МЕТОДОЛОГИЯ НАТУРНЫХ ИССЛЕДОВАНИЙ МАССООБМЕНА ТЯЖЁЛЫХ МЕТАЛЛОВ В СИСТЕМЕ «ВОДА–ДОННЫЕ ОТЛОЖЕНИЯ»
Толкачёв Г.Ю.
Кандидат географичеких наук, Институт водных проблем Российской академии наук
МЕТОДОЛОГИЯ НАТУРНЫХ ИССЛЕДОВАНИЙ МАССООБМЕНА ТЯЖЁЛЫХ МЕТАЛЛОВ В СИСТЕМЕ «ВОДА–ДОННЫЕ ОТЛОЖЕНИЯ»
Аннотация
Одним из важнейших показателей качества воды является содержание тяжёлых металлов (ТМ) и их соединений. Поступая в речную сеть и откладываясь в донных отложениях, эти соединения начинают играть весьма активную роль в процессах биогеохимической миграции, могут в определённых условиях переходить в поровые растворы и далее в придонные воды. Распределение и миграция металлов в водных системах контролируются в основном характером взаимодействия донных отложений, водной массы и биоты. Направленность потока массообмена в системе «вода–донные отложения» определяет опасность вторичного загрязнения водных масс водоёма.
Ключевые слова: донные отложения, тяжёлые металлы, подвижные формы, твёрдая фаза, поровый раствор.
Tolkachev G.Y.
PhD in Geography, Institute of Water Problems of Russian Academy of Sciences
METHODOLOGY OF STUDYING IN NATURE THE MASS EXCHANGE OF HEAVY METALS IN THE “WATER - BOTTOM SEDIMENTS” SYSTEM
Abstract
Bottom sediments reveal a considerable accumulation of different pollutants, including heavy metals and their compounds as most dangerous for the water quality. Under definite conditions the heavy metals being accumulated in bottom sediments can transfer into the water again. Their mass exchange in the “water - bottom sediments” system is largely dependent on the heavy metal forms, in which they are present in bottom sediments. The problem relating to translocation of heavy metals in the water - bottom sediments system and quantification of their intensive mass exchange in different seasons of the year becomes very acute and allows judging about unwanted hazardous second water contamination.
Keywords: bottom sediment, heavy metals, mobile forms, solid phase, pore solution.
IntroductionThe bottom sediments in water reservoirs play a double role being simultaneously not only as an accumulator of elements and their compounds but also as a source of the second water pollution. Many natural factors combined with those caused by human activities are conducive to peculiar element turnover being a matter of some difficulty for its comprehensive study. It concerns especially the turnover of heavy metals (HM), which exist in natural waters and bottom sediments as assigned to various chemical forms. The contamination with heavy metals occurs from many sources such as dry and wet precipitation, atmospheric deposition from smelting operations, soils and vegetation in the catchment area, ground waters, application of sewage-sludge, industrial and municipal waste, agricultural landscapes, surface runoff, etc. Having different ways for input to a water system, the heavy metals become affected by processes of hydrolysis, sorption, desorption, sequestration, deposition, thus changing the forms of their existence. In suspensions and bottom sediments they are transferred from liquid into solid phase, precipitated as hardly soluble compounds to enter the water again. The dominating processes occur in dependence on geographical location of the water system, its hydrological regime, peculiar chemical composition of water, bioproductivity, peculiarities of soil and vegetation cover in the catchment area and a massive human intervention.
The mechanisms responsible for HM accumulation in bottom sediments are the following:
- sorption by suspended mineral particles derived from the catchment area to be precipitated again,
- sorption and accumulation by hydrobionts with subsequent sedimentation of postletal residues,
- sorption by pellets, living hydrobionts to be deposited later,
- sorption by bottom sediments,
- sedimentation of heavy metals at the water/bottom interface resulted from the outcrop of ground water,
- sedimentation of hardly soluble compounds to be precipitated again together with Fe and Mn hydroxides,
- molecular diffusion from water into the pore solution,
- removal of suspended particles resulted from water filtration by hydrobionts.
