan, A. Guerra, A. Quinn, P. Brown, C.J. Stagg // The Journal of Neuroscience. — 2017. — V. 37, № 17. — P. 4481–4492.
- Kirsch, D.L. The Science Behind Cranial Electrotherapy Stimulation / D.L. Kirsch — P. 234.
- Wischnewski, M. NMDA Receptor-Mediated Motor Cortex Plasticity After 20 Hz Transcranial Alternating Current Stimulation / M. Wischnewski, M. Engelhardt, M.A. Salehinejad, D.J.L.G. Schutter, M.-F. Kuo, M.A. Nitsche // Cerebral Cortex. — 2019. — V. 29, № 7. — P. 2924–2931.
- Pikovsky, A. Synchronization: A universal concept in nonlinear sciences [Electronic resource] / A. Pikovsky, M. Rosenblum, J. Kurths. — Cambridge: Cambridge University Press, 2001.
- Thut, G. Entrainment of Perceptually Relevant Brain Oscillations by Non-Invasive Rhythmic Stimulation of the Human Brain [Electronic resource] / G. Thut, P.G. Schyns, J. Gross // Frontiers in Psychology. — 2011. — V. 2. — DOI: 10.3389/fpsyg.2011.00170.
- Herrmann, C.S. Transcranial alternating current stimulation: a review of the underlying mechanisms and modulation of cognitive processes [Electronic resource] / C.S. Herrmann, S. Rach, T. Neuling, D. Strüber // Frontiers in Human Neuroscience. — 2013. — V. 7. — DOI: 10.3389/fnhum.2013.00279.
- Gomez Palacio Schjetnan, A. Transcranial Direct Current Stimulation in Stroke Rehabilitation: A Review of Recent Advancements / A. Gomez Palacio Schjetnan, J. Faraji, G.A. Metz, M. Tatsuno, A. Luczak // Stroke Research and Treatment. — 2013. — V. 2013. — P. 1–14.
- Gonzalez-Burgos, G. GABA Neurons and the Mechanisms of Network Oscillations: Implications for Understanding Cortical Dysfunction in Schizophrenia / G. Gonzalez-Burgos, D.A. Lewis // Schizophrenia Bulletin. — 2008. — V. 34, № 5. — P. 944–961.
- Brittain, J.-S. Tremor Suppression by Rhythmic Transcranial Current Stimulation / J.-S. Brittain, P. Probert-Smith, T.Z. Aziz, P. Brown // Current Biology. — 2013. — V. 23, № 5. — P. 436–440.
- Nitsche, M.A. On the Use of Meta-analysis in Neuromodulatory Non-invasive Brain Stimulation / M.A. Nitsche, M. Bikson, S. Bestmann // Brain Stimulation. — 2015. — V. 8, № 3. — P. 666–667.
- Batsikadze, G. Partially non-linear stimulation intensity-dependent effects of direct current stimulation on motor cortex excitability in humans: Effect of tDCS on cortical excitability / G. Batsikadze, V. Moliadze, W. Paulus, M.-F. Kuo, M.A. Nitsche // The Journal of Physiology. — 2013. — V. 591, № 7. — P. 1987–2000.
- Hoy, K.E. Testing the limits: Investigating the effect of tDCS dose on working memory enhancement in healthy controls / K.E. Hoy, M.R.L. Emonson, S.L. Arnold, R.H. Thomson, Z.J. Daskalakis, P.B. Fitzgerald // Neuropsychologia. — 2013. — V. 51, № 9. — P. 1777–1784.
- Moliadze, V. Close to threshold transcranial electrical stimulation preferentially activates inhibitory networks before switching to excitation with higher intensities / V. Moliadze, D. Atalay, A. Antal, W. Paulus // Brain Stimulation. — 2012. — V. 5, № 4. — P. 505–511.
- Kanai, R. Transcranial alternating current stimulation (tACS) modulates cortical excitability as assessed by TMS-induced phosphene thresholds / R. Kanai, W. Paulus, V. Walsh // Clinical Neurophysiology. — 2010. — V. 121, № 9. — P. 1551–1554.
- Shpektor, A. Effects of Transcranial Alternating Current Stimulation on the Primary Motor Cortex by Online Combined Approach with Transcranial Magnetic Stimulation / A. Shpektor, M. Nazarova, M. Feurra // Journal of Visualized Experiments. — 2017. — № 127. — P. 55839.
- Feurra, M. State-Dependent Effects of Transcranial Oscillatory Currents on the Motor System: What You Think Matters / M. Feurra, P. Pasqualetti, G. Bianco, E. Santarnecchi, A. Rossi, S. Rossi // Journal of Neuroscience. — 2013. — V. 33, № 44. — P. 17483–17489.
- Feurra, M. Frequency-Dependent Tuning of the Human Motor System Induced by Transcranial Oscillatory Potentials / M. Feurra, G. Bianco, E. Santarnecchi, M. Del Testa, A. Rossi, S. Rossi // Journal of Neuroscience. — 2011. — V. 31, № 34. — P. 12165–12170.
- Fertonani, A. What do you feel if I apply transcranial electric stimulation? Safety, sensations and secondary induced effects / A. Fertonani, C. Ferrari, C. Miniussi // Clinical Neurophysiology. — 2015. — V. 126, № 11. — P. 2181–2188.
- Huber, R. Human Cortical Excitability Increases with Time Awake / R. Huber, H. Mäki, M. Rosanova, S. Casarotto, P. Canali, A.G. Casali, G. Tononi, M. Massimini // Cerebral Cortex. — 2013. — V. 23, № 2. — P. 1–7.
- Sharples, S.A. Cortical Mechanisms of Central Fatigue and Sense of Effort / S.A. Sharples, J.A. Gould, M.S. Vandenberk, J.M. Kalmar // PLOS ONE. — 2016. — V. 11, № 2. — P. e0149026.
- Deans, J.K. Sensitivity of coherent oscillations in rat hippocampus to AC electric fields / J.K. Deans, A.D. Powell, J.G.R. Jefferys // The Journal of Physiology. — 2007. — V. 583, № 2. — P. 555–565.
- Cortes, M. Transcranial Magnetic Stimulation as an Investigative Tool for Motor Dysfunction and Recovery in Stroke: An Overview for Neurorehabilitation Clinicians: TMS IN MOTOR DYSFUNCTION AND RECOVERY IN STROKE / M. Cortes, R.M. Black-Schaffer, D.J. Edwards // Neuromodulation: Technology at the Neural Interface. — 2012. — V. 15, № 4. — P. 316–325.