ХИМИЧЕСКАЯ МОДИФИКАЦИЯ АМИНОПОЛИСАХАРИДА ИЗ ПАНЦИРЯ КРЕВЕТОК ПРЯМЫМ СОЕДИНЕНИЕМ С ПОЛИАНИЛИНОМ
ХИМИЧЕСКАЯ МОДИФИКАЦИЯ АМИНОПОЛИСАХАРИДА ИЗ ПАНЦИРЯ КРЕВЕТОК ПРЯМЫМ СОЕДИНЕНИЕМ С ПОЛИАНИЛИНОМ
Научная статья
Нгуен Тхи Линь1, *, Нго Tхань Хай2, Ву Ван Тоан3, Ле Динь Чиен4
1 ,2, 3 Ханойский университет горного дела и геологии, Вьетнам, Ханой;
4 Вьетнамская государственная нефтегазовая компания, Вьетнам, Ханой
* Корреспондирующий автор (nguyenthilinh[at]humg.edu.vn)
АннотацияАминополисахарид был модифицирован деацетилированием хитина из панциря креветок с использованием фермента в качестве биокатализатора и микроволновой энергии. Модифицированный аминополисахарид имеет степень деацетилирования на 83 выше, чем при использовании обычного химического метода. Аминополисахарид был успешно соединен по свободно-радикальному механизму с инициатором персульфата аммония. Реакцию проводили в водной среде. Влияние температуры реакции, времени реакции, массового соотношения аминополисахарида к анилину было исследовано. Полученные результаты показали, что наилучшими условиями для соединения аминополисахарида были следующие: массовое соотношение аминополисахарида к анилину составляло 1:2, температура реакции - 5°С и время реакции 60 минут. Модифицированный аминополисахарид характеризован Фурье-спектроскопией и сканирующей электронной микроскопией. Степень соединения модифицированного аминополисахарида получена 41%.
Ключевые слова: хитин, аминополисахарид, прямое соединение, модифицированный аминополисахарид; Степень деацетилирования, степень соединения; полианилин.
CHEMICAL MODIFICATION OF AMINOPOLYSACCHARIDE FROM SHRIMP SHELL BY DIRECT GRAFTING WITH POLYANILINE
Research article
Nguyen Thi Linh1, *, Ngo Thanh Hai2, Vu Van Toan3, Le Dinh Chien4
1, 2, 3 Hanoi University of Mining and Geology, Hanoi, Vietnam;
4 Vietnam Oil and Gas Group, Hanoi, Vietnam
* Corresponding author (nguyenthilinh[at]humg.edu.vn)
AbstractAminopolysaccharide was modified by deacetylation of chitin from shrimp shell using enzyme as a biocatalyst and microwave energy. Modified aminopolysaccharide has degree of deacetylation of 83 higher than that of using normal chemical method. Aminopolysaccharide was successfully grafted with aniline by free radical mechanism with ammonium persulfate initiator. The reaction was carried out in aqueous medium. The effect of reaction temperature, reaction time, aminopolysaccharide: aniline weight ratio were investigated. The results showed that the best conditions for grafting of aminopolysaccharide were aminopolysaccharide: aniline weight ratio of 1:2, reaction temperature of 5oC and reaction time of 60 minutes. The modified aminopolysaccharide was characterized by Fourier transform infrared spectroscopy and Scanning electron microscopy. Degree of Grafting of modified aminopolysaccharide obtained 41%.
Keywords: Chitin, aminopolysaccharide, direct grafting, modified aminopolysaccharide; Degree of Deacetylation, Degree of Grafting; polyaniline.
- Introduction
Chitin is valuable material and is known to be the natural polysaccharide. Chitin can be extracted from the crust of marine invertebrates (prawn shells, lobster shells, scrab cuttlefish…), mollust, insects, fungi but mainly from crab and shrimp shells of waste byproducts from the seafood processing industries to protect environment [1], [2]. Chitin is a heteropolymer made up of β (1 → 4)-2-acetamido-2-deoxy-β-D-glucopyranose units with acetamide group at C2 positions can attend in the deacetylation reaction to form aminopolysaccharide (chitosan) [3], [4]. Chitosan has found application in many fields, including food processing, medicine, and wastewater treatment.
Aminopolysaccharide is modified from chitin shell that called natural aminopolysaccharide containing β(1,4)-D glucosamine units and N-acetyl- D-glucosamine units. Aminopolysaccharide and its derivatives are non-toxic, degradable biopolymers [8], [9]. Beside all these value properties, aminopolysaccharide has remained underutilized biopolymer primarily due to its coilling in liquid medium result in the functional groups is counteracted [10].