The above processes lead to HM accumulation in bottom sediments. The heavy metals enter an aquatic system in different forms frequently inherited from soils of the catchment area. They can be redistributed and change their forms due to diagenetic processes occurring in bottom sediments. The HM forms exert an effect on the composition and concentration of the pore solution, close connection with the solid phase, availability by hydrobionts, toxicity level.
The heavy metals can be removed from bottom sediments in abiotic and biotic way. The mechanisms of abiotic removal are as follows:
- molecular diffusion from the pore solution into the water,
- convective removal during the outcrop of ground water
- removal resulted from the suspended surface layer of bottom sediments,
- sequestration and desorption by organic ligands,
- dissolution of Fe and Mn hydroxides under definite redox conditions in the water system,
- compaction of sediments.
The biotic way is a complex of processes promoting the vital activity of hydrobionts and leading to changes in the acidic properties of bottom sediments, redox conditions, salinity, concentration of organic compounds as well as to the increase in the effective porosity. Biological mixing and absorption of macrophytes by roots contribute to removal of heavy metals from bottom sediments as well.
All these processes are proceeding simultaneously but show different trend and intensity in space and time. The available methods to measure the heavy metal flows at the water/bottom interface will entail great difficulties. As a matter of fact, some processes cannot be assessed under natural conditions, a quantitative assessment of the other processes requires permanent observations for a long period of time. Even the method widely applied to quantify the diffusion current using the mean concentration gradient in water near the bottom and the pore solution needs in every effort to provide approximate results of measuring the HM removal from bottom sediments.
In laboratory the study of HM removal in samples taken in bottom sediments fails to simulate the processes of diagenesis and accumulation, which are constantly changed in time and control not only the removal of heavy metals but also their turnover in water reservoirs.
The existing methods of experimental measuring are subject to some restriction. They don’t permit to determine a resultant of all the accumulation and removal processes, the forms of HM existence, in which they are transferred at the water/bottom interface, and to assess diagenetic processes causing transformation of geochemical conditions so dangerous for the second contamination of the water reservoir.
In view of this, there is an urgent need in searching another methods to study the heavy metal turnover in aquatic systems with slower water exchange, where great amounts of loose debris are accumulated being enriched with heavy metals and the other pollutants.
Object of research
The idea to determine element changes in pore solution and solid phase of bottom sediments for a short period of time and to quantify accumulation and removal of pollutants has been implemented in the catchment area of Ivankovsky water reservoir in the Upper Volga region.
Aims and objectives
The present study was carried out with the aim at identifying the HM foms in the pore solution and solid phase of bottom sediments and quantifying the accumulation and removal of these forms in nature.
The objectives of this study were:
- to assess the recent contamination level of bottom sediments by heavy metals taking into complete account the forms of their existence,
- to determine the distribution pattern of heavy metals in bottom sediments and to give quantitative characteristics of their total accumulation,
- to study the seasonal dynamics of HM mobile forms in the solid phase,
- to estimate deposits of HM mobile forms, their seasonal dynamics in the solid phase and the possible accumulation and removal from bottom sediments in different reaches of Ivankovsky water reservoir.
Materials and methods
To assess a resultant of accumulation and removal processes of microelements in bottom sediments a combined method was employed to conduct the survey of bottom sediments in definite seasons of a year and monthly observations in two definite points. As a result of bottom surveying the upper (10cm) layer of sediments was studied using cross profiles at a distance from 1 to 5 km between them. Sampling was taken by stratometer to define vertical distribution of elements as well as by bottom scoop (Petersen D-25). The samples were subject to centrifugation to extract the pore solution. The total HM content in samples and the pore solution was determined by atomic absorption spectrometer (Perkin-Elmer-460).
In points of observations the samples of water near the bottom and bottom sediments were taken in different seasons of a year and separated into the solid phase and the pore solution. However, it is impossible to study HM migration, accumulation by aquatic organisms and water toxic properties using only the total content of heavy metals. The investigation results may be reliably interpreted as based upon data about the forms, in which the heavy metals are present in water near bottom and in bottom sediments. By this reason, apart from the total content of heavy metals their forms have been analyzed in pore solution and in the solid phase of bottom sediments with the help of sequential extraction.
