СИНТЕЗ И СВОЙСТВА НОВЫХ N-АРИЛ-1,2,4-ТРИАЗОЛОВ
Аль-Саеди Раад Муслим Мухабес1, Викрищук Н.И.2, Душенко Г.А.3, Попов Л.Д.4, Белдовская А.Д.5, Михайлов И.Е.6
1Магистр 2-года обучения, Южный федеральный университет, химический факультет, Ростов-на-Дону, Россия, Высшее образование Ирака, Майсон университет, медицинский факультет, кафедра клинической биохимии, Ирак,
2Доцент, кандидат химических наук, Южный федеральный университет, химический факультет, Ростов-на-Дону, Россия,
3Главный научный сотрудник, доктор химических наук, Южный научный центр РАН, отдел химии Ростов-на-Дону, Россия,
4Ведущий инженер, кандидат химических наук, Южный федеральный университет, химический факультет, Ростов-на-Дону, Россия,
5Младший научный сотрудник отдела молекулярной биологии Института аридных зон ЮНЦ РАН, Ростов-на-Дону, Россия,
6Профессор, заведующий отделом химии Южного научного центра РАН, доктор химических наук, Ростов-на-Дону, Россия
СИНТЕЗ И СВОЙСТВА НОВЫХ N-АРИЛ-1,2,4-ТРИАЗОЛОВ
Аннотация
Рециклизацией перхлората 2-метил-4-оксо-1,3-бензоксазиния под действием арилгидразинов (п-гидразинобензойной кислоты и п-нитрофенилгидразина) синтезированы ранее неизвестные о-гидроксифенилзамещенные N-арил-1,2,4-триазолы и изучены их реакции метилирования и ацетилирования.
Ключевые слова: синтез, 1,2,4-триазол, рециклизация, производные.
Al saedi Raad Muslim Mhaibes1, Vikrishchuk N.I. 2, Dushenko G.А.3, Popov L.D.4, Beldovskaya A.D.5, Mikhailov I.Е.6
1Magistrant of the 2nd year education, Southern Federal University, Chemistry Faculty, Rostov-on-Don, Russia, High education of Iraq, Maysan University - Faculty of Medicine, Department of Clinical biochemistry, Iraq,
2Assistant professor, candidate of chemistry sciences, Southern Federal University, Chemistry Faculty, Rostov-on-Don, Russia,
3Principal research worker, doctor of chemistry sciences, Southern Scientific Center, Russian Academy of Sciences, Rostov-on-Don, Russia,
4Leading engineer, candidate of chemistry sciences, Southern Federal University, Chemistry Faculty, Rostov-on-Don, Russia,
5Junior research worker, Institute of Arid Zones, Southern Scientific Center, Russian Academy of Sciences, Rostov-on-Don, Russia,
6Professor, doctor of chemistry sciences, Southern Scientific Center, Russian Academy of Sciences, Southern Federal University, Rostov-on-Don, Russia
SYNTHESIS AND PROPERTIES OF NOVEL N-ARYL-1,2,4-TRIAZOLES
Abstract
By recyclization of 2-methyl-4-oxa-1,3-benzoxazinium perchlorate under action of arylhydrazines of p-hydrazinobenzoic acid and p-nitrophenylhydrazine novel o-hydroxyphenyl-substituted N-aryl-1,2,4-triazoles were synthesized. Their reactions of methylation and acylation were studied.
Keywords: recyclization of 2-methyl-4-oxa-1,3-benzoxazinium perchlorate, arylhydrazines, methyl and acyl derivatives of 1,2,4-triazoles.
Increasing interest of chemists to derivatives of 1,2,4-triazoles can be explained first of all by high biological activity of this class compounds. Among 1,2,4-triazoles have been found anticonvulsants [1], compounds with antifungous [2], antibacterial [3] and antiviral [4] effects. Besides derivatives of 1,2,4-triazoles reveal other interesting properties, so triphenyltriazoles are described in literature as effective organic luminophors. They are also applied as chemosensors [5]. o-Hydroxyphenyltriazoles containing chelating coordinative =N-(O-phenol) center [6-8] can form stable metal complexes possessing useful properties for practice [9-15].