The chemistry of aminopolysaccharide is interesting due to the presence of hydroxyl groups at C3 and C6 and amine group at C2 position [11], [12]. Recently, there has been great interest in chemical modification of aminopolysaccharide to prevent its coilling in order to improve its linear flexible chains and widen its application in environmental and biomedical fields [13], [14].
Chemically modified aminopolysaccharide was carried using pyridine, grafting with poystyrene, glycidyl methacrylate, indirect grafting with aniline, etc., to make biosensors, adsorbents for wastewater treatment. However, the grafting of aminopolysaccharide was carried indirectly through organic bridges.
In the present work, we report for the first time the chemical modification of aminopolysaccharide by direct grafting with polyaniline using ammonium persulfate as an initiator.
- Materials and methods
2.1. Materials
Shrimp shell was collected from refrigerated shrimp production of Minh Hai company (Vietnam), ammonium persulfate, enzyme papain were purchared from Sigma-Aldrich, sodium hydroxide, hydro chloride, ethanol, acetic acid were purchared from Xilong chemicals company (China), aniline from Merck.
2.2. Extraction and conversion of chitin for aminopolysaccharide
Shimp shell was treated with 10% HCl solution (solid/liquid ratio 1/10) at ambient temperature in 12h until gas bubble disappears (heating method) or placed in microwave oven at 500W in 8 minutes (microwave method) and then the shimp shell must be washed with demineral water to neutral state (pH=7) to remove minerals.
The demineralized shells were immersed in papain enzyme/phosphate 7 buffer solution at 60oC for 30 minutes to remove protein. The sample was finally washed with distilled water and then dried in a vacuum oven at 60oC to constant weight. Deproteinated shell has light pink what decolorizated by exposing in the natural light to remove pigments. After that all, the product of extraction is extracted chitin.
Chemical deacetylation was carried out by treatment of extracted chitin with 50% sodium hydroxide (NaOH) at 90oC in 4.5h using heating method or using microwave method in 8 minutes at 350W. Deacetylated product has opalesque white, is aminopolysaccharide.
2.3. Synthesis of aminopoysaccharide grafting polyaniline
In a typical procedure, an aqueous solution of aminopolysaccharide (0.3g) was prepared by dissolving aminopolysaccharide in 150mL of aqueous acetic acid 1% for 10 minutes with stirring of 600 rpm. Adding 5.63 mmol aniline in hydrochloride 1M to form a homogenous solution. The homogenous solution fed into aminopolysaccharide solution.
Ammonium persulfate was added to the above and interact with the aminopolysaccharide solution for 30 min before adding the monomer. Copolymers of soluble aminopolysaccharide and aniline were prepared using different parameters namely aminopolysaccharide:monomer weight ratio, temperature, reaction time, with constant stirring at 600 rpm. While the reaction was transferring from emeraldine color to dark green, ethanol was poured into the reaction to precipitate the crude grafted polymer. The precipitate was filtered using sintered glass funnel, dried under vacuum at 40◦C as powdery material until a constant weight was achieved. The crude graft polymer was washed with aniline (Soxhlet extraction) for 24 h to remove any polyaniline present. The extracts were filtered using sintered funnel, washed with ethanol to rinse the copolymer. The clean graft copolymers were dried to a constant weight in a vacuum oven at 60◦C for 24h. The direct grafting of aminopolysaccharide by aniline was expressed in scheme 1.
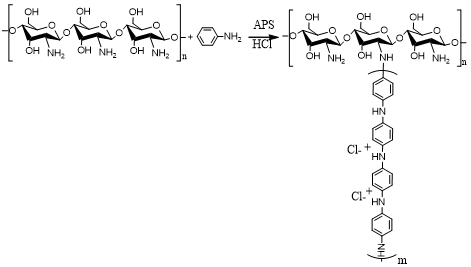
Fig. 1 – Direct grafting of aminopolysaccharide by aniline
2.4. Characterization techniques
Fourier transform infrared (FTIR) spectra was recorded between 400- 4000 cm-1 with a 4 cm-1 resolution from KBr pallets on a JASCO FT/IR- 4600 spectrometer to define the molecular structure of parent aminopolysaccharide and grafted aminopolysaccharide. The morphologies of grafted polymer were determined by scanning electron microcopy (SEM) working at 5 kV.