The 1st extract – treatment of samples with ammonium-acetate buffer at pH=48 (108 ml 98% CH3COOH + 78 ml 25% NH4OH + 800 ml H2O) to extract exchangeable and soluble forms of heavy metals.
The 2nd extract – treatment of samples with 30% H2O2 to extract forms bound to the organic matter.
The 3rd extract – treatment with 0.5 M Na + NaHCO3 + Na2S2O4 to extract forms bound to amorphous Fe and Mn hydroxides at pH=7.3.
Different HM forms were extracted by ionites: diethylaminoethyl cellulose (DEAE) and carboxymethyl cellulose (CM). They permit to determine the forms by charge sign. DEAE-cellulose promotes extracting the forms bound to fractions of humic-and fulvoacids and low molecular organic acids, whilst CM-cellulose – ionic forms and complexes with aminoacids, protein and polypeptides. Only the forms bound to neutral colloides, polysaccharides and free reducing saccharides remain in the solution [1]. The mass-spectrometer (“Plasma Quad 3” produced by “Fisons Instruments Elemental Analysis, England) was used for these purposes.
Based upon the data of bottom surveying it seemed reasonable to determine the total content of heavy metals in the solid phase of the upper (10cm) layer of bottom sediments and in the pore solution in samples taken in different reaches of Ivankovsky water reservoir.
The data obtained in points of observation allowed determining heavy metals stabilized in different extent in the solid phase and the dynamics of their changes for a year.
Results
The bottom sediments of the above water reservoir are formed at the expense of suspended elements annually accumulated in the amount of 15665 thou t or 71% from the total content of loose debris, represented by products of beach destruction (66%) and a share of suspended elements (34%) entering the reservoir together with river waters [3].
In the Volga river higher than Ivankovsky reservoir the heavy metals are moving in the kind of suspensions. Due to sedimentation of suspensions taking place in Ivankovsky water reservoir the balance is sharply changed towards dissolved HM forms. It is especially typical for such elements as Pb, Cr, Cd, Ni, Fe, Zn and speaks about their accumulation in the solid phase of bottom sediments. The major part of microelements is uniformly distributed in bottom sediments, what is confirmed by variation coefficients (Cv) of HM concentrations (Table 1).
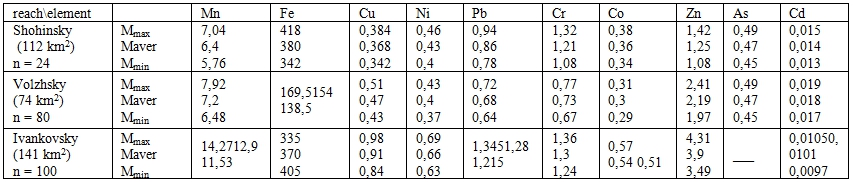
For calculating the HM amount in the solid phase and the pore solution of the upper (10cm) layer in the above reaches we used the mean values of properties including the bulk density of bottom sediments – 1.5 g/cm3 and the porosity of the upper horizon – 0.6. Table 2 demonstrates the mass of accumulated heavy metals in intervals at α = 0.95. It is evident that Ivankovsky reach being the largest and the last link in a chain reveals the maximum content of heavy metals with exception of Cd.
Table 1 - Standard deviations (σ) and variation coefficients (Cv) of the total content of heavy metals in bottom sediments in different years
| (σ/Сv) | |||
| Shoshinsky reach | Volzhsky reach | Ivankovsky reach | |
| Mn | 4,6/0,17 | 5,1/0,19 | 5,77/0,2 |
| Zn | 4,2/0,11 | 4,9/0,13 | 5,63/0,16 |
| Cu | 2,7/0,1 | 2,64/0,14 | 2,82/0,15 |
| Pb | 2,45/0,11 | 3,02/0,12 | 3,86/0,15 |
| Cd | 0,12/0,01 | 0,18/0,01 | 0,22/0,015 |
| Ni | 1,2/0,07 | 1,7/0,08 | 2,03/0,1 |
| Cr | 1,9/0,09 | 2,52/0,1 | 3,24/0,13 |
Table 2 - Maximal, minimal and mean values of the heavy metal content (thou t) in the upper (10cm) layer of bottom sediments (summer survey in 2002)
As a result of summer survey it was found that the maximum of HM dissolved forms is in the pore solution separated from samples of Ivankovsky reach (Table 3).