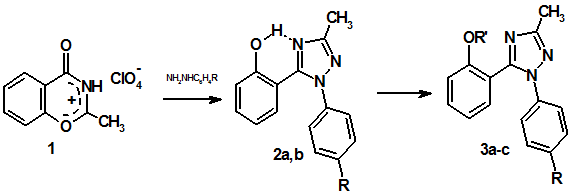
With aim of widening of range of similar compounds novel 1,2,4-triazoles have been obtained by us according to the following scheme:
Hydroxyphenyl substituted triazoles 2 were synthesized by recyclization of perchlorate 1 under action of arylhydrazines of p-hydrazinobenzoic acid and p-nitrophenylhydrazine in ice acetic acid. Alkylation of triazoles 2 with methyl iodide in the presence of silver oxide under refluxing in chloroform gave methoxy derivatives 3а,с, at that in the case of triazolbenzoic acid 2a methylation of phenol hydroxy group were accompanied by reaction of carboxyl group and led to the ester 3а. Acetates 3b,d were synthesized by reaction of triazoles 2 with acetic anhydride in the presence of 70% perchloric acid.
Structure of the newly obtained compounds was determined by element analysis, IR and NMR 1Н spectroscopy.
In the IR spectra of compounds 2,3 the stretching vibration bands of С=N and С=С were observed at 1460-1684 cm–1, at that for hydroxyphenyl substituted triazoles 2 also the vibration bands of OH groups were found at 3063 and 3083 cm–1. In the IR spectra of acetates 3b,d intensive bands were appeared at 1754 and 1767 cm–1 (COCH3). In the NMR 1H spectra of 2,3 in CDCl3 signals of protons of methyl group at triazole cycle are at 2.05-2.48 ppm and signals of aromatic protons – at 6.82-8.29 ppm.
EXPERIMENTAL
IR spectra of compound 2,3 were recorded with Varian Excalibur 3100 FT-IR spectrometer in thin layer. 1Н NMR (250.13 MHz) spectra in CDCl3 were registered with Bruker DPX-250 spectrometer at 22оС. The chloroform signals (δ H 7.27 ppm and δ С 77.00 ppm) were used as internal standard.
2-Methyl-4-oxa-1,3-benzoxazinium perchlorate 1 were synthesized as described in [16].
4-[5-(2-hydroxyphenyl)-3-methyl-1,2,4-triazol-1-yl]benzoic acid 2а. Mixture of 3.2 g of perchlorate 1 and 3.15 g of p-hydrazinobenzoic acid in 13 ml of ice acetic acid was refluxed in during 3 min, then was cooled down and left for 10 h. at room temperature. Then 80 ml of water was added. The precipitate was filtered off. Yield 65%, brown crystals, mp 261-263°C (from butan-1-ol). IR spectrum, ν, cm–1: 1595, 1630 (C=Car, С=N), 1698 (С=О). NMR 1Н spectrum (CDCl3) δ, ppm: 2.47 s (3Н, CH3), 6.82-7.49 m (6Н, Нarom), 8.04 d (2Н, Нarom, 3J 7.5 Hz). Calculated, %: C 65.04; H 4.40; N 14.23. С16H13N3O3. Found, %: C 65.30; H 4.30; N 13.90.
2-[5-methyl-2-(4-nitrophenyl)-1,2,4-triazol-3-yl]phenol 2b was obtained analogously to compound 2а. Yield 58%, brown crystals, mp 170-172°C (from propan-2-ol). IR spectrum, ν, cm–1: 1514 (С=С), 1594, 1613 (C=N), 3083 (ОH). NMR 1Н spectrum (CDCl3) δ, ppm: 2.48 s (3Н, CH3), 6.88-7.64 m (6Н, Нarom), 8.29 d (2Н, Нarom, 3J 10 Hz), 9.95 s (1Н, OH). Calculated, %: C 60.71; H 4.05; N 18.91. С15H12N4O3. Found, % : C 60.10; H 4.00; N 18.30.
Мethyl 4-[5-(2-methoxyphenyl)-3-methyl-1,2,4-triazol-1-yl]benzoate 3а. Mixture of 0.8 g of triazole 2а, 0.56 g of silver oxide and 0.4 ml of methyl iodide was refluxed in 10 ml of chloroform for 3 h., then cooled down. The precipitate was filtered off. The obtained filtrate was passed through a column with aluminum oxide (eluent – chloroform). The residue prepared after concentration by evaporation was recrystallized from petroleum ether. Yield 60%, colorless crystals, mp 115-117°C. IR spectrum, ν, cm–1: 1460, 1518 (С=С), 1582, 1606, 1649 (C=N), 1716 (C=О). NMR 1Н spectrum (CDCl3) δ, ppm: 2.46 s (3Н, CH3), 3.34 s (3Н, CH3), 3.91 s (3Н, ОCH3), 6.91 d (1Н, Нarom, 3J 7.5 Hz), 7.12 t (1Н, Нarom), 7.35 d (2Н, Нarom, 3J 10 Hz), 7.45-7.59 m (2Н, Нarom), 7.98 d (2Н, Нarom, 3J 10 Hz). Calculated, %: C 66.8; H 5.26; N 13.01 С18H17N3O3 Found, %: C 66.70; H 5.10; N 12.50.