FTIR spectroscopy was also determined degree of deacetylation (DD) of parent aminopolysaccharide and grafting degree (GD) of modified aminopolysaccharide. The DD of the aminopolysaccharide samples was calculated base on the absorbances at about 1650cm-1 and 3450 cm-1 according to the follow equation [19]:
DD (%) = 100 – [(A1650/A3540)×100/1.33]
Where A1650 and A3450 are absorbance at 1650 cm-1 of the amide I of the N-acetyl group and 3450 cm-1 of amine group in aminopolysaccharide.
The DG is the percentage of grafting of aminopolysaccharide by aniline. The DG was suggested by calculating the DD of parent aminopolysaccharide and the DD of modified aminopolysaccharide by grafting by follow equation:
DG(%) = DDparent – DDmodified
Where DDparent and DDmodified are the DD of parent aminopolysaccharide and the DD of modified aminopolysaccharide by grafting.
3. Results and discussion3.1. Characterisation of chitin from shrimp shell
Chitin was extracted from shrimp shell using enzyme papain as a catalyst to remove protein, chemical method and microwave were used for the elimination of mineral salts of shrimp shell. FT-IR spectra of chitin are shown in Fig. 1.
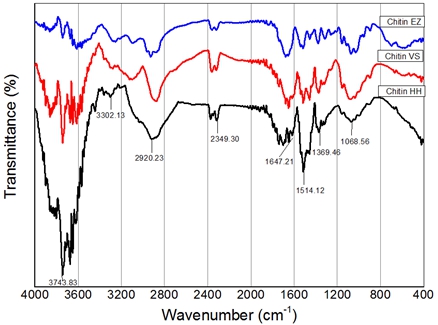
Fig. 2 – FT-IR of chitin from shimp shell using chemical method (Chitin HH), microwave method (Chitin VS) and enzyme (Chitin EZ)
FTIR spectra of chitin from shrimp shell using chemical method (Chitin HH), microwave method (Chitin VS) and enzyme (Chitin EZ) shows that bands characteristic for three chitin samples are similar. Broad band at 3302 cm-1 ascribe H- bonded of NH stretching vibration. Bands at 2920 cm-1 and 2349 cm-1 refer to CH stretch in acetyl amino of chitin. The amide I brand is split at about 1647 cm-1 with low absorption. This result illustrated that extracted chitin structure contains a little amide I. The absorbance at 1514 cm-1 is due to protein is not present in chitin after deproteination by enzyme in 30 minutes while the band is present if deproteination is carried out using normal chemical method with 40% NaOH in 6h. The brand at 1369 cm‑1 represent symmetric streching vibration of CH3 of acetyl amino group of chitin. The brand at 1068 cm-1 assign to C-O-C streching vibration. The absence of brand around 870 cm-1 indicates that mineral has been removed.
3.2. Characterisation of aminopolysaccharide
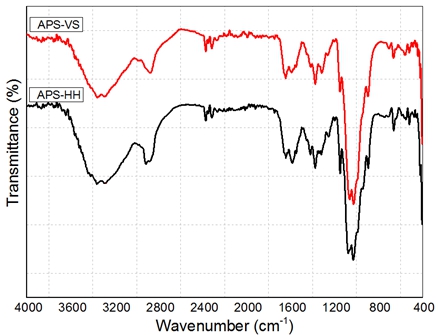 Fig. 2 – FTIR of aminopolysaccharide from chitin using normal method (APS-HH) and microwave method (APS-VS)
Fig. 2 – FTIR of aminopolysaccharide from chitin using normal method (APS-HH) and microwave method (APS-VS)
The FTIR spectrum of aminopolysaccharide Fig. 2 shows that the characteristic absorption bands at 3340 cm-1 – 3400 cm-1 and 2800- 2900 cm-1 corresponding to OH, NH stresching regions of aminopolysaccharide. The brand at about 1600 cm-1 was assigned to amide I of aminopolysaccharide. The disappearence of amide II of chitin shown that deacetylation of chitin was occurred to convert amide I into amine group.
FTIR spectroscopy was also used to define the degree of deacetylation of aminopolysaccharide (DD). The DD of the aminopolysaccharide samples was calculated from the absorbances at 1658 cm-1 and 3450 cm-1. Degree of deacetylation of normal method aminopolysaccharide (APS-HH) is 78 while by using microwave the DD of APS is 83.