Comparing the mass of heavy metals in the solid phase and the pore solution shows that their share in the pore solution is practically insignificant in the summer period. It is worth emphasizing that the role played by pore solution in the second water contamination doesn’t limited by the content of dissolved heavy metals. A part of them assigned to the solid phase of bottom sediments is transferred into the pore solution and then into the water under definite conditions. Having migrated from bottom sediments into the water the heavy metals display a transit through the pore solution.
Table 3 - Annual accumulation of heavy metals in the pore solution (numerator) and in the solid phase of the upper (10cm) layer of bottom sediments in Ivankovsky reach (denominator)
| reach\element | Cu | Ni | Pb | Cr | Co | Zn |
| Shoshinsky (112 km2) | 0,8/ 2,19 | 9,8/ 2,45 | 1,8/ 0,21 | 20,9/ 1,72 | 1,4/ 0,39 | 25,4/ 1,45 |
| Volzhsky (74 km2) | 3/ 0,81 | 6,6/ 1,74 | 1,8/ 0,26 | 3,2/ 0,38 | 2,6/ 0,87 | 28/ 1,08 |
| Ivankovsky (141 km2) | 85/ 7,08 | 34/ 3,54 | 32/ 2,5 | 38/ 3,27 | 13,8/ 1,63 | 165/ 2,39 |
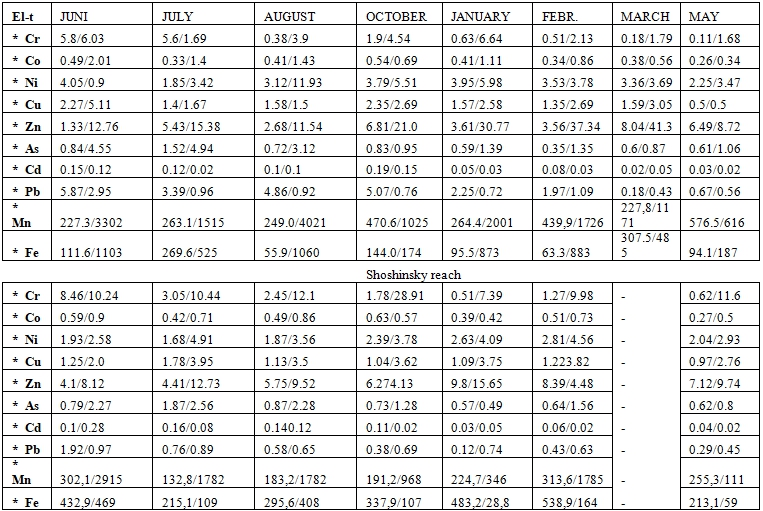
The content of Cr, Co, Ni, Zn, As, Cd, Pb, Fe, Mn in the layer near bottom, in the pore solution and the solid phase of bottom sediments was studied for a year in two points of observation located in different hydrodynamic conditions. The point of observation “Ploski” near Ploski village has a very active water exchange, while in “Shoshinsky reach” point the water exchange is slower. In both points of observation the water temperature gets higher from March to August and then it falls reaching its minimum in the winter. In May and Juni the content of dissolved oxygen (8-8.5 mg/l) is rather high in the layer near bottom being declined to 1.3 mg/l in July in “Ploski” and 1.6 mg/l in August in “Shoshinsky reach”. It is explained by appearance of stable water stratification. In the autumn homothermy (in October) the oxygen content increases up to 8.5 mg/l being sharply decreased after the formation of the ice cover (2.7-3.3 mg/l) in February-March. In the spring it shows an increase to 7.5-8.2 mg/l. Thus, in spite of decreasing the dissolved oxygen content during the summer and winter period the redox conditions remain in the water near the bottom sediments and their potential varies from +340 mB in winter to +440 mB in spring. One should mention that the bottom sediments have an oxidized layer of 3cm thick (Eh = 70-80 mB), which becomes reduced with depth (Eh =-170-180 mB at a depth of 10-12cm). pH is neutral and slightly alkaline for a year. So, one should stress that the water near bottom and the pore solution reveal no sharply expressed seasonal changes in redox potential and pH value. The data about the HM content in the water near bottom and in the pore solution obtained in two points of observation are presented in Table 4. In all the seasons the concentration of heavy metals except Pb and Cd is higher in the pore solution as compared to the water near bottom. It speaks about their constant flow from bottom sediments into the water. Regularities of changes in concentration of the major heavy metals haven’t been found in the water near bottom and pore solution. The absence of correlation between the HM concentration in water and the pore solution serves as evidence that their chemical composition is formed independently from each other under influence of different processes.