4-[5-(2-acetoxyphenyl)-3-methyl-1H-1,2,4-triazol-1-yl]benzoic acid 3b. To a mixture of 0.5 g of triazole 2а and 0.67 ml of acetic anhydride 0.04 ml of 70% HClO4 was dropped. The mixture was left for 10 h. at room temperature. Then 60 ml of water was added. The precipitate was filtered off. Yield 57%, yellow crystals, mp 190-192°C (from toluene). IR spectrum, ν, cm–1: 1448, 1474, 1525 (С=С), 1606, 1647 (C=N), 1707, 1754 (C=О). NMR 1Н spectrum (CDCl3) δ, ppm: 2.05 s (3Н, CH3), 2.52 s (3Н, CH3), 7.20-7.48 m (6Н, Нarom), 7.04 d (2Н, Нarom, 3J 10 Hz). Calculated, %: C 64.05; H 4.44; N 12.46. С18H15N3O4. Found, %: C 63.50; H 4.10; N 12.10.
5-(2-methoxyphenyl)-3-methyl-1-(4-nitrophenyl)-1,2,4-triazole 3с was obtained analogously to compound 3а. Yield 60%, yellow crystals, mp 153-155оC (from propan-2-ol). IR spectrum, ν, cm–1: 1472, 1516 (С=С), 1596, 1613 (C=N). NMR 1Н spectrum (CDCl3) δ, ppm: 2.48 s (3Н, CH3), 3.39 s (3Н, ОCH3), 6.96 d (1Н, Нarom, 3J 7.5 Hz), 7.14 t (1Н, Нarom), 7.50 d (2Н, Нarom, 3J 10 Hz), 7.48-7.61 m (4Н, Нarom), 8.23 d (2Н, Нarom, 3J 7.5 Hz). Calculated, %: C 61.90; H 4.51; N 18.06. С16H14N4O3. Found, %: C 61.10; H 4.11; N 17.80.
[2-[5-methyl-2-(4-nitrophenyl)-1,2,4-triazol-3-yl]phenyl] acetate 3d was obtained analogously to compound 3b. Yield 55%, yellow crystals, mp 140-142оC (from toluene). IR spectrum, ν, cm–1: 1477, 1506, 1518 (С=С), 1595, 1618, 1684 (C=N), 1767 (C=О). NMR 1Н spectrum (CDCl3) δ, ppm: 2.05 s (3Н, CH3), 2.48 s (3Н, CH3), 7.20-7.57 m (6Н, Нarom), 8.04 d (2Н, Нarom, 3J 10 Hz). Calculated, %: C 60.70; H 4.05; N 18.91. С17H14N4O4. Found, %: C 60.11; H 3.95; N 18.21.
References
- Almasirad A, Tabatabai S, Faizi M, Kebriaeezadeh A, Mehrabi N, Dalvandi A, et al. Synthesis and anticonvulsant activity of new 2-substituted-5-[2-(2-fluorophenoxy)phenyl]-1,3,4-oxadiazoles and 1,2,4-triazoles // Bioorganic & medicinal chemistry letters. 2004. Vol. 14(24). P. 6057-6059.
- Dan Z, Zhang J, Yu S, Hu H, Chai X, Sun Q, et al. Design and synthesis of novel triazole antifungal derivatives based on the active site of fungal lanosterol 14a-demethylase (CYP51) // Chinese Chemical Letters. 2009. Vol. 20(8). P. 935-938.
- Bayrak H, Demirbas A, Demirbas N, Karaoglu S. Synthesis of some new 1,2,4-triazoles starting from isonicotinic acid hydrazide and evaluation of their antimicrobial activities // European journal of medicinal chemistry. 2009. Vol. 44(11). P. 4362-4366.
- Al-Soud Y, Al-Dweri M, Al-Masoudi N. Synthesis, antitumor and antiviral properties of some 1, 2, 4- triazole derivatives // Il Farmaco. 2004. Vol. 59(10). P. 775-783.
- Arduini M., Fellunga F., Mancin F., Rossi P., Tecilla P., Tonellato U., Valentinuzzi N. Aluminium fluorescence detection with a FRET amplified chemosensor // Chem. Commun. 2003. N 13. P. 1606-1607.