Fig 3 – Image SEM of aminopolysaccharide using microwave (APS-VS) (a) and normal chemical methods (APS-HH) (b)
The result of SEM images showed that aminopolysaccharide is very uiniform with a lamellar organization and crystical structure that converted under microwave condition beside the sample of chemical method with dense block appears. The lamellar structure of APS-VS was suggested for modification of aminopolysaccharide by direct grafting with aniline.
3.3. Characterisation of polyaniline grafted aminopolysaccharide
3.3.1. FTIR analysis
The grafting of APS with aniline was carried out with present of ammonium persulfate such as an initiator. The grafting is favored with free redical mechanism in here ammonipersulfate can be decomposed thermally under suitable temperature to form pair of initiating radicals. The radicals abstract the hydrogen atom attached to the amine group of aminopolysaccharide molecule to form macroradical thereby generating an active center that could be used to graft aniline onto the backbone [16], [20]. Aminopolysaccharide macroradical were treated with aniline to initiate graft copolymerization by free radical mechanism. The macroradical attacked the amine group of the aniline and generate radicals onto it which resulted to repeate propagation processes. The resulting graft copolymer was precipitated by pouring ethanol and finally the precipitate was washed with aniline by Shoxhlet extraction to remove any free poyaniline.
The preparation of graft copolymers was carried out with different degree of grafting (DG) by considering the reaction parameters.
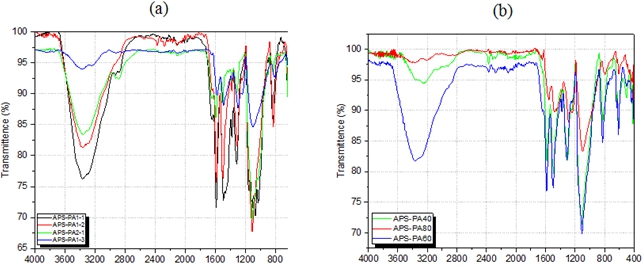
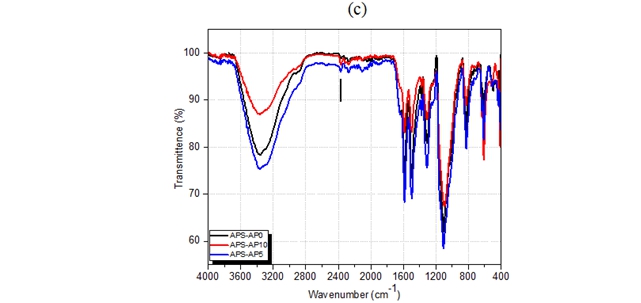
Fig. 4 – FTIR of aminopolysaccharide grafting polyaniline in different conditions: (a) APS-PA synthesized
in different APS:PA ratios, (b) different reaction times, (c) different temperatures
The FTIR spectra of aminopolysaccharide grafting polyaniline samples with different ratio of APS and aniline are shown in Fig.4. It can be seen from the FTIR spectra that the grafted polymers have characteristic bands of both aminopolysaccharide and substituted polyaniline. The absorption brand at 3380 cm-1 with different transmittence belonging to -NH2 group of aminopolysaccharide. The diffence of -NH2 group transmittence due to this group attend to grafting to form bond with polyaniline. The band around 1641 cm-1 are ascribed to the -NH2 group bending vibration. New absorption bands found in the APS-PA at 1550 cm-1, 1370 cm-1, 807 cm-1 are characteristic of the nitrogen quinone structure in the polyaniline bands, phenyl bands, aromatic C-H bending with a sharp vibration in the aromatic ring structure containing parasubstitutes of polyaniline bands, respectively. The results of investigation shown that the best condition for synthesis of polyaniline grafted aminopolysaccharides were APS:PA ratio of 1:2, reaction time of 60 minutes and temperature of 5oC. Moreover, the band at 1641 cm-1 has changed to lower wavenumber and the new bands are characteristics of the polyaniline side chains on the backbond of aminopolysaccharide. This result indicates interaction between polyaniline and aminopolysaccharide.
The reaction time was determined in the range of 40-80 minutes. The reaction was stopped by allowing air into the reaction vessel and then the reaction mixture was poured into ethanol. It was observed that the reaction time of 60 minutes yielded the highest DG (41%) in aminopolysaccharide-g-PA copolymer as shown in Table 1. Longer reaction time would result to homopolymerization due to chain transfer and termination of the growing radicals. Since the grafting was manifested only on the surface of the trunk polymer granules as such, this has effect on the rate of graft copolymerization.