Sequential extraction of samples taken in both points of observation allowed determining the amount of heavy metals in exchangeable forms and in forms bound to organic matter and absorbed by amorphous Fe and Mn hydroxides (Table 5). One should indicate that the sum of HM mobile compounds is very high in the solid phase of bottom sediments. It is especially characteristic of Cu, Zn, Cd, Cr in Ploski and - Zn, Cd in Shoshensky reach.
As seen from Table, the data obtained in Ploski point of observation show the ion-exchangeable forms of Co, Cd, Mn and this form is predominant for Cr, Ni, Pb and Fe. The Cu mobile form is assigned to those bound to the organic matter of bottom sediments. The form bound to Fe and Mn hydroxides is typical for Pb, As, Fe and practically not typical for Co, Ni, Cu, Cd, Mn. All these forms of Zn reveal an identical distribution. As regards Shoshinsky reach there are the same regularities in distribution of mobile forms. Co, Zn, Cd are found in ion-exchangeable forms, this form is also characteristic of Cr, Ni, Pb and As especially in the winter. The major part of mobile Cu is bound to the organic matter; this form is inherent to Cr, Co, Ni, Zn, Cd. The form bound to Fe and Mn hydroxides is dominant for Pb and As only in the summer period, whilst ion-exchangeable forms become prevailed in winter and spring. One should stress again that Co, Cd, Zn and in the winter period Pb, As are found to be most mobile in bottom sediments of Shoshinsky reach.
Table 4 - The content of microelements in the water near bottom (numerator) and the pore solution (denomenator)
Note: - no observationsTable 5 - The share of mobile forms in the total content of heavy metals in the solid phase of bottom sediments in Ploski (numerator) and Shoshensky reach (denominator) in %
| JUNI | AUGUST | OCTOBER | JANUARY | MAY | |
| Cr | 58.1/10.88 | 51.35/13.02 | 50.7/15.31 | 50.78/11.16 | 54.08/15.6 |
| Co | 17.3/19.12 | 20.64/21.5 | 19.11/24.23 | 20.01/6.42 | 24.27/17.48 |
| Ni | 23.14/23.0 | 24.36/27.08 | 22.46/22.19 | 23.24/4.93 | 21.49/17.78 |
| Cu | 75.56/18.11 | 75.27/21.58 | 78.03/22.35 | 87.74/13.59 | 70.08/17.09 |
| Zn | 52.9/59.1 | 58.55/64.47 | 37.46/70.25 | 43.13/41.02 | 41.12/58.59 |
| As | 14.4/11.68 | 10.42/13.27 | 9.2/15.21 | 12.14/5.96 | 13.04/15.7 |
| Cd | 60.16/58.16 | 68.69/66.91 | 61.1/61.09 | 59.74/26.95 | 60.44/49.86 |
| Pb | 11.05/8.48 | 12.49/11.75 | 15.16/16.43 | 15.13/4.17 | 15.1/11.0 |
| Mn | 19.1/23.3 | 31.4/59.6 | 19.1/25.5 | 20.9/15.7 | 20.7/17.6 |
| Fe | 39.2/30.1 | 56.8/42.1 | 56.7/56.8 | 54.4/22.1 | 67.8/38.7 |
The mobile forms of all the studied microelements are highly changed, their variation coefficient varies in the range of 0.18-1.04 in Ploski (Volzhsky reach) and 0.42-1.06 in Shoshinsky reach. It is worth of note that slightly mobile (crystalline) forms have a very low variation coefficient equaled to 0.001-0.055 in Volzhsky reach and 0.002-0.040 in Shoshinsky reach.