- Shasheva E.Yu., Vikrishchuk N.I., Popov L.D., Vikrishchuk A.D., Mikhailov I.E., Lysenko K.A., Kletskii M.E. Reactions of hydroxyphenyl-substituted 1,2,4-triazoles with electrophilic reagents // Zhurnal Obshchei Khimii. 2009. Vol. 79. N 10. P. 1739-1748 (Russian Journal of General Chemistry. 2009. Vol. 79. N 10. P. 2234-2243).
- Shasheva E.Yu., Vikrishchuk N.I., Popov L.D., Minkin V.I., Kletskii M.E., Antipin M.Yu., Vikrishchuk A.D., Mikhailov I.E. Synthesis and properties of substituted [3-(2-hydroxyphenyl)-1H-1,2,4-triazol-1-yl]acetonitriles // Zhurnal Organicheskoi Khimii. 2010. Vol. 46. N 7. P. 1080-1085 (Russian Journal of Organic Chemistry 2010. Vol. 46. N 7. P. 1079-1084).
- Vikrishchuk A.D., Popov L.D., Vikrishchuk N.I., Morozov P.G., Mikhailov I.E. Synthesis of 1-(benzothiazol-2-yl)-5-(o-hydroxyphenyl)-1H-1,2,4-triazoles // Zhurnal Obshchei Khimii. 2013. Vol. 81. N 1. P. 135-137 (Russian Journal of General Chemistry. 2011. Vol. 81. N 1. P. 132-134).
- Ryabukhin Yu.I., Shibaeva N.V., Kuzharov A.S., Korobkova V.G., Khokhlov A.V. Garnovskii A.D. Synthesis and investigation of complex compounds of transition metals with di-(o-oxyphenyl)-1,2,4-oxadiazole and 1,2,4-triazoles // Koordinatsionnaya Khimiya. 1987. Vol 13. P. 869-874 (Sov. J. Coord. Chem. (Engl. Trans.). 1987. Vol. 13. P. 493-499)
- Antsyshkina A.S., Porai-Koshits M.A., Garnovskii D.A., Faleeva L.N., Ryabukhin Yu.I., Garnovskii A.D. Synthesis and crystal structure of solvate of zinc (II) {2-[5-(1,3-diphenyl-1,2,4-triazolyl)]phenolate} with methanol // Koordinatsionnaya Khimiya. 1990. Vol 16. H. 1628-1632 (Sov. J. Coord. Chem. (Engl. Trans.). 1990. Vol. 16. P. 876-880)
- Hage R., Haasnoot J.G., Wang R., Ryan E.M., Vos J.G., Spec A.L., Duisenberg J.M. Synthesis, Spectroscopic and Electrochemical Properties and X-ray Studies of Bis(2,2-bipyridil)(3-(2-hydroxy-phenyl)-5-(pyridine-2-yl)-1,2,4-triazole)-ruthenium(II)hexafluorphosphate // Inorg. Chim. Acta. 1990. Vol. 174. P. 77-85.
- 12. Steinhauser S., Heinz U., Bartholoma M., Weyhermuller T., Nick H., Hegetschweiler K. Complex Formation of ICL670 and Related Ligands with FeIII and FeII // Eur. J. Inorg. 2004. N 22. P. 4177-4191.
- Keyes T.E., Evrard B., Vos. J.G., Johannes G., Brady C., McGarvey J.J., Jayaweera P. Electronic and photophysical properties of novel phenol bound dinuclear ruthenium complex: evidence for a luminescent mixed valence state // Dalton Trans. 2004. Vol. 15. P. 2341-2346.
- Dushenko G.A., Vikrishchuk A.D., Mikhailov I.E., Vikrishchuk N.I. Quantum chemical investigation of structure and conformational non-rigidity of 5-(2'-hydroxyphenyl)-3-methyl-1-benzthiazolyl-1,2,4-triazole // Vestn. Yuzhn. Nauch. Tsentra. 2013. Vol. 9. No. 1. P. 37-41.
- Beldovskaya A.D., Dushenko G.А., Vikrishchuk N.I., Popov L.D., Revinskii Yu.V., Mikhailov I.Е. Synthesis, structure, and spectral luminescent properties of novel 1,2,4-triazole derivatives containing benzthiazole group // Zhurnal Obshchei Khimii. 2013. Vol. 83. N 11. P. 1882–1891 (Russian Journal of General Chemistry. 2013. Vol. 83. N 11. P. 2075–2083).
- Ryabukhin Yu.I., Mezheritskii V.V., Dorofeenko G.N. Method of preparation of 4H-1,3-benzoxazin-4-onium salts // Zhurnal Obshchei Khimii. 1974. Vol. 44. N 12. P. 2792-2793.
Acknowledgements- Al saedi Raad Muslim Mhaibes is grateful to the High education of Iraq for sending him to study in Russia.