Reaction temperature has great role on copolymerization. The results obtained in Table 1 showed that graft copolymerization was favoured at temperature of 5◦C; as percentage grafting of 41%. Higher temperature promotes decomposition of the initiator, which could lead to creating more active sites (radicals) on the backbone of the chitin molecule (Scheme 1). At temperature above 5◦C, the percentage grafting was reduced. This could be a result of the increase in conversion of initiator to free radicals which would promote termination of the growing radicals and could result to low molecular weight copolymer.
Grafting degree of aminopolysaccharide with polyaniline at various synthesis conditions is shown in Table 1.
Table 1 – Grafting degree of the APS-PA samples
| Samples | Synthesis conditions | DDmodified (%) | DG (%) | ||
| Ratio APS:PA (w/w) | Reaction temperature (0oC) | Reaction time (min) | |||
| APS-PAopt | 1:2 | 5 | 60 | 42 | 41 |
| APS-PA1-1 | 1:1 | 5 | 60 | 62 | 21 |
| APS-PA 1-3 | 1:3 | 5 | 60 | 56 | 27 |
| APS-PA 40 | 1:2 | 5 | 40 | 45 | 38 |
| APS-PA 80 | 1:2 | 5 | 80 | 62 | 21 |
| APS-PA0 | 1:2 | 0 | 60 | 58 | 25 |
| APS-PA10 | 1:2 | 10 | 60 | 54 | 21 |

Fig. 5 – SEM images of polyaniline grafted aminopolysaccharide samples in different conditions
Fig 5. shows scanning electron micrographs of APS-PA samples that were synthesized under different conditions. The morphology of the samples are compared at magnification of 30 000 times. The APS/PA1-1, APS/PA1-3, APS-PA40, APS/PA-80, APS/PA-10,APS/PA0 have ununiform structure with the arrange randomly. At the ratio of aminopolysaccharide and aniline is lower than ratio of ½ resulting in the grafting occurred uncompletely. On the contrary, at the high ratio, long reaction time, the appearance of exessed polyaniline covered on the aminopolysaccharide surface.
The mophology of APS-PAopt has fibers to make porous structure. From the images, it can be deduced that APS-PAopt has more homogenous structure than others.
- Conclusions
Aminopolysaccharide was converted from chitin of shimp shell by using enzyme and microwave energy in order to reduce used chemicals and energy. The FTIR of aminopolysaccharide confirmed that conversion of amide group in chitin into amine group in aminopoysaccharide. The aminopolysaccharide using microwave obtained degree of deacetylation of 83%. Grafted polyaniline on aminoplysaccharide copolymer was synthesized by chemical oxidation under free radical mechanism. The grafting aminopolysaccharide was synthesized under optimal condition with APS/PA ratio of ½, reaction time of 60 minutes, reaction temperature of 5oC. APS-PAopt sample carries polyanilne bands on the side chains due to grafting of aniline. The samples have porous structure and uniform that are suitable candidates for applications in environmental treatment.
| Благодарности Авторы выражают благодарность Ханойскому горно-геологическому университету а также Министерству образования и обучения Вьетнама (MOET) за финансовую поддержку. | Acknowledgement The authors are grateful to Hanoi University of Mining and Geology and Vietnam Ministry of Education and Traning (MOET) for financial support. |
| Конфликт интересов Не указан. | Conflict of Interest None declared. |
Список литературы / References
- Musarrat H. Extraction of chitin from prawn shells and conversion to low molecular masschitosan / Musarrat H. Mohammed A, Peter A. Williams A, Tverezovskaya O.. // Food Hydrocolloids, 31, 2013.- P 166-171.
- Sagheer F.A. Al. Extraction and characterization of chitin and chitosan from marine sourcesin Arabian Gulf / Sagheer F.A. Al, M.A. Al-Sughayer, S. Muslim, M.Z. Elsabee. // Carbohydrate Polymers, 77, 2009. – P. 410–419.
- Guilherme L. Surface modification of chitin using ultrasound-assisted and supercritical CO2 technologies for cobalt adsorption / Guilherme L. Dotto, Jeanine M. Cunha, Camila O. Calgaro, Eduardo H. Tanabe, Daniel A. Bertuol. // Journal of Hazardous Materials, 295, – P. 29–36.
- Muryeti, Estuti Budi Mulyani Adsorption of Carbon Black Using Chitosan in the Deinking / Muryeti, Estuti Budi Mulyani, Elya Sinurat. // ProcessProcedia Chemistry, 17, 2015. – P.106 – 110.