It seems reasonable to conclude that due to changes in the amount of HM mobile forms the total content of heavy metals is also changed in bottom sediments.
The study carried out in Kuibyshev water reservoir and later in the other reservoirs of the Volga river cascade showed that the ratio between different forms of heavy metals in the solid phase of bottom sediments remains unchanged within the entire area of water reservoir; it is changed only in seasons [2]. By this reason, to evaluate the content of heavy metals assigned to different forms in the solid phase of bottom sediments in Ivankovsky water reservoir, the investigation results obtained in Ploski point and Shoshinsky reach found an application in our study.
The data about the total content of heavy metals obtained in both points of observation during the summer period seemed identical to those characteristic of reaches, that is why they may be used for calculating the areas of all the reaches in Ivankovsky water reservoir. These results have an estimate character and may be specified in future by those obtained in a greater quantity of points for observation.
In our study the investigation results obtained in both points of observation were used for determining the percentage of heavy metals with different mobility degree in every reach. Table 6 presents the data about the monthly content of differently mobile elements in the solid phase of the upper (10cm) layer of bottom sediments. They provide evidence that the amount of HM mobile forms is rather significant.
It is worth emphasizing that the mobile forms can take part in mass exchange processes within the “solid phase – pore solution – water” system and their amount is largely dependent on factors responsible for changing constituents of element balance, hydrodynamic conditions in the layer near bottom, physical-chemical properties of the upper 10cm layer in bottom sediments. One should conclude that the mobile forms of the studied heavy metals show considerable changes in this upper layer which is intensively involved into the mass exchange process with water. Seasonal changes in the content of these elements are characteristic not only of ion-exchangeable forms but also mobile forms.
Based on the data obtained in all the reaches it is possible to estimate a summary annual removal and accumulation of heavy elements in the solid phase of bottom sediments (Table 6).
Table 6 - Annual removal and accumulation of heavy metals in the solid phase of bottom sediments in Ivankovsky water reservoir
| Element | Volzhsky reach | Ivankovsky reach | Shoshinsky reach | |||
| removal | accumulation | removal | accumlation | removal | accumulation | |
| Cr | 162,76 | 135,76 | 67,42 | |||
| Co | 106,64 | 203,66 | 72,5 | |||
| Ni | 7,56 | 24,83 | 34,85 | |||
| Cu | 51,68 | 129,75 | 24,39 | |||
| Zn | 307,88 | 777,94 | 15,46 | |||
| Cd | 1,22 | 0,71 | 4,82 | |||
| Pb | 18,02 | 61,49 | 33,38 | |||
| Mn | 37,7 | 371,7 | 436,5 | |||
| Fe | 99,2 | 257,75 | 413 | |||
In the winter season the groundwater bed is subject to upload in the volume of 0.5-5 m3/24 hours per one linear meter. In this time the microelements are removing from pore solution due to convective mixing followed by molecular transfer. The total suspension current of microelements from bottom sediments is formed by convective transfer with average filtration velocity (ν) and diffusional transfer with coefficient of molecular diffusion (Dm). The possible value of this flow is estimated by the following way. In physical chemistry the process of mass transfer in the sorbing porous medium is described by the sum of convective and diffusional flows:
j = – Def σС/σx + vC (1),
where Def – effective coefficient of diffusion with account of the influence exerted by dispersion in dependence of the filtration velocity and sorption upon the diffusional mass transfer at the expense of concentration gradient σС/σx.