- Rathinam Karthik. Removal of Pb(II) and Cd(II) ions from aqueous solution using polyaniline grafted chitosan / Rathinam Karthik, Sankaran Meenakshi. // Chemical Engineering Journal, 263, 2015. – P. 168–177.
- Ayse Gul Yavuz A comparison study of adsorption of Cr(VI) from aqueous solutions ontoalkyl-substituted polyaniline/chitosan composites / Ayse Gul Yavuz, Elif Dincturk-Atalay, Aysegul Uygun, Fethiye Gode, Erhan Aslan. // Desalination, 279, 2011. – P. 325–331
- Thirugnanasambandham K. Application of chitosan as an adsorbent to treat rice mill wastewater—Mechanism, modelling and optimization / Thirugnanasambandham K., V. Sivakumar, J. Prakash Maran. // Carbohydrate Polymers, 97, 2013. – P. 451–457.
- Álvaro J. Leite Synthesis and characterization of bioactive biodegradable chitosan composite spheres with shape memory capability / Álvaro J. Leite, Sofia G. Caridade, João F. Mano. // Journal of Non-Crystalline Solids, 2015.
- Gre´ gorio Crini Application of chitosan, a natural aminopolysaccharide, for dye removal from aqueous solutions by adsorption processes using batch studies: A review of recent literature / Gre´ gorio Crini, Pierre-Marie Badot. // Polym. Sci. 33, 2008. – P. 399–447.
- Odile Carrier, Rudy Covis Inverse emulsions stabilized by a hydrophobically modified polysaccharide / Odile Carrier, Rudy Covis, Emmanuelle Marie, Alain Durand // Carbohydrate Polymers, 84, 2011. – P. 599–604.
- Junhua Wang Recent progress on synthesis, property and application of modified chitosan: An overview / Junhua Wang, Li Wang, Haojie Yu, Zain-ul-Abdin, Yongsheng Chen, Qing Chen, Weidong Zhou, Hongtao Zhang, Xiao Chen. // International Journal of Biological Macromolecules, 88, 2016. – P. 333–344.
- Jian-Ya Qian Antioxidation and a-glucosidase inhibitory activities of barley polysaccharides modified with sulfation / Jian-Ya Qian, Ye-Yu Bai, Jing Tang, Wei Chen // LWT - Food Science and Technology, 64, 2015. – P. 104-111.
- Xuemei He Removal of direct dyes from aqueous solution by oxidized starch cross-linked chitosan/silica hybrid membrane / Xuemei He, Mei Du, Hui Li, Tianchi Zhou. // International Journal of Biological Macromolecules, 82, 2016. – P. 174–181.
- Huacai Ge Synthesis and characterization of poly(maleic acid)-grafted crosslinked chitosan nanomaterial with high uptake and selectivity for Hg(II) sorption / Huacai Ge, Tingting Hua. // Carbohydrate Polymers, 153, 2016. – P. 246–252.
- Cintia S. Oliveira Pyridine derivative covalently bonded on chitosan pendant chains for textile dye removal / Cintia S. Oliveira, Claudio Airoldi // Carbohydrate Polymers, 102, 2014. – P. 38–46.
- Ahmedy Abu Naim Chemical modification of chitin by grafting with polystyrene using ammonium persulfate initiator / Ahmedy Abu Naim, Abdulganiyu Umar, Mohd Marsin Sanagi, Noraimi Basaruddin. // Carbohydrate Polymers, 98, 2013. – P.1618–1623.
- Tahia B. Mostafa An approach toward construction of tuned chitosan/polyaniline/metal hybrid nanocomposites for treatment of meat industry wastewater / Tahia B. Mostafa, Atef S. Darwish. // Chemical Engineering Journal, 243, 2014.- P. 326–339.
- Sayyah S.M. Kinetic studies and grafting mechanism for methyl aniline derivatives onto chitosan: Highly adsorptive copolymers for dye removal from aqueous solutions / Sayyah S.M., Amr A. Essawy, A.M. El-Nggar. // Reactive and Functional Polymers, 96, 2015. – P. 50–60.
- Baskar D. Effect of deacetylation time on the preparation, properties and swelling behavior of chitosan films // Baskar D. , T.S. Sampath Kumar. / Carbohydrate Polymers, 78, 2009. – P. 767–772.
- Huacai Ge Synthesis and characterization of poly(maleic acid)-grafted crosslinked chitosan nanomaterial with high uptake and selectivity for Hg(II) sorption / Huacai Ge, Tingting Hua. //Carbohydrate Polymers, 153, 2016. – P. 246–252.