Def = Dm/Г (2),
where Г – coefficient of distribution between solid and liquid phases. Coefficient of molecular diffusion is n*10-5 cm2/sec.
Using the formula (1) the maximum of unloaded ground waters in winter is taken as 5m3/24 hours in one linear meter. In the water reservoir of 111 km in length (with exception of Shoshinsky reach) the volume of unloaded ground waters accounts for 555*103m3/24 hours or 6.424 m3/sec in winter. Filtration takes place in a part of the area (15%) of Volzhsky and Ivankovsky reaches, but the major part of bottom has no filtration. The entire area of water reservoir is 327 km2. The average linear filtration velocity in a part (15%) of the water reservoir is estimated as v = (6,424 m3/с)/(32,25*106m2) = 2*10-4 cm/sec
Its average local velocity in the pore solution of bottom sediments (porosity = 0.85) is v’ = v/m = 4,45*10-4 cm/sec.
For a month (2.6*106 sec) the water is filtered in the volume of 5.18 m3 per one square meter. In the upper 10cm layer the volume of pore solution is 0.04 m3, that’s why 130 free volumes of pore water will be filtrated for a month at the maximum velocity (5 m3/24 hours).
When estimating the possible removal of sorbed microelements in three mobile forms it seemed feasible to use a sorption model. For this purpose the coefficient of distribution between solid and liquid phases was calculated, i.e. Г as a ratio of concentrations α and C, where α – concentration of microelement in solid phase and C – its concentration in the pore water. Using these data the coefficient of distribution is calculated as
Г = Csp/Cps (3),
where Csp is a summary concentration of mobile forms, Cps – concentration in pore solution. According to this model the water in volume of Г*V is required for desorption of pore volume V, i.e. 104 volumes of the pore space (Table 7). Thus, only an insignificant part of mobile forms seems desorbed and removed by filtration with maximum velocity in the area of 15% from the bottom.
Table 7 - Distribution coefficients of HM concentration between the solid phase and pore solution in all the studied reaches of the water reservoir
| Element | Volzhsky reach | Ivankovsky reach | Shoshinsky reach |
| Cr | 1,4*104 | 2,1*104 | 1,4*103 |
| Co | 3,2*104 | 5,7*104 | 2,4*103 |
| Ni | 2,4*104 | 1,4*104 | 2,7*103 |
| Zn | 5*103 | 1,2*104 | 1,1*104 |
| As | 1,1*104 | 2,6*104 | 1,3*103 |
| Cd | 1,4*104 | 104 | 1,4*103 |
| Pb | 2,7*104 | 3,1*104 | 1,6*103 |
| Cu | 2*104 | 2,3*104 | 1,1*103 |
It is evident that the microelements are sorbed by the solid phase of bottom sediments to a significant extent and distribution coefficients calculated by the sum of three mobile forms are varying insignificantly. Such data about mobile forms have been obtained for the first time. They allow specifying the possible diffusion-induced removal of heavy metals from bottom sediments.
Using the formula (2) and the data presented in Table 8 it is possible to calculate the effective coefficient of diffusion
Def ~ 10-9
Let’s calculate the diffusional flow of any element Q per 1 cm2 of the area using the diffusion coefficient 10-9cm2/sec for a month (2 592 000 sec). It will be equal to Q ≡ 2.6 10-3 ∆C/∆x mg/(cm2*month),
where ∆C – difference in concentrations between the layer near bottom and bottom sediments, mg/ml (maximal difference of concentrations in pore solution and the water near bottom is not more than 10-3 mg/cm3 for all the heavy metals, except Fe and Mn). ∆x is a specific size between measurements of the layer near bottom and bottom sediments calculated as 1.0 cm. The mass of component that can be removed by diffusion from the upper 10cm layer of bottom sediments proves to be smaller then the mass of heavy metals in this layer. There are no significant changes in the mass of HM mobile forms for a month.
Based upon the above data it is impossible to conclude that removal by diffusion and by filtration should be considered as real mechanisms of microelement removing into the water. The main reason for such removal is probably the effect of channel processes.
In Volzhsky reach Co, Pb, Fe, Mn mobile forms reveal an accumulation for a year, whilst the mass of Zn, Cr, Cu, Ni gets declined. In winter the mass of HM mobile forms is depleted, i.e. in the winter period the bottom sediments can serve as a source of the second water contamination in Volzhsky reach.
In Ivankovsky reach an annual accumulation of mobile forms is characteristic of Zn, Cu, Cr, Ni. The mass of Zn mobile forms is the greatest.
In Shoshinsky reach the mobile forms of such elements as Co, Fe, Cr, Pb are largely accumulated in bottom sediments but Zn, Mn, Ni, Cu, Cd display a decreased accumulation. In October-January the mass of mobile forms is intensively depleted in this reach.
As regards the mass of microelements in the pore solution it is rather high, however the assessment of its changes in all the reaches for the whole period of observations is of great importance.
The obtained results give evidence that the accumulated mass of heavy metals in pore solution is insignificantly changed as compared to changes in their mass in the solid phase. The pore solution plays a role of “migration” channel, in which the heavy metals reveal their transfer from the solid phase into the water and the bottom sediments. This transfer occurs at the expense of sedimentation and sorption by the upper later of the bottom.
Such elements as Zn, Cu, Cr, Ni, Cd are predominantly removed from bottom sediments into the water in the kind of dissolved compounds. The removal of Zn, Cr, Cu is the highest in running Volzhsky and Ivankovsky reaches.
Conclusions
- A new approach is offered to study in nature the mass exchange of heavy metals in the “water – bottom sediments” system, approbated in Ivankosky water reservoir as the basic source of water supply in Moscow.
- The total mass of heavy metals containing in the pore solution of the upper 10cm layer of bottom sediments is insignificant as compared to that accumulated in the solid phase.
- Investigation results obtained in points of observation showed that the concentration of the studied heavy metals in pore solutions exceeds that in the layer near bottom. The diffusional flow of major heavy metals occurs from bottom sediments into water layers near bottom in every season for a year. Moreover, the variation coefficient of their concentration in pore solutions is also higher than that in water near bottom. One should stress that the seasonal dynamics of heavy metals is not affected by water pH, pore solution and the content of dissolved oxygen.
- The study of three heavy metal mobile forms in the solid phase of bottom sediments (exchangeable, organic complexes and bound to Fe and Mn hydroxides) conducted in points of observation allowed determining a higher percentage of mobile forms as compared to the total content of heavt metals. It is shown that the composition of microelements closely connected with the solid phase is rather stable, the composition and ratio of mobile forms reveal great changes. The monthly measurements made it possible to establish that the mobile forms are highly redistributed. There exist regularities in seasonal changes of mobile forms for each studied microelement. With changing the content of heavy metals in bottom sediments the amount of mobile forms is also changed.
- The suspension current was estimated for the first time taking into account the distribution coefficient of microcomponents between the pore water and the solid phase of bottom sediments. It is worth of note that the mass of mobile forms accumulated in the solid phase is insignificant.
- Estimation of changes in the amount of mobile forms in the solid phase and in pore solution of bottom sediments in intervals between time of observations made it possible to identify the value and trend in resultant of two processes – accumulation of elements in bottom sediments and their removal. The balance for every microelement permitted to assess seasonal changes in resultant of its accumulation and removal for the whole period of observation (2001-2002).
References
- Varshal, G.M., Velyukhanova, T.K., I.M. Koshcheeva. Humic substances in the biosphere. Moscow, Nauka, 1993, 97 p.
- Venitsianov, Ye.V., Kocharyan, A.G., Safronova, N.S., Ye.P. Serenkaya. Seasonal changes in heavy metal forms in bottom sediments of Kuibyshev water reservoir // Water resources, 2003, No. 4, v.15, p. 443-451.
- Zakonnov, V.V. Spatial-temporary heterogeneity of distribution and accumulation of bottom sediments in the Upper Volga cascade // Water resources, 1995. No. 3, v. 22, p. 362-371.